Sanitary fitting selection for GMP facilities is not just a hardware choice. It is a cleanability, drainability, seal-control, documentation, and contamination-control decision. In pharmaceutical piping, the wrong fitting may still look sanitary in a catalog but create a dead leg, trap rinse water, complicate inspection, weaken traceability, or introduce an unnecessary elastomer-controlled risk at the exact point where the process is least tolerant of it.
That is why engineers do not select hygienic fittings by connection style alone. A fitting in a GMP facility has to be judged by where it will be installed, how it will be cleaned, whether it will be opened routinely, what seals it introduces, what finish and weld condition it requires, and how easily QA can trace and release the final assembly. A clamp ferrule, a welded branch, a sample connection, and a diaphragm valve interface can all be “correct” in one location and wrong in another.
This guide explains how engineers choose sanitary fittings for GMP facilities, where clamp-type fittings make sense, where more permanent welded connections are usually the safer engineering choice, how local geometry changes the risk, and why fittings should always be specified together with material, finish, seal, and documentation requirements rather than as independent purchasing items. In real GMP projects, fitting decisions rarely fail because the team picked a visibly “cheap” part. They fail because the connection looked acceptable on a line list but created the wrong hygienic behavior once installed in a sample branch, valve cluster, or SIP/CIP-exposed local assembly.
Why Sanitary Fitting Selection in GMP Facilities Is a High-Risk Engineering Decision
This Is Not Just a Hardware Choice
In GMP environments, a sanitary fitting is part of the product-contact system, not just a connector between pipe sections. It influences cleanability, drainability, inspection access, seal reliability, and the evidence chain needed for QA release. That is why fitting choice belongs in the same technical conversation as material, finish, weld quality, and cleaning strategy.
FDA 21 CFR 211.65 is useful here because it requires product-contact surfaces to be non-reactive, non-additive, and non-absorptive. That requirement sounds simple, but in practice it means the fitting has to be evaluated as an installed wetted assembly, not as an isolated catalog component.
Engineering note: in GMP piping, the fitting only succeeds when the installed wetted assembly meets the hygienic intent of the process, the inspection method, and the documentation package together.
Why GMP Facilities Cannot Choose Fittings Like General Sanitary Plants
Pharmaceutical hygienic fitting selection is usually held to a tighter standard than general sanitary service because the consequence of poor cleanability or weak traceability is higher. In food or general sanitary plants, a fitting may be judged mainly by serviceability and corrosion resistance. In GMP facilities, the same fitting may also have to satisfy a tighter contamination-control logic, stronger documentation expectations, and stricter release scrutiny.
Typical industry case: a fitting family that performs acceptably in a general sanitary application may still become problematic in a GMP transfer assembly because the real issue is not bulk corrosion or pressure containment. It is that the seal interface, branch geometry, or documentation package does not support the hygienic and QA burden of the pharmaceutical environment. This is especially visible in sample branches and local distribution points, where one seemingly minor connection can dominate the whole investigation when a deviation occurs.
What Standards and GMP Logic Actually Affect the Choice
The most useful standards are the ones that change what the engineer writes into the RFQ and inspects before release. In this topic, the most relevant references usually include:
- ASME BPE for materials, design, fabrication, inspection, testing, and certification logic in bioprocessing and pharmaceutical systems.
- FDA 21 CFR 211.65 for product-contact surface suitability.
- FDA 21 CFR 211.67 for cleaning, maintenance, disassembly/reassembly procedures, inspection before use, and related records.
- ASTM A270 for sanitary tubing, including the fact that pharmaceutical quality may be specified as a supplementary requirement.
- EU GMP Annex 1 for contamination-control strategy and risk-managed design expectations in more demanding sterile-related settings.
Those frameworks matter because they push fitting selection away from “what connects easily” and toward “what creates the most controlled hygienic outcome in the actual process area.”
What Engineers Really Evaluate When Selecting Sanitary Fittings
Cleanability
The first question is whether the fitting helps or hurts cleanability at the exact point where it is installed. Internal geometry, crevice tendency, branch shape, seal position, and the relationship between the fitting and adjacent welds all affect whether the assembly can be cleaned reliably. In GMP piping, the fitting is often not judged by how the main run cleans. It is judged by whether the hardest local assembly still cleans predictably.
Typical engineering example: a transfer loop may look acceptable in the main run, yet the same system keeps producing concern at one sample tee because the fitting arrangement created a local flush weakness. The lesson is not that the line is generally wrong. The lesson is that cleanability is decided at the worst local assembly, not the most obvious straight run.
Drainability
A fitting that does not drain well can create a repeated hygienic problem even when the rest of the loop appears acceptable. Residual rinse water, retained product, or trapped condensate at a local branch or fitting body can distort the next cleaning cycle and complicate investigation. This is one reason why fitting orientation matters as much as fitting type.
In practical startup work, one of the most common surprises is that the overall line slope looks acceptable on the isometric, but one reducer, sample connection, or branch body still becomes the true low point after installation. That local retention then shows up later as inconsistent post-clean inspection, unexplained residue, or recurring concern around one repeated assembly.
Seal Control
Many sanitary fitting risks are actually seal risks. A clamp-style fitting may be mechanically sound but still create hygienic instability if the gasket material is wrong, the compression is uneven, or the seal interface is repeatedly opened without strong control. In pharmaceutical systems, a good metal choice cannot rescue a weak elastomer strategy.
Expert advice: the more a fitting depends on a replaceable seal for hygienic success, the more that seal should be managed as a controlled wetted component rather than as a generic consumable.
Inspection and Maintenance Access
A good hygienic fitting has to support the inspection reality of the facility, not just the installation preference of the contractor. Some assemblies benefit from controlled disassembly and gasket access. Others are safer when the number of disassembly interfaces is minimized. The right choice depends on how the assembly will actually be verified, cleaned, and maintained through its lifecycle.
This is where facilities often diverge. A pilot or development environment may value controlled modular access more strongly. A fixed high-consequence production system may value reduced interface count more strongly. The mistake is not choosing one philosophy or the other. The mistake is applying the same philosophy everywhere without considering local hygienic consequence.
Material, Finish, and Documentation Compatibility
Fittings should never be specified independently from material, finish, and documentation requirements. In GMP facilities, the fitting material, wetted finish, certificate package, and traceability burden are part of the same technical decision. A correct fitting geometry with the wrong finish expectation or weak records can still become a release problem.
For the broader material and finish logic, connect this article with Pharmaceutical Hygienic Piping Material Selection and Electropolished 316L vs Standard 316L Stainless Steel.
The Main Sanitary Fitting Types Used in GMP Facilities
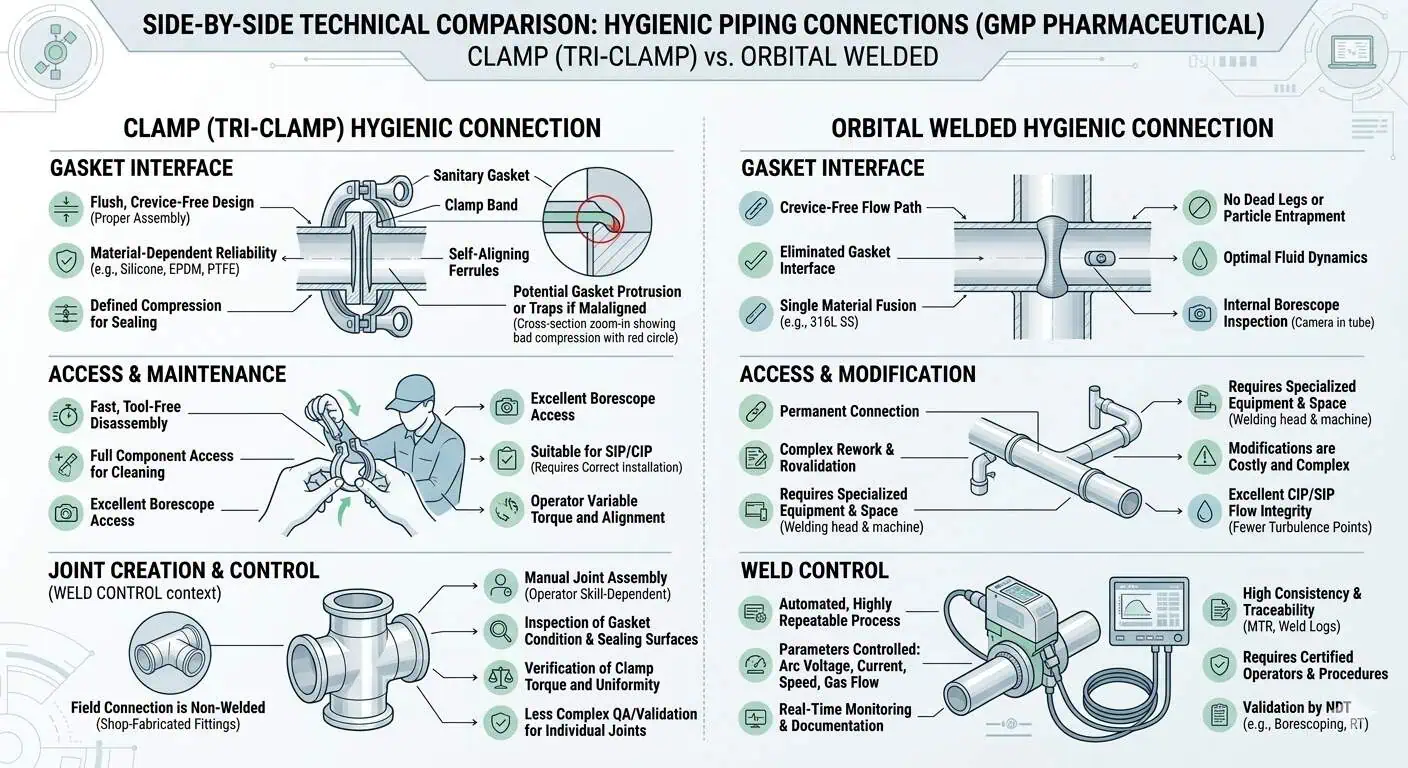
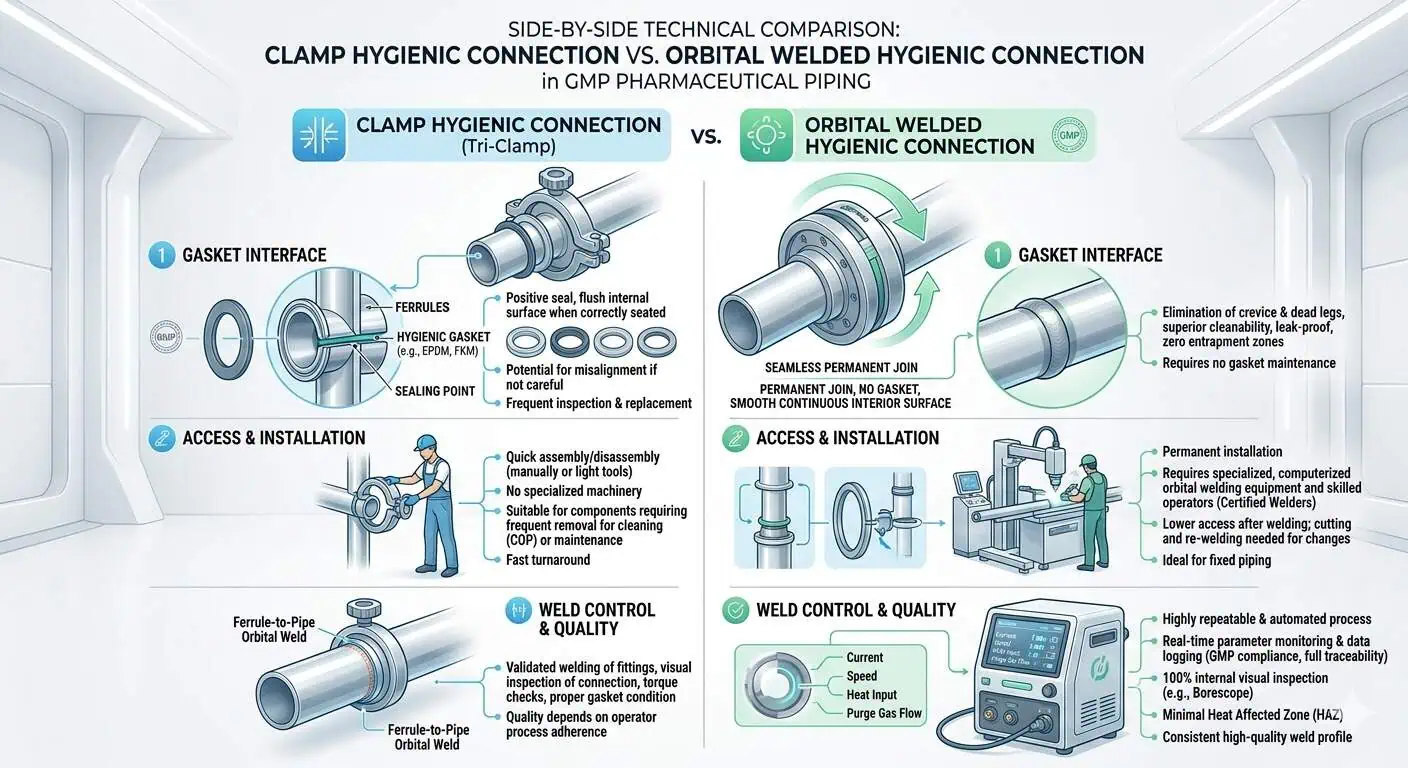
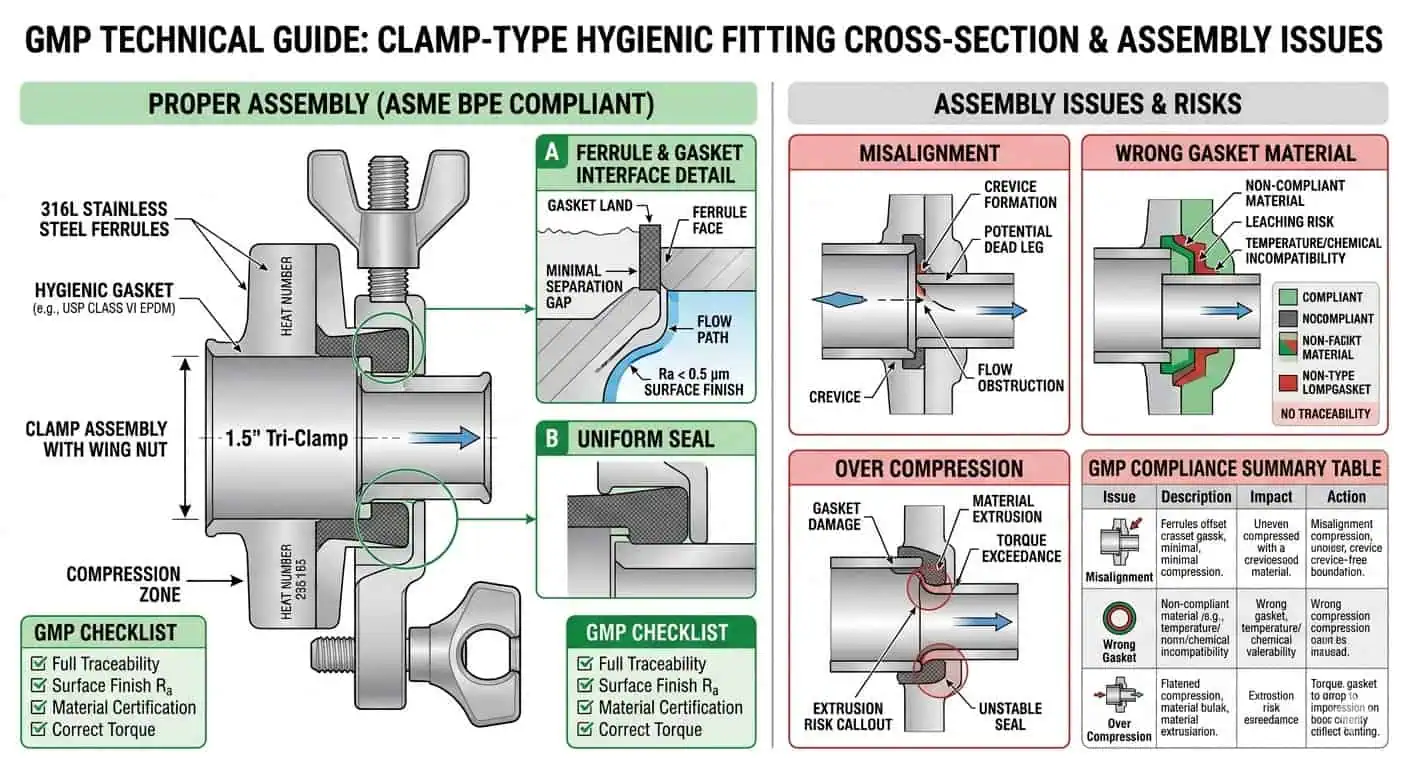
Clamp-Type Hygienic Connections
Clamp-type hygienic fittings are useful where controlled access, gasket replacement, modularity, or inspection convenience are real project needs. Their advantage is not only speed. It is that they allow controlled opening, local component replacement, and flexibility in assemblies that would be impractical to cut and reweld frequently.
The risk, however, is equally clear: every clamp-style interface introduces a controlled seal, a compression condition, and an alignment requirement. If that interface is placed in the wrong location or handled loosely in operation, it can become the hygienic weak point of the assembly. This is why clamp connections in GMP systems should be justified by actual lifecycle benefit, not by habit alone.
Orbital Welded Connections
Orbital welded connections are often preferred where the process should minimize repeated disassembly interfaces and reduce seal-count risk. In fixed product-contact GMP systems, a well-controlled welded connection can remove a layer of assembly variability that clamp-style fittings necessarily retain. That does not make welding automatically better everywhere. It means the tradeoff is usually between access and control, not between good and bad hardware.
Expert advice: choose the permanently welded option when the location is high consequence, relatively stable, and does not benefit enough from routine opening to justify the added seal interface.
Threaded Connections in GMP Environments
Threaded connections are generally not the preferred answer for critical product-contact pharmaceutical hygienic piping. In lower-risk utility or non-product-contact service they may still appear, but inside critical hygienic assemblies they usually introduce geometry and cleanability questions that are harder to defend in a GMP setting. The issue is not whether a thread can physically hold pressure. It is whether the resulting interface supports the hygienic and documentation target of the project.
Diaphragm Valve and Sample Assembly Interfaces
Some of the highest-risk fitting decisions in GMP facilities sit around diaphragm valves, sample assemblies, and branch-heavy local manifolds. These zones combine weld density, seal count, local stagnation risk, and higher inspection sensitivity. In practice, the wrong fitting choice here does not usually fail dramatically. It fails by becoming the assembly that keeps raising hygienic questions during qualification, inspection, or maintenance.
Typical engineering example: a manifold may meet the nominal line-list requirement yet still become difficult to release because the fitting and branch package around one diaphragm valve or sample point creates the local inspection burden that the rest of the skid does not.
Special Transition Fittings and Instrument Connections
Reducers, tees, instrument ports, sensor fittings, and transition pieces deserve more review than their size suggests. They often look secondary on the drawing, but in service they can dominate local cleanability and drainability. A small transition fitting in the wrong orientation can create more GMP risk than a much longer straight run of correctly installed tube.
How to Choose the Right Fitting by GMP Process Area
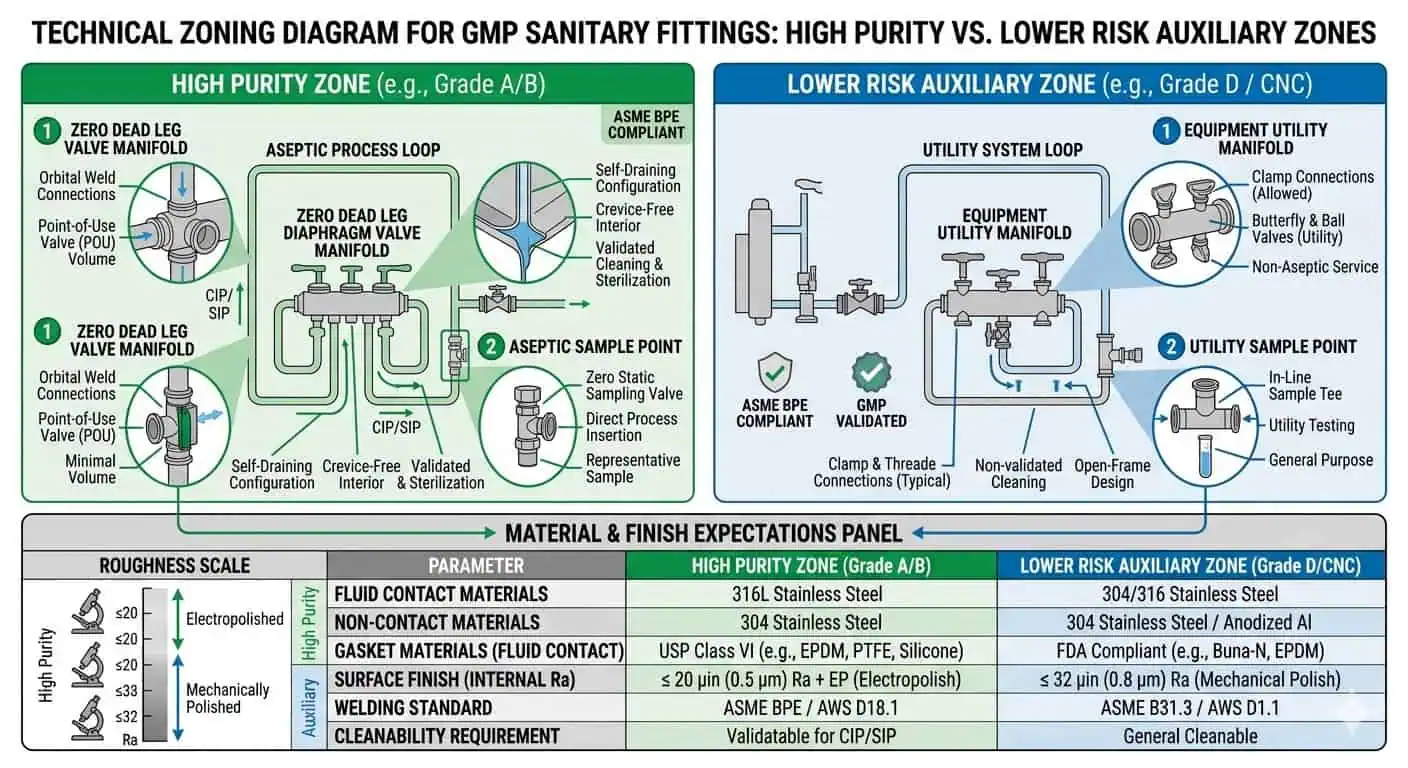
High-Purity Product-Contact Lines
In high-purity product-contact lines, fitting selection should favor the most controlled hygienic outcome, not the most convenient installation method. That often means minimizing unnecessary seal interfaces, dead-leg opportunities, and uncontrolled local complexity. Where a connection will remain fixed for long periods and the hygienic consequence is high, the balance often shifts toward more permanent solutions.
Sampling Points and Branch Connections
Sample points and small branch connections are where fitting selection errors become visible fastest. They combine local flow weakness, branch geometry sensitivity, seal risk, and repeated operator interaction. If a project wants to know where to spend more design attention, the answer is usually not the easy straight run. It is the sample point, branch take-off, or local manifold that is hardest to clean and easiest to overlook.
Industry case pattern: the main loop appears acceptable in qualification, but one branch repeatedly draws attention because the selected fitting strategy created a local hygienic burden that was never obvious at the main line level.
Valve Manifolds and Distribution Assemblies
As assembly complexity increases, fitting choice becomes more about system behavior than component preference. In valve manifolds and distribution areas, the engineer has to review weld density, disassembly need, seal count, inspection burden, and local dead-leg risk together. A fitting that seems acceptable as a stand-alone component may become the wrong choice once installed inside a branch-dense manifold.
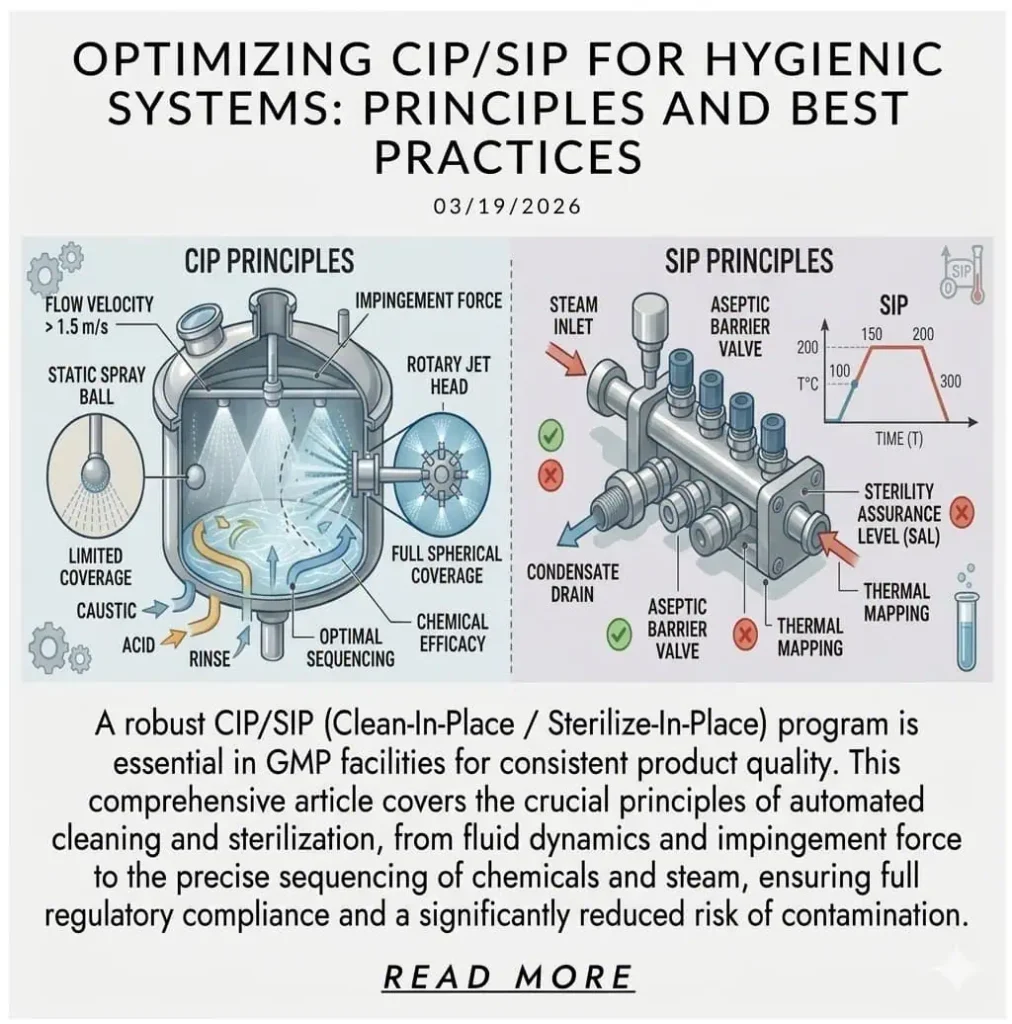
CIP/SIP-Exposed Assemblies
CIP/SIP-exposed assemblies should be reviewed as thermal-chemical systems, not only as product-contact systems. Repeated hot water, clean steam, caustic, acid, or sanitizer cycles can change how seals age, how interfaces behave, and how quickly a local assembly becomes maintenance-sensitive. In these areas, a fitting choice that seems acceptable at installation may prove weak after repeated cleaning and sterilization cycles.
Lower-Risk Auxiliary or Noncritical Service
Not every facility area carries the same hygienic consequence. Lower-risk auxiliary zones can sometimes tolerate fitting strategies that would be too loose for critical product-contact service. The mistake is not using a less demanding approach in a genuinely lower-risk zone. The mistake is letting that lower standard migrate into the wrong process area without a conscious engineering boundary.
How Sanitary Fittings Create or Prevent GMP Risk
Dead Legs and Branch Geometry
One of the most important fitting-related GMP risks is the creation of dead-leg behavior at the assembly level. A fitting may be dimensionally correct and still create a local hygienic weakness if the branch is too long, the take-off direction is wrong, or the fitting body creates a poor-exchange zone. This is why fitting selection must stay tied to branch geometry rather than being handled as a purely mechanical choice.
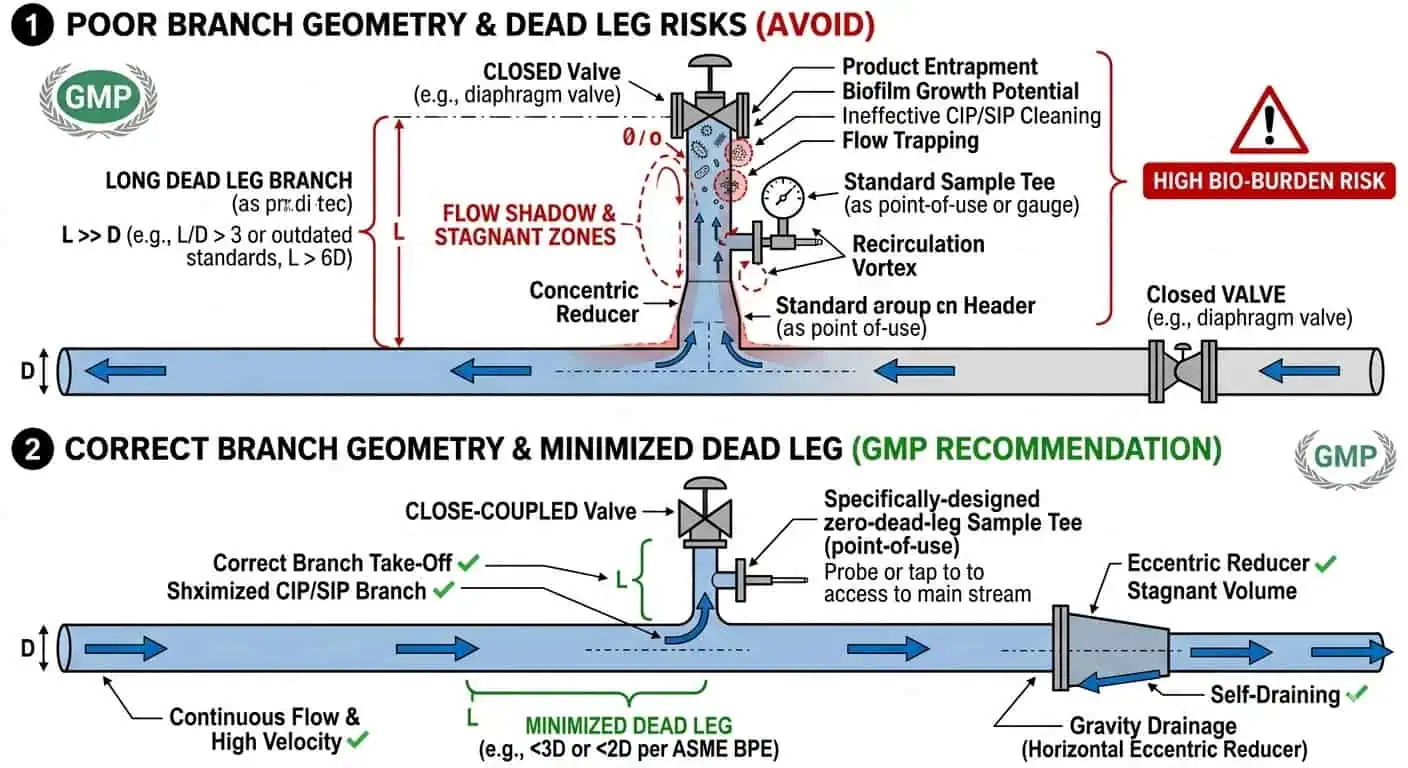
Poor Drainback and Low-Point Retention
Local retention is often a fitting problem before it becomes a system problem. A line may appear sloped correctly overall while one fitting body, reducer, or branch arrangement still traps fluid after cleaning. In real troubleshooting work, this is one of the most common reasons a local assembly behaves worse than the rest of the loop.
Gasket-Controlled Risk
The more a connection depends on a gasket or similar seal interface, the more that interface has to be treated as a controlled GMP component. Material compatibility, replacement control, compression history, and local reassembly practice all matter. Many hygienic deviations that appear to be “fitting problems” are really uncontrolled seal problems at the fitting interface.
False Confidence from Sanitary-Looking Hardware
A polished hygienic-looking fitting is not automatically a GMP-ready fitting. The catalog style can create false confidence if the assembly still suffers from poor local geometry, difficult drainback, or weak documentation. In pharmaceutical projects, appearance should never outrank assembly behavior.
Typical Industry Case: The Fitting Was Correct on Paper but Wrong in the Assembly
This is one of the most realistic fitting failures in GMP work. The selected fitting met the nominal material and connection requirement, but once installed in a branch or sample assembly it created a local cleanability problem. The lesson is not that the part was “bad.” The lesson is that the part was judged as a component instead of as a hygienic assembly element.
Materials and Finish Requirements for GMP Sanitary Fittings
Why Alloy Selection Still Matters
Fittings in GMP facilities are often specified around 316L for the same reason tubing and product-contact assemblies are: it provides a practical balance of corrosion margin, weldability, and industry acceptance in higher-purity service. But fitting selection does not end with the base alloy. The same alloy can still produce very different hygienic outcomes depending on finish, weld integration, and seal strategy.
Why Finish Can Matter More Than Buyers Expect
Finish matters because fittings often sit at the exact locations where local surface condition becomes more consequential. Branches, ferrules, small local assemblies, and inspection-sensitive product-contact points may justify tighter finish expectations than the rest of the system. The better question is not “Do all fittings need the highest finish?” It is “Which fittings sit in the assemblies where finish changes the real hygienic risk?”
Why the Same Alloy Does Not Guarantee the Same Hygienic Result
A 316L fitting with poor surface control, weak weld integration, or the wrong gasket is not equivalent to a 316L fitting inside a well-controlled hygienic assembly. In other words, the alloy family is only one layer of the decision. The installed wetted condition is what actually matters during qualification and operation.
Expert Advice: Never Specify the Fitting Without Specifying the Wetted Condition
The strongest fitting specifications define the wetted condition, not just the base component. That means material, finish, gasket or seal requirement, relevant documentation, and any inspection expectation should be written together. A brief fitting description without those details is usually not strong enough for GMP project control.
Documentation and Traceability Requirements for GMP Fittings
What QA Typically Wants to See
For GMP fittings, QA usually wants proof that the installed component matches the approved technical and hygienic intent. Depending on project scope, that may include material certificates, heat traceability, surface-finish records where relevant, inspection records, weld maps for integrated assemblies, and any related turnover evidence needed to support release.
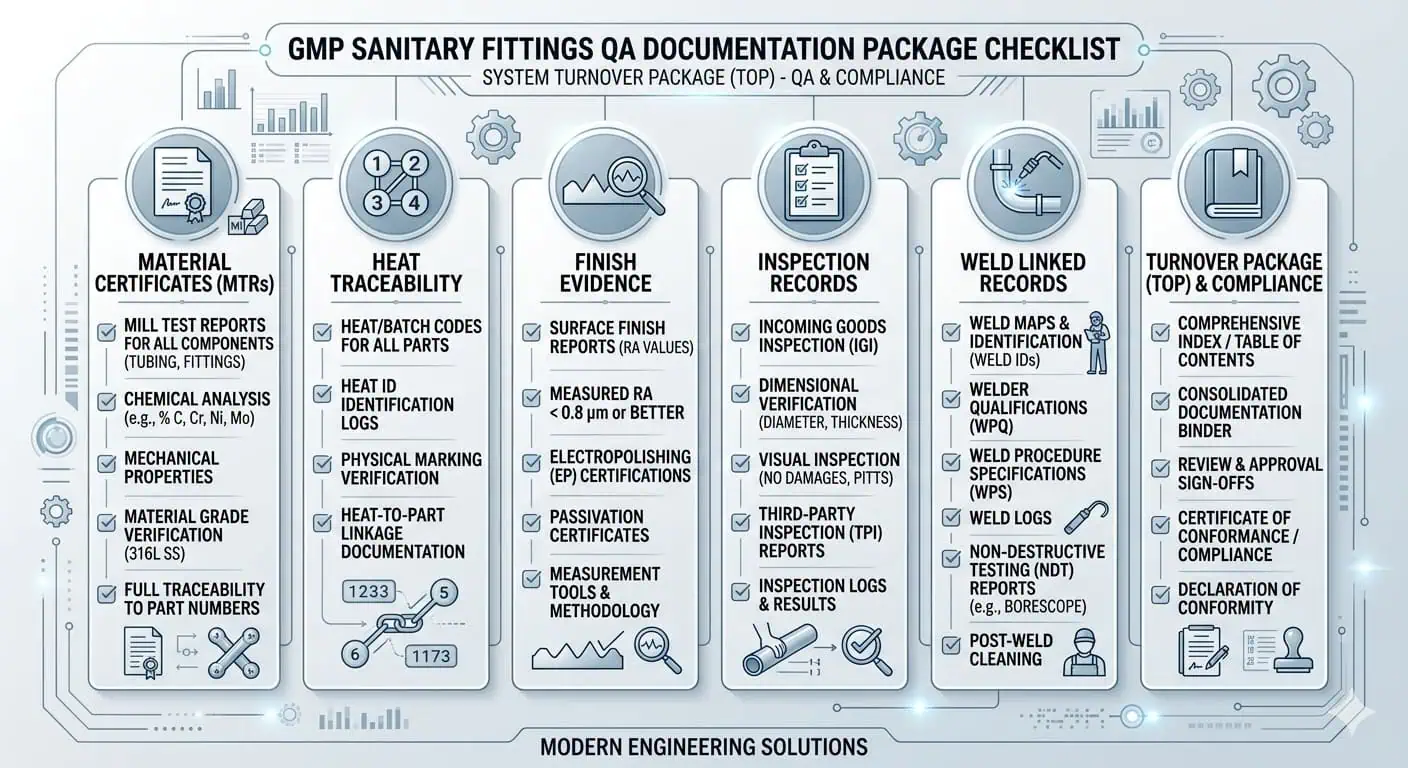
| Documentation Item | Why It Matters |
|---|---|
| Material certificates / MTC | Confirms the specified wetted material was supplied |
| Heat traceability | Links installed fittings back to the approved material lot |
| Finish evidence | Supports the specified wetted surface expectation where relevant |
| Inspection records | Shows the installed fitting or assembly was reviewed before release |
| Weld-linked records | Important when the fitting is part of an integrated fabricated assembly |
Why Fittings Become a Turnover Bottleneck
Fittings become a turnover bottleneck because they are numerous, easy to substitute, and often treated too casually during procurement. A project may manage tubing traceability well and still lose time because small fittings, seals, or branch components were not controlled to the same standard. In practice, the weakest documentation chain often appears in the smallest wetted parts.
Typical Industry Case: The Installed Fitting Was Fine but the Record Package Failed
Another common GMP problem is a fitting assembly that appears technically acceptable but still delays release because the records do not close cleanly. The installed hardware may be correct, but if traceability, finish evidence, or linked inspection documentation is incomplete, QA may still treat the assembly as unresolved. This is one of the clearest examples of why fitting selection is also a documentation decision.
Link This to the Project RFQ
The strongest fitting projects define documentation expectations before RFQ, not after fabrication. If the facility expects certificates, traceability, finish evidence, or specific inspection records, those requirements should be written directly into the procurement package. For the broader turnover side of this topic, connect this article with Documentation and Traceability in Pharma Piping Projects.
Research Examples, Industry Cases, and Expert Advice
Research Insight: Hygienic Performance Is Assembly-Level, Not Part-Level
ASME BPE is valuable not because it names one perfect fitting type, but because it frames materials, design, fabrication, inspection, testing, and certification as one hygienic system. That is the correct lens for GMP fitting selection. A fitting that is acceptable in isolation may still be wrong once the surrounding welds, seals, branch geometry, and cleaning expectations are considered.
Industry Case: A Sample Branch Failed Repeatedly Even Though the Main Line Passed
A realistic GMP case is a loop that looks acceptable in the main run while one sample branch keeps generating hygienic concern. The main pipe may be fine. The problem is the local fitting strategy, branch geometry, or the seal-controlled interface around that sample assembly. This is why fittings should be selected for the hardest-to-control local assembly, not for the easiest straight section of tube.
Expert Advice: Select the Fitting for the Hardest-to-Control Location, Not the Easiest Straight Run
This is the simplest rule that prevents many GMP fitting mistakes. Do not choose the connection style based on how quickly it installs on the straight run. Choose it based on whether it still behaves hygienically at the branch, sample point, valve cluster, or other local assembly where the process is least forgiving.
How Engineers Actually Decide: A Practical Selection Method
Step 1: Define the Real Process and Cleaning Exposure
Start with the fluid, the cleaning regime, and the hygienic consequence of failure. A fitting in a SIP-exposed sample assembly is not the same decision as a fitting in a lower-risk auxiliary zone.
Step 2: Identify the Hardest-to-Clean Local Assemblies
Then identify where the actual hygienic risk concentrates. In many GMP systems, this is not the main transfer line. It is the branch, manifold, valve interface, or sample point.
Step 3: Decide the Connection Strategy
Next decide whether the location benefits more from modular access or from reduced interface count. That is the real clamp-versus-weld decision in hygienic engineering.
Step 4: Define Material, Finish, Seal, and Documentation Together
Do not finalize the fitting until the wetted condition is fully defined. Material, finish, gasket or diaphragm choice, and the documentation package should be written together.
Step 5: Check Whether the Fitting Solves the Dominant Risk
The last question is simple: does this fitting reduce the dominant hygienic risk in this exact assembly, or does it only look appropriate because it is common elsewhere in the facility?
Common Mistakes in Sanitary Fitting Selection for GMP Facilities
Choosing by Catalog Appearance
A polished or sanitary-looking fitting does not guarantee a GMP-ready outcome once installed.
Selecting the Fitting Before Reviewing the Branch Geometry
A fitting chosen too early can lock in a local dead-leg or drainability problem that becomes visible only after startup.
Ignoring Gasket and Seal Control
Many fitting-related deviations are actually uncontrolled seal-interface problems.
Debating Alloy but Ignoring Finish
The base metal matters, but the wetted finish and local fabricated condition often determine the hygienic outcome more directly.
Treating Documentation as a Procurement Afterthought
In GMP projects, a technically correct fitting without a clean evidence chain is still a project risk.
Using One Fitting Logic Across All Process Areas
Different areas of the facility do not carry the same hygienic burden, so they should not be fitted to the same standard by default.
Practical Selection Checklist
Use Clamp-Type Hygienic Fittings When
- Controlled modular access is genuinely needed
- Inspection and gasket replacement are part of the normal lifecycle
- The local geometry remains cleanable and drainable
- Seal control is strong enough to support the added interface
Use More Permanently Welded Connections When
- The hygienic consequence of added interfaces is high
- The assembly is relatively fixed and does not benefit enough from routine opening
- The project wants to reduce local seal count and assembly variability
- The location is a critical product-contact or higher-consequence GMP zone
Escalate Review If
- Sample points or branch connections are involved
- Dead-leg or low-drain risk exists
- SIP/CIP cycling is severe
- The area is high-purity, aseptic, or release-sensitive
- QA requires tighter finish and documentation evidence
Questions to Answer Before RFQ
- What process fluid contacts this fitting?
- How will the assembly be cleaned and, where needed, sterilized?
- Is the location self-draining under installed conditions?
- What seal material and replacement logic apply?
- What finish and documentation package are required?
- Is this location better served by access or by reduced interface count?
Conclusion: The Right GMP Fitting Is the One That Reduces the Real Hygienic Risk
The best sanitary fitting is not the most expensive one or the most common catalog option. It is the fitting that reduces the actual hygienic risk in the exact assembly where it will be used. In GMP facilities, fitting selection should always be made together with material, finish, seal, cleanability, and documentation control.
For the full pharma topic cluster, connect this article with Pharmaceutical Hygienic Piping Material Selection, Electropolished 316L vs Standard 316L Stainless Steel, Documentation and Traceability in Pharma Piping Projects, and CIP Design Considerations for Hygienic Piping.
FAQ
How do you select sanitary fittings for GMP facilities?
Start with the real process, cleaning regime, and local hygienic risk. Then choose the fitting that best supports cleanability, drainability, seal control, inspection access, and documentation expectations in that exact assembly.
Are clamp fittings acceptable in pharmaceutical GMP piping?
Yes, in the right locations. Clamp fittings are useful where controlled access, gasket replacement, and modularity are real needs, but they also introduce a controlled seal interface that must be justified and managed properly.
What is the biggest fitting risk in GMP piping?
The biggest risk is usually not the fitting body itself. It is the local hygienic consequence the fitting creates through dead legs, poor drainback, uncontrolled seals, or weak traceability in the installed assembly.
Do sanitary fittings affect dead-leg and cleanability risk?
Yes. Fitting geometry, branch length, orientation, and local assembly design can materially change how well a system cleans and drains.
What material is most common for GMP sanitary fittings?
316L is a common baseline for critical wetted pharmaceutical service, but the final hygienic result still depends on finish, weld integration, seal choice, and documentation, not alloy name alone.
What documentation should sanitary fittings have in pharma projects?
The exact package depends on project scope, but material traceability, certificates, any relevant finish evidence, and linked inspection or turnover records are often needed to support QA release.




