
Electropolished 316L is not automatically the better choice for every pharmaceutical hygienic piping project. It becomes the better choice when the process needs tighter surface consistency, easier inspection response, stronger cleanability confidence, or higher purity expectations than standard 316L finish can reliably support. If the real problem is poor weld quality, bad geometry, dead legs, or weak documentation, electropolishing alone will not fix it.
That is why the real comparison is not “premium finish” versus “basic finish.” It is a question of engineering fit. In pharmaceutical hygienic piping, surface finish has to be judged by product-contact risk, cleanability target, fabrication quality, inspection method, and QA release requirements. A finish upgrade only makes sense when it changes one of those outcomes in a meaningful way.
This guide explains how engineers compare electropolished 316L and standard 316L stainless steel in pharmaceutical hygienic piping, where electropolishing genuinely adds value, where standard 316L is fully acceptable, and why finish selection must stay tied to weld quality, local geometry, and documentation control instead of being treated as a cosmetic decision. In real GMP projects, the finish decision rarely changes because of one abstract theory. It usually changes because of a repeated pattern: local welds are harder to release than expected, finish evidence is weaker than QA wants, or the process ends up needing more inspection confidence than the original RFQ assumed.
Why This Comparison Matters in Pharmaceutical Hygienic Piping
This Is Not a Cosmetic Comparison
In pharmaceutical systems, finish choice affects how the surface behaves in cleaning, inspection, and release, not just how it looks. ASME BPE is useful here because it treats materials, surface requirements, design for cleanability and sterility, fabrication, examinations, inspections, testing, and certifications as part of one integrated engineering framework rather than separate topics. That is exactly how finish decisions should be handled in GMP piping projects.
Engineers do not upgrade to electropolished 316L because the pipe “looks better.” They upgrade when the surface condition is expected to influence hygienic performance, visual or borescope inspection confidence, residue behavior, or the consistency of the turnover package.
Engineering note: in pharma piping, finish is not a styling choice. It is one part of the evidence chain that supports cleanability, inspectability, and QA confidence.
Why GMP Teams Ask This Question So Often
GMP teams ask about electropolished versus standard 316L because finish can become a release issue before it becomes a corrosion issue. In real projects, the question usually appears when one of four things happens:
- The process is more sensitive to surface condition than originally assumed.
- The project expects tighter inspection response or visual consistency.
- The system contains many welded local assemblies where surface condition carries more weight.
- QA wants clearer evidence that the installed surface matches the hygienic target defined during design.
A common project pattern is that the base alloy decision is accepted early, but the finish decision stays open longer because the team is still aligning process risk, fabrication capability, and turnover expectations. That is normal engineering behavior, not indecision. The finish requirement usually becomes clearer only after the team understands what failure mode they are actually trying to avoid.
What the Standards Framework Implies
The standards framework does not treat finish as decoration. FDA 21 CFR 211.65 requires product-contact surfaces to be non-reactive, non-additive, and non-absorptive, while 21 CFR 211.67 ties equipment choice directly to cleaning and maintenance practice. ASTM A270 allows pharmaceutical quality to be requested as a supplementary requirement, which is an important reminder that the tubing decision in pharma projects often goes beyond basic sanitary tubing supply. EU GMP Annex 1 then pushes contamination control strategy and risk management logic into sterile-related environments, which makes uncontrolled finish variation harder to defend in higher-risk applications.
For the broader material baseline, connect this article with Pharmaceutical Hygienic Piping Material Selection.
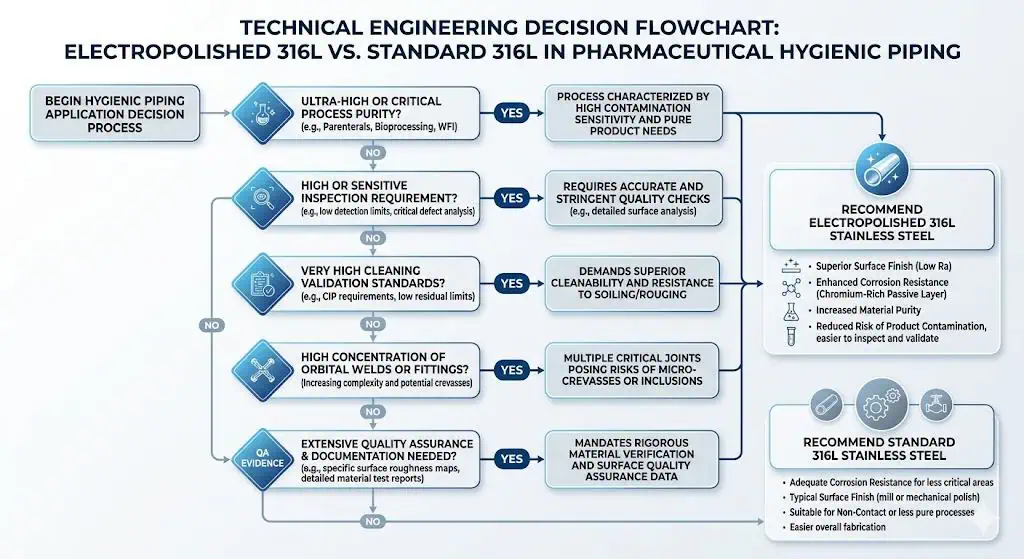
What Is the Real Difference Between Standard 316L and Electropolished 316L
The Alloy Family Is the Same, but the Surface Condition Is Not
This comparison is not about changing the stainless steel family. It is about changing the condition of the product-contact surface. Both options may start from 316L stainless steel, but the final hygienic behavior depends in part on how the surface is finished, how consistently that finish is delivered, and how that surface interacts with cleaning, inspection, and local fabrication details.
That difference matters because pharmaceutical hygienic piping rarely fails in the abstract. It fails at local details such as weld-adjacent zones, ferrule transitions, branch assemblies, valve interfaces, and places where QA or operations need more confidence than a generic sanitary finish can always provide.
What Electropolishing Changes in Practical Terms
Electropolishing is usually selected to improve surface consistency, inspection confidence, and the practical cleanability margin of the system. In project language, that often means:
- A more controlled product-contact surface condition
- More confidence during visual, borescope, or release-related review
- A stronger fit for higher-purity or more demanding hygienic expectations
- Better alignment with projects that treat finish as part of the qualification logic rather than only a fabrication detail
Typical industry case: a team may accept standard 316L for general hygienic service but move to electropolished 316L for a more inspection-sensitive product-contact loop because the finish decision improves release confidence, not because the alloy family itself was wrong.
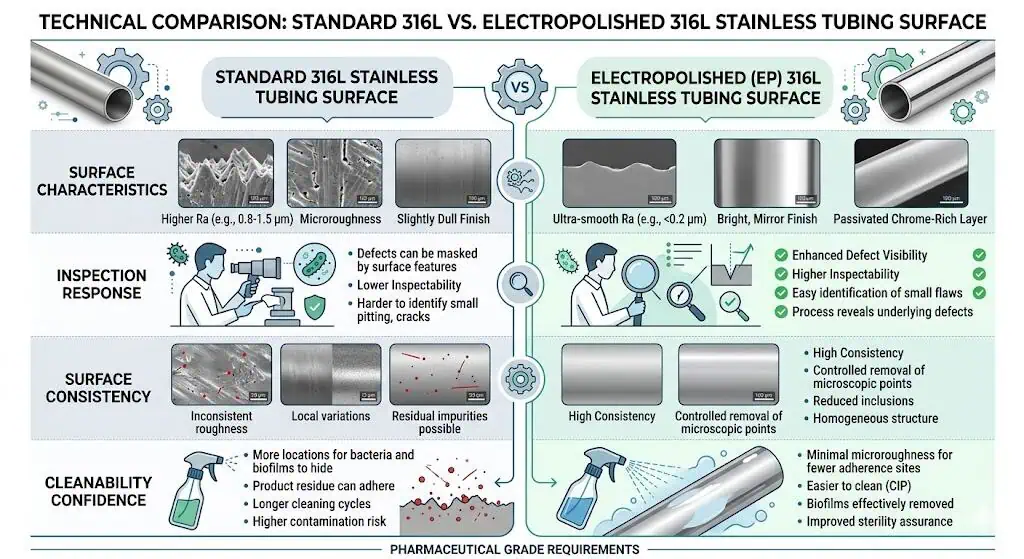
What Electropolishing Does Not Automatically Change
Electropolishing does not automatically fix the rest of the hygienic system. It does not correct poor weld purge, bad branch geometry, trapped dead legs, weak drainability, or incomplete traceability. It can improve the surface condition, but it does not excuse a system that was badly designed or fabricated upstream.
This is one of the most important engineering boundaries in the whole topic. A common mistake is to use electropolish as a compensating choice when the real issue is weld quality or assembly design. In those cases, the finish upgrade may improve part of the risk while leaving the dominant failure mode untouched.
When Standard 316L Is Usually Enough
Moderate-Risk GMP Hygienic Service
Standard 316L is often enough when the hygienic target is already achievable through good fabrication, controlled weld quality, appropriate cleaning, and moderate finish expectations. Not every pharmaceutical line requires electropolish to perform reliably. In many GMP systems, the dominant success factors are still weld execution, geometry control, proper materials documentation, and realistic maintenance discipline.
In practical terms, if the process is not especially surface-sensitive, the inspection burden is manageable, and the team already has good control over fabrication quality, standard 316L can be a sound engineering choice.
Where Teams Over-Spec Electropolish
Teams often over-specify electropolish when they have not clearly defined what hygienic or QA benefit they actually need from it. This usually happens when finish is treated as a prestige feature rather than a risk-control tool. The result can be higher cost without a corresponding reduction in the system’s dominant failure mode.
Expert advice: if the team cannot explain whether the upgrade is meant to improve cleanability confidence, inspection response, release consistency, or another defined project outcome, the finish requirement is probably not mature enough yet.
Typical Industry Case: The Line Performed Well with Standard 316L
Many pharmaceutical hygienic loops perform well with standard 316L because the real engineering basics were done correctly. In those systems, weld quality was controlled, local geometry was kept cleanable, documentation closed properly, and the process did not impose a finish-sensitive burden that justified going further. The lesson is not that electropolish has no value. The lesson is that a well-fabricated standard 316L system can outperform a poorly executed electropolished system.
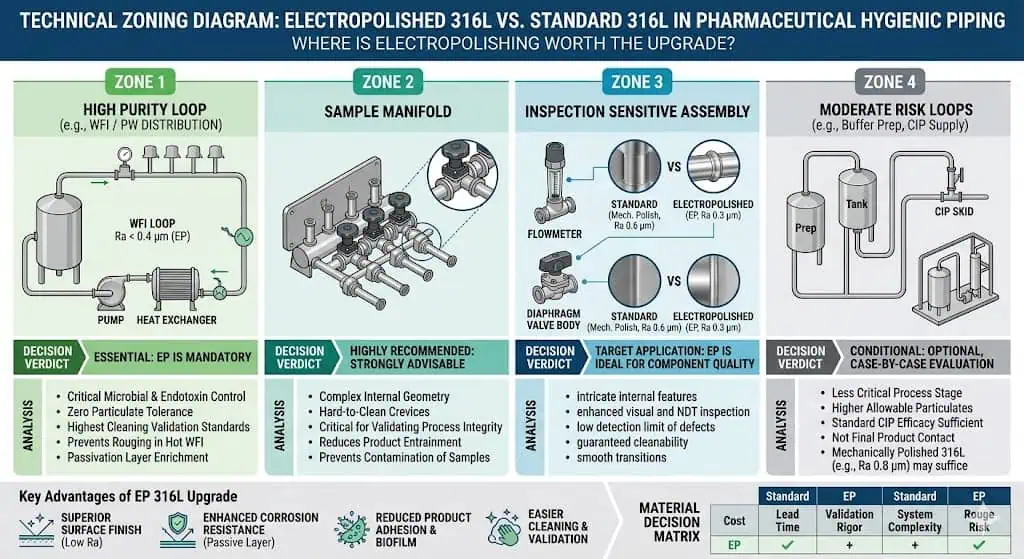
When Electropolished 316L Is Worth the Upgrade
High-Purity and High-Inspection Environments
Electropolished 316L is more likely to justify itself in high-purity and inspection-sensitive applications. When the project expects tighter surface uniformity, stronger inspection confidence, or more demanding hygienic scrutiny, the finish upgrade can become easier to defend. That is especially true in systems where release decisions depend heavily on local surface condition and where visual inconsistency is likely to trigger repeated technical discussion.
When the Process Is More Sensitive to Surface Condition
Some processes are more sensitive to local surface behavior than others. In these cases, the finish decision carries more engineering weight because the system is less tolerant of variability in inspection response, residue behavior, or local hygienic confidence. The question is not whether electropolish is objectively better in every environment. The question is whether the process is sensitive enough that the finish difference changes the practical risk.
Typical Engineering Case: Electropolish Was Chosen for Inspection and Cleanability Confidence
A common engineering case is a project that originally considered standard 316L acceptable, then moved to electropolished 316L because the required inspection and cleanability confidence became tighter during design review. The key point is that the change was driven by the hygienic target, not by a belief that standard 316L was defective by default. This is the correct logic: define the expected performance first, then decide whether electropolish materially improves the chance of meeting it.
Expert Advice: Upgrade EP Only When You Can Name the Benefit
The strongest electropolish decisions are specific. Engineers who specify it well can usually say exactly what they expect it to improve: inspection consistency, cleanability confidence, higher-purity surface expectations, or QA comfort during release. If the project cannot name that benefit clearly, the finish upgrade may be more emotional than technical.
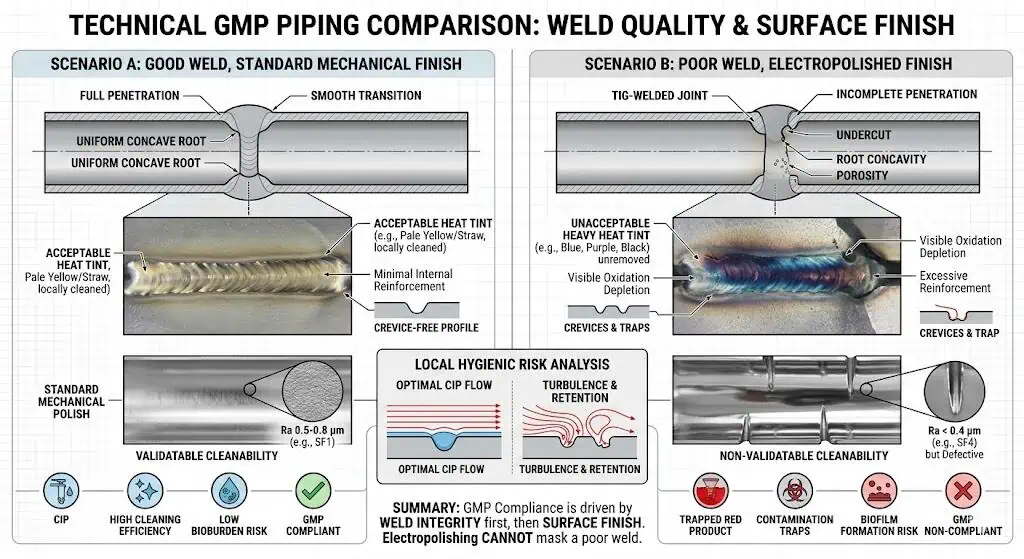
What Electropolishing Cannot Fix
Poor Weld Quality
Electropolishing does not replace weld quality. If the weld zone is poorly purged, irregular, difficult to inspect, or inconsistent with the hygienic intent of the system, the finish upgrade does not erase that risk. In practical GMP work, weld quality often dominates the local surface performance more than the general finish category does.
Typical industry case: the specification correctly upgraded the finish, but a small number of weld-adjacent locations still triggered concern because the local weld condition, not the general tubing finish, remained the weakest point in the loop.
Bad Geometry and Dead Legs
Electropolishing does not make a poorly designed assembly cleanable. If a branch is too long, a sample point traps fluid, or a valve cluster creates a local hygienic shadow, the system can still underperform even with a tighter finish requirement. This is why finish selection should never be separated from connection design and local cleanability logic.
For that side of the problem, connect this article with Sanitary Fitting Selection for GMP Facilities.
Weak QA Documentation
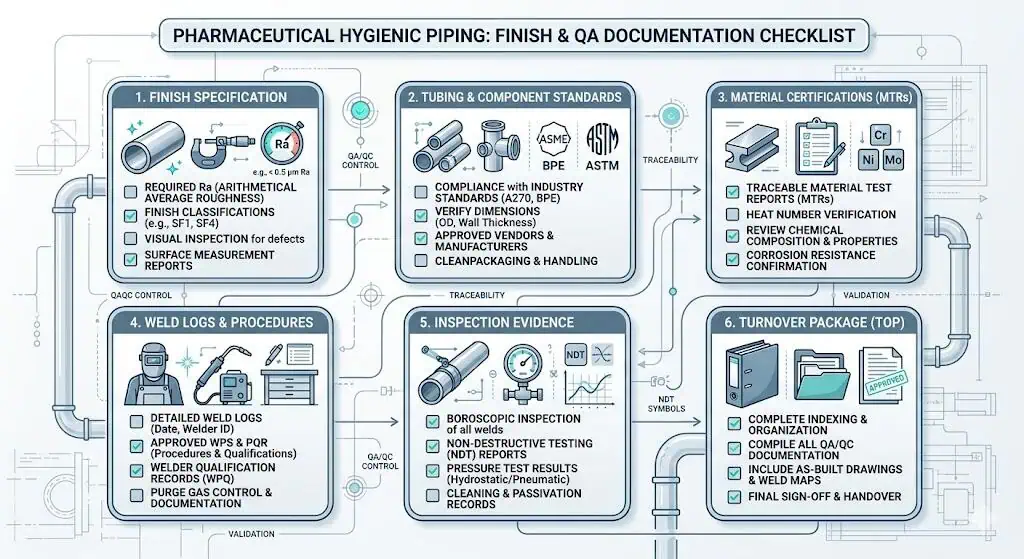
Electropolishing also does not replace documentation. In GMP projects, the finish decision enters the turnover package. If the project requires finish-related evidence and the record set is incomplete, the system can still become difficult to release even when the installed surface looks acceptable to the eye.
This is why finish choice and QA documentation belong in the same discussion, not in separate project silos.
Typical Industry Case: The Project Upgraded Finish but Still Failed Hygienic Intent
One of the most common missteps is a project that upgraded to electropolished 316L and still did not solve the real hygienic problem. Later review showed that the dominant issue was weld condition, geometry, or documentation closure rather than the finish category. This is exactly why electropolish should be treated as one control measure within a larger hygienic system decision, not as a universal correction for every risk.
How Finish Choice Connects to Welds, Inspection, and Cleanability
Why Weld Zones Still Dominate the Risk
In many pharmaceutical piping investigations, the first hygienic concern appears at the weld zone, not the general tube wall. That is why finish discussions that ignore welds usually stay too abstract to be useful. The more welded the system becomes, the more the project has to care about local execution quality, because that is where the intended hygienic performance is most likely to weaken first.
Why Inspection Strategy Changes the Finish Decision
The stricter the inspection and release strategy, the more likely finish will matter as a project variable. If visual review, borescope expectations, or QA scrutiny are tight, the finish choice becomes easier to justify because it directly affects how the surface will be judged in practice. In other words, some finish upgrades are driven as much by inspection reality as by process chemistry.
Research Insight: Surface Requirements Only Matter When the Whole Fabrication Chain Supports Them
ASME BPE treats surface requirements as part of the broader fabrication, inspection, and testing chain rather than as an isolated decoration step. That is the right way to interpret finish requirements in pharmaceutical hygienic piping. A surface target only becomes meaningful when fabrication practice, local weld quality, inspection methods, and turnover controls are capable of supporting it consistently.
How Engineers Decide in Real Projects
Start with the Failure Mode, Not the Finish Name
The most reliable way to choose between standard 316L and electropolished 316L is to start with the failure mode you are trying to control. A practical sequence looks like this:
- Define the process purity and hygienic target.
- Review cleaning and sterilization exposure.
- Review weld density and local assembly risk.
- Define how the system will be inspected and released.
- Then decide whether standard 316L is enough or electropolish changes the practical risk margin.
Questions to Ask Before Writing the RFQ
- Is the process finish-sensitive enough to justify electropolish?
- Is the real concern cleanability, inspection, or QA release?
- Are weld quality expectations already defined?
- Will the project require finish-related evidence beyond basic material certification?
- Is electropolish solving a real engineering problem or only increasing cost?
A Simple Engineering Rule
Use standard 316L when the hygienic target is achievable through good fabrication, controlled weld quality, and moderate finish expectations.
Move to electropolished 316L when finish consistency, inspectability, and cleanability confidence must be tighter than standard finish can reliably support.
Do not use electropolish as a substitute for fixing weld, geometry, or documentation problems.
Common Mistakes When Comparing Electropolished 316L and Standard 316L
Treating EP as a Luxury Upgrade
Electropolish should be justified by hygienic or QA value, not by appearance or prestige.
Treating Standard 316L as Automatically Insufficient
Many GMP systems perform well with standard 316L when fabrication, geometry, cleaning, and documentation are all controlled properly.
Ignoring Welds While Debating Finish
A finish argument that ignores weld execution usually misses the real local risk.
Choosing Finish Before Defining QA Expectations
In regulated projects, finish choice and release expectations need to be aligned early.
Using EP to Hide a Design or Fabrication Weakness
A tighter finish cannot rescue a fundamentally weak hygienic assembly.
Practical Selection Checklist
Choose Standard 316L If
- The hygienic target is moderate and well defined
- Weld and fabrication controls are already strong
- Finish-related inspection sensitivity is not unusually high
- The real system risk is manageable without a tighter finish spec
Choose Electropolished 316L If
- The process is high-purity or inspection-sensitive
- Finish consistency matters materially to qualification or release
- Surface-driven cleanability confidence is important
- The project has a real, defined reason to tighten finish expectations
Reassess the Whole System If
- The team is arguing about finish while weld criteria remain vague
- Branch geometry is still poor
- Local assemblies are hard to clean
- The documentation package is not yet clear
Conclusion: The Best Finish Choice Is the One That Solves the Right Problem
Electropolished 316L is not automatically the best answer, and standard 316L is not automatically the cheaper compromise. The right finish choice depends on what hygienic failure, inspection burden, or QA risk the project is actually trying to control.
For the full pharma topic cluster, connect this article with Pharmaceutical Hygienic Piping Material Selection, Sanitary Fitting Selection for GMP Facilities, and Documentation and Traceability in Pharma Piping Projects.
FAQ
Is electropolished 316L required for pharmaceutical piping?
No. It is justified when the project needs tighter surface consistency, cleanability confidence, or inspection and release certainty than standard 316L can reliably support.
What is the difference between standard 316L and electropolished 316L?
The alloy family may be the same, but the finished product-contact surface is not. The comparison is mainly about surface condition, consistency, and how that affects cleanability and inspection response.
Does electropolishing improve cleanability?
It can improve cleanability confidence and surface consistency, but it does not automatically fix a system with poor welds, dead legs, or bad geometry.
Can electropolishing replace good weld quality?
No. Weld quality still dominates many of the highest-risk local hygienic zones in pharmaceutical piping.
When is standard 316L enough in GMP piping?
It is often enough when the hygienic target is achievable through good fabrication, moderate finish expectations, controlled weld quality, and realistic inspection demands.
Is electropolishing a documentation issue as well as a finish issue?
Yes. In GMP projects, finish choice may affect how the system is specified, inspected, and supported in the turnover package.




