Documentation and traceability in pharma piping projects are not paperwork added after installation. They are the evidence chain that proves the installed system matches the approved material, finish, weld, inspection, and hygienic design intent. In GMP projects, a piping loop can be mechanically complete and still remain unreleasable because the documentation does not cleanly prove what was installed, how it was fabricated, what changed in the field, and whether the final assembly still meets the approved project standard.
That is why experienced engineers do not treat turnover documents as an administrative handoff. They treat them as part of project design control from the beginning. If material traceability, weld records, finish records, inspection evidence, redlines, and turnover deliverables are not defined before RFQ, the project often tries to rebuild the evidence chain later at much higher cost and with lower confidence. This guide explains what documentation and traceability really mean in pharma piping projects, which records matter most, where the evidence chain usually breaks, and how GMP teams can define the right documentation package before fabrication starts.
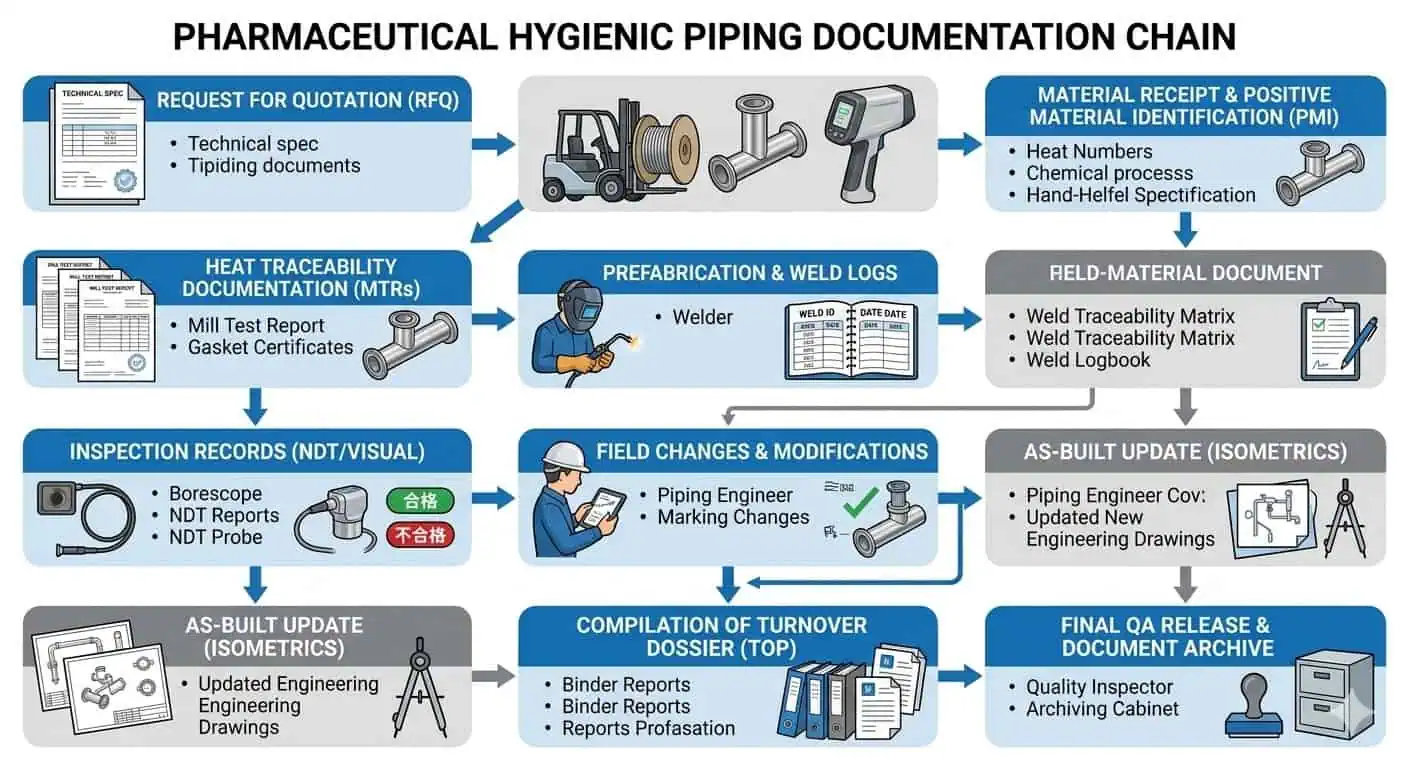
In real pharmaceutical work, documentation problems rarely appear as dramatic mechanical failures. They show up as slower QA closure, unresolved turnover comments, unclear weld lineage, mismatched spool histories, or uncertainty around whether the final installed loop still reflects the approved hygienic intent. That is why strong documentation is not a paperwork preference. It is an engineering control.
Why Documentation and Traceability Matter in Pharma Piping Projects
This Is a QA and Release Topic, Not Just an Engineering Filing Topic
In pharma piping, documentation is part of release control, not just project administration. EU GMP Chapter 4 does not describe documentation as a filing convenience. It describes good documentation as an essential part of the quality assurance system and the mechanism used to establish, control, monitor, and record activities that affect product quality. FDA logic points in the same direction: product-contact equipment must be suitable for use, and cleaning, maintenance, and equipment history must be controlled and recorded. In other words, the installed loop has to be technically right and provably right at the same time.
Typical industry case: a product-contact transfer loop is fully installed, pressure tested, and visually acceptable. Operations sees a finished piping system. QA sees an incomplete evidence chain because the final dossier still cannot clearly tie together approved material identity, weld history, and final as-built configuration. The piping is physically there, but the release logic is still unresolved.
Expert advice: if the project team treats documentation as something that starts at handover, it is already too late. In GMP piping, the release story has to be built while the loop is being designed, fabricated, inspected, and changed—not after the last clamp is tightened.
Engineering note: in GMP piping, the installed loop is only as releasable as the evidence chain that proves its final wetted condition.
Why Pharma Piping Projects Often Fail at Turnover, Not at Fabrication
Many pharma piping projects do not fail because the loop was badly built. They fail because the project cannot prove, in release-ready form, that the final installed loop still matches the approved hygienic intent. That is a different kind of failure, but in GMP work it is often the more dangerous one because it appears late, after the system already looks mechanically complete. EU GMP Chapter 4 and FDA recordkeeping logic both support the same practical conclusion: if cleaning history, component identity, weld status, and installed configuration cannot be reconstructed reliably, the system becomes harder to defend at release and later during operation.
Typical industry case: the field installation team finishes a hygienic loop and assumes the difficult work is over. Later review shows that several spool pieces were revised in the field to improve routing and slope, but the final weld map, material-location matrix, and as-built package were never fully reconciled. No one is claiming the metal is wrong. The problem is that the project can no longer prove cleanly which certified materials, weld records, and inspection results belong to the final installed arrangement. At that point, the loop is not fighting a fabrication defect. It is fighting a release-evidence defect.
Experienced project engineers therefore define the documentation chain before RFQ, not after mechanical completion. Once fabrication starts, every uncontrolled field cut, spool swap, or branch reroute becomes harder to close retroactively. The engineering lesson is simple: in GMP piping, if you wait until turnover to decide what must be traceable, the project usually ends up rebuilding the evidence chain at higher cost and lower confidence.
What the GMP and Regulatory Framework Actually Requires
The most useful regulations are the ones that change what the project team defines and records before turnover. In this topic, the core framework usually includes:
- ASME BPE, which covers materials, design, fabrication, inspections, testing, and certification logic for bioprocessing and pharmaceutical equipment.
- FDA 21 CFR 211.65, which requires product-contact surfaces to be non-reactive, non-additive, and non-absorptive.
- FDA 21 CFR 211.67, which ties equipment cleaning and maintenance to written procedures and records.
- FDA 21 CFR 211.182, which addresses equipment cleaning, maintenance, and use logs for major equipment.
- FDA 21 CFR 211.184, which covers component, container, closure, and labeling records.
- EU GMP Chapter 4, which defines documentation as an essential part of the quality assurance system and requires records to be controlled, accurate, traceable, and retained appropriately.
Research takeaway: these sources do not treat documentation as background support. They treat it as a controlled part of GMP execution. For piping teams, that means the question is never just “what records do we have?” but “do these records still prove the final wetted system clearly enough for QA release?”
What Must Be Traceable in a GMP Piping Project
Tubing, Fittings, Valves, and Other Wetted Components
Traceability should follow the wetted path, not just the largest or easiest components. Many teams manage tubing well but treat smaller fittings, diaphragms, sample valves, ferrules, gaskets, and branch components too casually. In GMP projects, that is a mistake. The smallest wetted parts are often the easiest to substitute, the hardest to track later, and the first to create QA questions during turnover.
Typical industry case: tubing traceability is complete, but the turnover package becomes weak at local assemblies because fittings, gaskets, or sample-valve internals were handled outside the main control workflow. The line looks fine in walkthrough, yet the weakest traceability link sits exactly where the hygienic assembly is most complex.
Expert advice: if a component is important enough to be specified as part of the hygienic design, it is important enough to be traceable as part of the turnover logic. In practice, teams get better QA closure when they define traceability around the full wetted path instead of around tubing alone.
Material Certificates, Heat Numbers, and Lot Identity
Material traceability is only useful when the installed location can be tied cleanly back to the original record set. Material test reports, EN 10204 3.1 certificates, heat numbers, and receiving records all matter, but only if the project preserves the link between supplier identity and installed spool, fitting, or assembly location. A pile of certificates without a clean location matrix is not a strong traceability package.
Typical industry case: the project has the correct material certificates in hand, but after field cutting, re-spooling, and local rerouting, the turnover team cannot quickly prove which heat numbers ended up in which installed branch sections. The problem is not missing paper. The problem is that the paper no longer maps clearly to the installed reality.
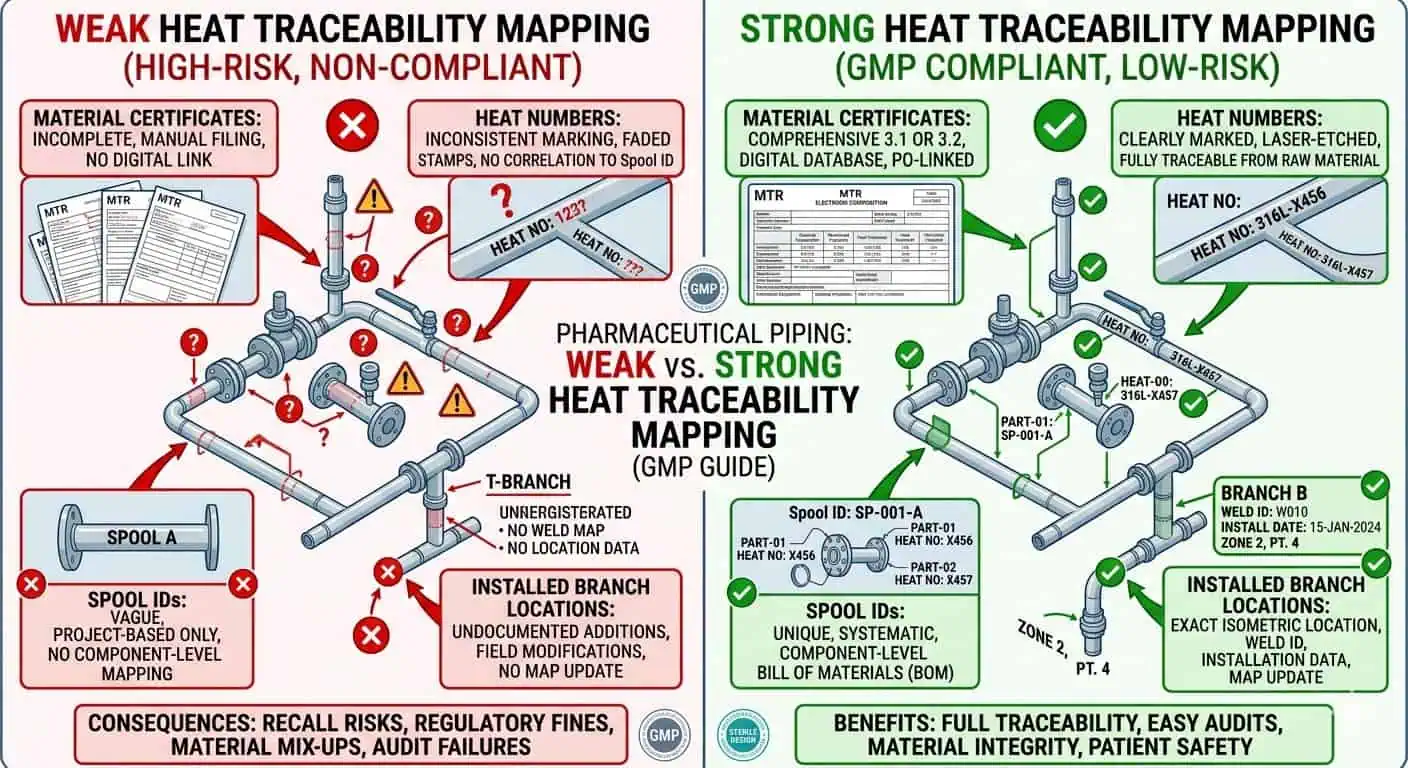
Expert advice: in high-consequence product-contact systems, define the traceability matrix early instead of allowing material records and spool records to evolve separately. Once field modifications begin, rebuilding that link becomes slower and less reliable.
Weld Maps, Orbital Weld Logs, and Inspection Status
Weld documentation is often the most operationally important part of the piping evidence chain. In hygienic pharma systems, weld numbers, machine settings, operator identity, dates, inspection status, repair history, and acceptance status are often more decisive than teams expect. That is because the weld is where material identity, fabrication quality, inspection evidence, and final hygienic performance converge. ASME BPE is useful here not because it creates paperwork for its own sake, but because it treats fabrication, examination, testing, and certification as one connected control framework. In pharma projects, weld logs are therefore not just shop records. They are release records.
Typical engineering case: a loop looks acceptable on walkthrough, but QA still flags it because the weld map and the final spool revision are no longer aligned after field changes. The installed welds may be physically acceptable, but the documentation set no longer proves which records belong to which final weld locations.
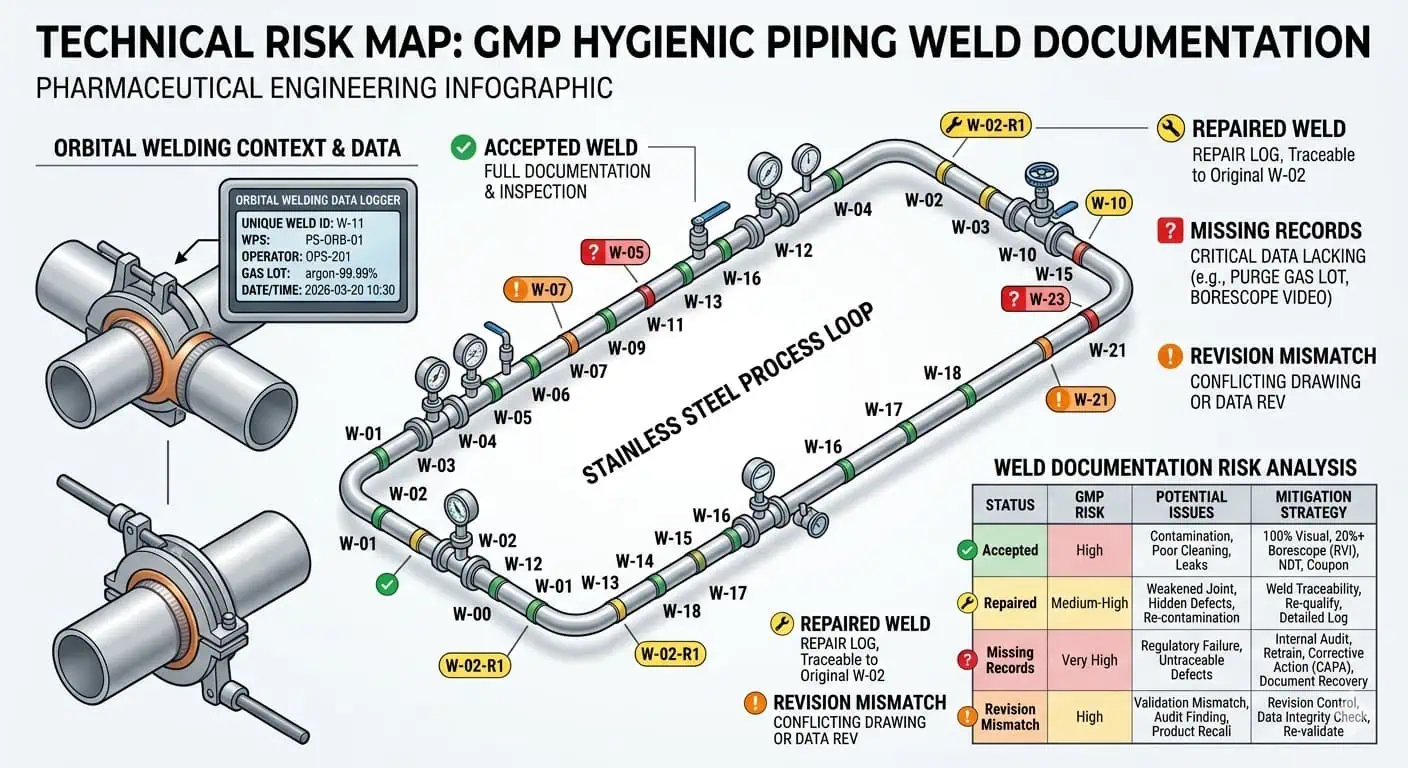
Expert advice: do not manage weld records as a fabrication-only dataset. Manage them as a release dataset that must still make sense after field changes, spool substitution, and final as-built closure.
Surface Finish, Electropolish, Passivation, and Related Records
Finish requirements matter only when the project can prove what finish was required, what was supplied, and how the final wetted condition was preserved. In more demanding GMP systems, the finish decision is not separate from documentation. If the project differentiates between standard 316L and electropolished 316L, or requires passivation and post-fabrication treatment evidence, those requirements need their own record path in the turnover dossier.
Typical industry case: the tubing grade was acceptable, but the release discussion shifted to whether the final surface condition of several product-contact spool pieces still matched the specified hygienic expectation after fabrication and rework. The alloy was not the weak point. The documented finish condition was.
Expert advice: define finish-related documentation before fabrication starts. If a project decides late that surface condition matters more than expected, the missing records are often much harder to rebuild after fabrication than the material certificates were.
Redlines, As-Builts, and Field Changes
Field changes are where traceability chains most often break. A branch is moved for access. A spool is remade to suit actual fit-up. An instrument point is revised. A valve orientation changes. None of these actions are automatically fatal, but each one creates documentation risk if the redline-to-as-built process is weak. One of the most important documentation principles in GMP practice is that records should be created at the time the work happens. That matters because reconstructed records are almost always weaker than contemporaneous ones.
Industry case: a GMP skid tie-in was changed in the field to improve routing and maintenance access. The change itself was reasonable. The problem was that the updated weld references, spool references, and as-built drawings lagged behind the installation. By turnover, the team was trying to reconcile two technically similar but documentationally inconsistent versions of the same loop.
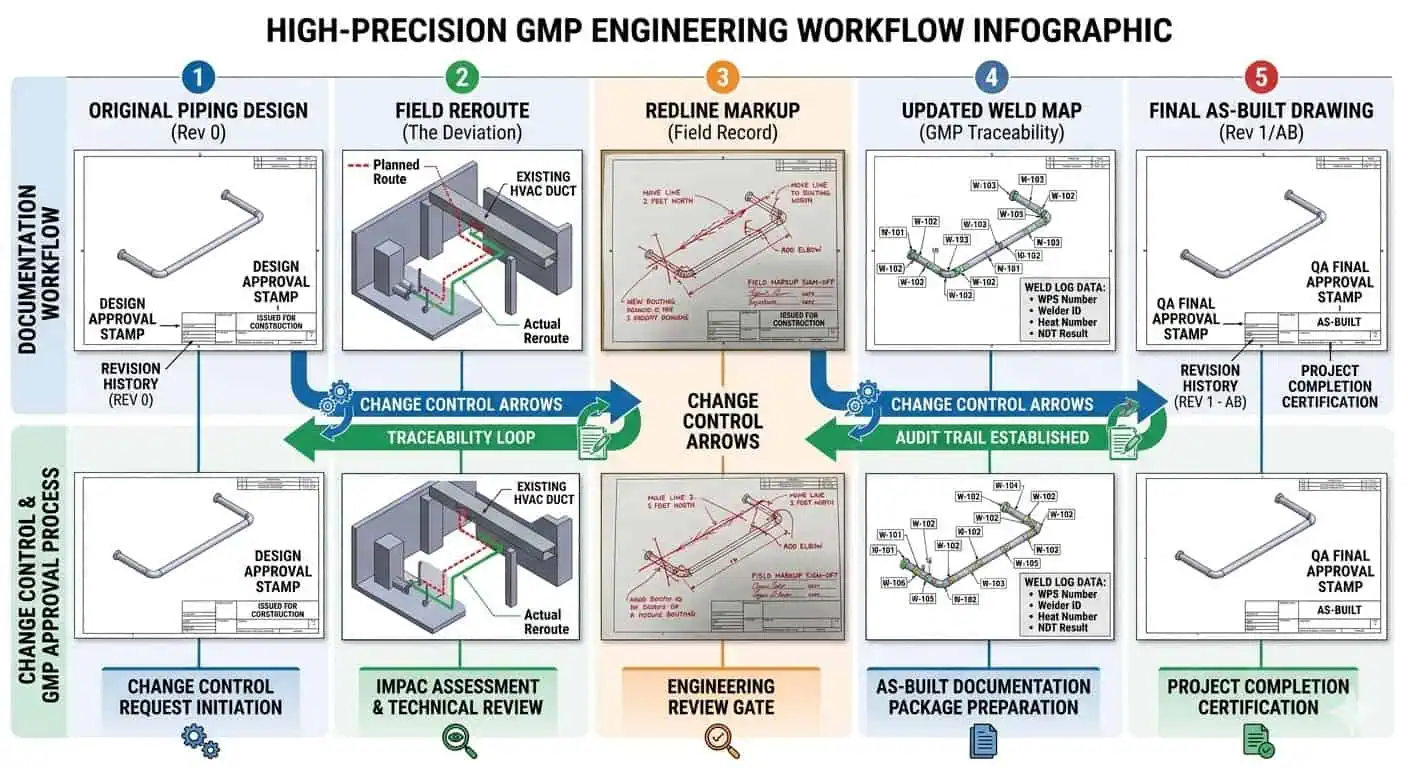
Expert advice: field reroutes are not the real problem. Unclosed evidence from field reroutes is the real problem. The project should treat redlines and as-builts as active control tools, not end-stage drafting tasks.
The Core Documentation Package GMP Teams Usually Expect
Material Documentation
Material documentation proves that the approved wetted materials actually entered the system. At minimum, this usually means material certificates, heat traceability, receiving control, and some form of location mapping that ties installed components to the approved material record set. The stronger the hygienic or product-contact consequence, the less acceptable it becomes to leave this relationship vague.
Fabrication and Weld Documentation
Fabrication documentation proves how the system became what it is. This is where weld maps, orbital weld logs, weld inspection results, repair histories, boroscope records where required, and fabrication status tracking usually sit. The point is not to create a thick dossier for its own sake. The point is to keep the installed loop auditable and technically defensible.
Surface and Treatment Documentation
Surface records matter whenever finish or post-fabrication treatment is part of the hygienic requirement. If the project specifies electropolish, passivation, or a defined finish target, the turnover package should show how that requirement was applied and how the relevant parts were identified. Otherwise, the project risks having a finish requirement on paper but no strong evidence that the installed surface still matches it.
Testing and Turnover Documentation
Testing and turnover records turn fabrication history into release evidence. Pressure testing, leak testing, inspection closure, punch-list closeout, and final dossier assembly are where the project demonstrates that the fabricated loop was not only built, but also verified and closed in a way that supports GMP operation.
Typical industry case: the project has all the individual records somewhere, but turnover still stalls because the package is not structured in the way QA needs to review and approve the loop. This is not a missing-document problem alone. It is a dossier-logic problem.

Expert advice: if turnover documents are being assembled for the first time only after mechanical completion, the project is already late from a documentation-control perspective.
Change Control and Deviation Records
Change documentation is what keeps the final evidence chain believable. The project should be able to show what changed, who approved it, what inspection or rework followed, and how the final as-built condition absorbed that change. Without that, even technically reasonable field changes can create unnecessary QA hesitation.
| Documentation Set | What It Proves | Where It Commonly Fails |
|---|---|---|
| Material certificates and traceability matrix | The approved wetted materials were actually installed | Location mapping becomes unclear after cutting or field rerouting |
| Weld maps and weld logs | The final welds were completed and inspected under control | Weld numbers stop matching final spool revisions |
| Finish / EP / passivation records | The specified surface condition was achieved and preserved | Rework or late finish changes are not fully documented |
| Redlines and as-builts | The installed loop matches the final approved configuration | Field changes happen faster than drawing closure |
| Turnover dossier | The complete evidence chain is ready for QA review | Records exist, but not in a release-ready structure |
Where Documentation Chains Usually Break
Material Was Correct, but the Traceability Matrix Was Weak
One of the most common failures is not missing material, but weak material-location mapping. Teams sometimes assume that having the certificates is enough. It is not enough if the project cannot quickly prove where the certified material ended up after cutting, fabrication, spooling, and field change.
Welds Were Acceptable, but the Weld Record Set Was Incomplete
A loop can be technically sound and still fail documentation closure because the weld record set is incomplete. Missing weld status, unclear repair history, or misaligned weld numbering usually creates more delay than teams expect. In GMP piping, weld evidence is not just a fabrication tool. It is part of release readiness.
Field Changes Happened Faster Than the Documentation Update
Documentation often falls behind the project at exactly the moment the project becomes most dynamic. Late-stage routing changes, tie-in revisions, field fit-up adjustments, and added branches often occur under schedule pressure. If the document set does not keep up, the final turnover package becomes a reconstruction exercise instead of a controlled handoff.
The Smallest Components Created the Biggest QA Problem
Small wetted parts create disproportionate documentation risk. Fittings, sample components, diaphragms, gaskets, and instrument-related wetted parts are often where the cleanest evidence chain breaks first. Teams tend to think in terms of pipe runs, but QA often experiences the project in terms of unresolved local assemblies.
Expert lesson from field closure work: the more local and replaceable the assembly, the less safe it is to assume its records will take care of themselves.
How Engineers Should Build Documentation Requirements Before RFQ
Define What Must Be Traceable
Before RFQ, define which wetted components must remain traceable through installation and turnover. If only tubing is controlled and the smaller wetted parts are left vague, the evidence chain will usually be weakest where the hygienic assembly is most complex.
Define What Must Be Recorded During Fabrication
Projects should define what must be recorded while the work is happening, not only what must exist at the end. Weld logs, inspection points, finish-related records, passivation evidence where relevant, and field-change updates should be part of the defined project flow.
Define What QA Must Receive at Turnover
The turnover package should be defined as a release deliverable, not a vague collection of whatever the contractor can assemble later. If QA expects a specific dossier structure, traceability matrix, or record format, the project should define that while it still has time to generate those records correctly.
Define What Must Be Maintained After Startup
The documentation chain should survive into operation, not stop at handover. Equipment logs, maintenance records, replacement-part traceability, and controlled updates to the installed system are all part of keeping the original evidence chain alive. A release-ready system can become much harder to defend later if lifecycle records are weak.
The strongest project advice is simple: define the evidence chain before procurement. Do not wait until fabrication is underway to decide whether heat traceability, finish records, weld status, or field-change documentation will matter. By that point, the project usually ends up collecting documents reactively instead of controlling them deliberately. This is one of the clearest practical lessons from GMP turnover work: projects that define the record set before RFQ usually close faster and with fewer QA surprises than projects that try to reconstruct it later.
How This Connects to Design, Material, and Fitting Selection
Material Selection Without Documentation Control Is Not Closed Risk
A good material decision only becomes a safe project decision when it is documented cleanly. For the broader material-side logic, connect this page with Pharmaceutical Hygienic Piping Material Selection.
Finish Requirements Only Matter If They Are Proved
Electropolish, finish targets, and post-treatment expectations do not close risk unless the records prove them. Link this topic with Electropolished 316L vs Standard 316L Stainless Steel.
Fittings and Sample Assemblies Need Their Own Evidence Chain
The smaller and more local the assembly becomes, the more likely its documentation will become the weakest part of the turnover package. Link this topic with Sanitary Fitting Selection for GMP Facilities.
As-Built Documentation Must Reflect the Real Cleanability Risk
Documentation should not only prove what was installed. It should also reflect the final local hygienic reality of the system. For that logic, connect this page with CIP Design Considerations for Hygienic Piping.
Common Mistakes in Documentation and Traceability for Pharma Piping Projects
Treating Documentation as a Turnover Activity Instead of a Design Input
Projects that define the documentation package late usually end up rebuilding records under schedule pressure.
Tracking Tubing but Not Tracking Small Wetted Components
The weakest traceability link often appears in fittings, seals, diaphragms, or local sample assemblies.
Keeping Weld Data Separate from Line-Level Release Logic
Weld records are much less useful if they do not map clearly to the final installed loop and its release status.
Letting Redlines Drift Away from the Installed Reality
When field changes move faster than controlled updates, the turnover package becomes harder to trust.
Assuming QA Will Accept It Later
In GMP piping, missing evidence is rarely easier to fix at the end of the project than during controlled execution.
Over-Collecting Documents but Under-Defining What Actually Matters
Large document volumes do not automatically create a strong turnover package. What matters is whether the record set proves the installed hygienic system clearly enough for release and later lifecycle control.
Practical Checklist: What to Ask Before You Release a Pharma Piping System
Before Fabrication Starts
- What wetted components must be traceable?
- What records must be created during fabrication?
- What weld, finish, and inspection evidence will QA require later?
- How will field changes be absorbed into controlled records?
Before Mechanical Completion
- Are redlines current?
- Do installed components map clearly to approved material records?
- Are weld status and repair history closed?
- Are finish and treatment records complete where required?
Before QA Release
- Does the turnover package match the actual installed loop?
- Are any local assemblies still unresolved in traceability or inspection records?
- Can the project prove the final as-built condition clearly enough for release?
After Startup
- How will equipment logs and maintenance records continue?
- How will replacement parts remain traceable?
- How will future changes be tied back into the original evidence chain?
| Project Stage | Key Documentation Question | What Good Control Looks Like |
|---|---|---|
| Before RFQ | What must be traceable and deliverable? | The record set is defined, not assumed |
| During fabrication | Are records being created in real time? | Weld, material, finish, and change records stay current |
| Before turnover | Does the dossier match the installed reality? | As-built, traceability, and inspection closure align |
| After startup | Will the evidence chain survive lifecycle changes? | Maintenance and replacement records continue under control |
Conclusion: In Pharma Piping, Documentation Proves the System You Think You Built Is the System You Actually Installed
Good documentation and traceability do not add value after the project. They protect the project from becoming unreleasable at the exact moment it should be ready to run. In pharma piping, the installed system is only as acceptable as the evidence chain that proves its material, fabrication, finish, inspection, and changes were controlled.
The strongest projects do not try to reconstruct this evidence chain at turnover. They define it before RFQ, maintain it during fabrication, and close it before release. For the full pharma topic cluster, connect this article with Pharmaceutical Hygienic Piping Material Selection, Electropolished 316L vs Standard 316L Stainless Steel, Sanitary Fitting Selection for GMP Facilities, and CIP Design Considerations for Hygienic Piping.
FAQ
What documentation is required for pharma piping projects?
The exact package depends on project scope, but it usually includes material certificates, traceability records, weld documentation, inspection records, finish or treatment evidence where required, redlines, as-builts, and turnover records that support QA release.
What traceability is required in GMP piping?
Traceability should follow the wetted path. That often means tubing, fittings, valves, local assemblies, and other critical product-contact components must be traceable back to approved records in a way that still matches the installed condition.
Why do pharma piping projects fail at turnover?
They often fail because the evidence chain is incomplete. The installation may be physically acceptable, but missing or weak material, weld, finish, or as-built records can still delay QA release.
Do fittings and gaskets need traceability in GMP projects?
In many critical hygienic applications, yes. Smaller wetted components are often where the weakest documentation link appears, so they should not be treated as casual consumables if they influence hygienic performance or release risk.
Are weld logs required for pharma hygienic piping?
Projects commonly rely on weld maps and weld documentation because they are central to proving fabrication and inspection status. The exact format varies by project, but weak weld documentation is one of the most common turnover problems.
What is the difference between as-built records and turnover documents?
As-built records show the final installed configuration. Turnover documents are the broader evidence package used to support release, including traceability, inspection, testing, and related project records.




