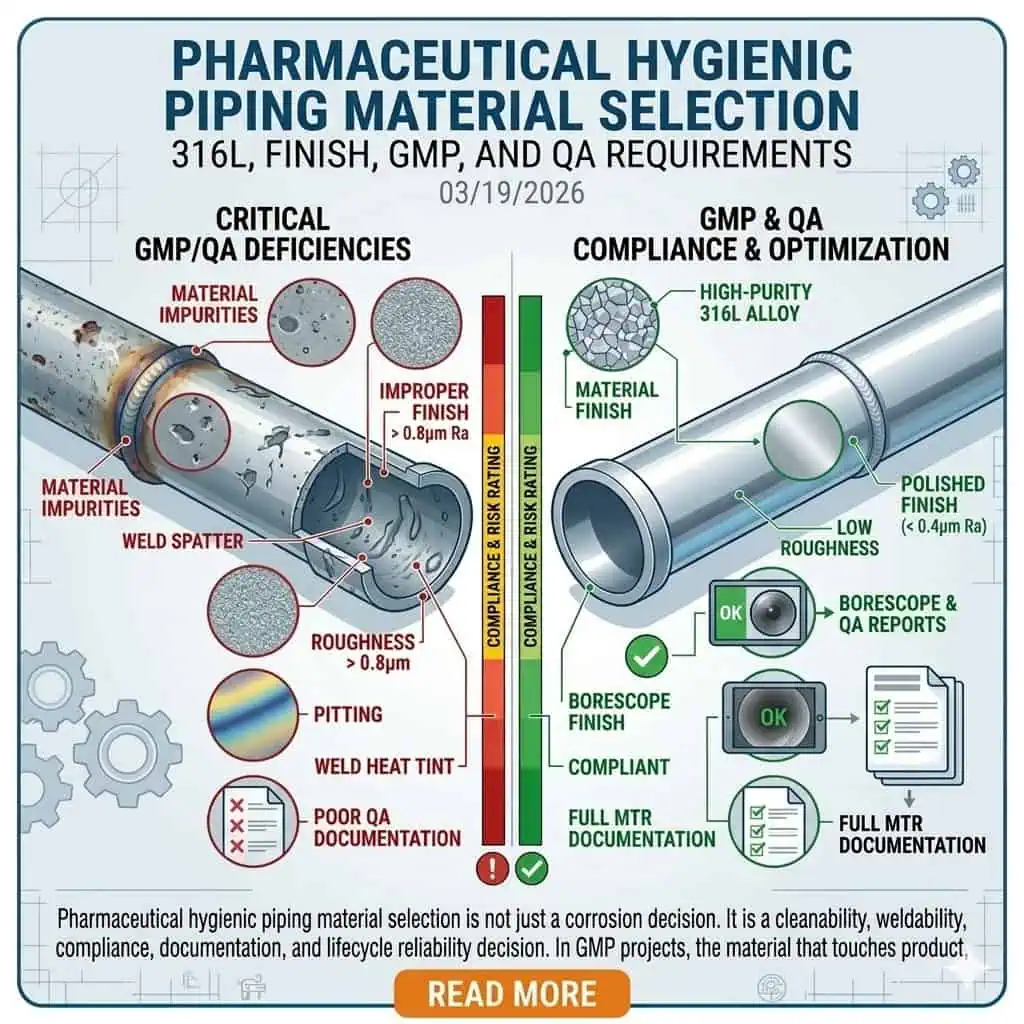
Pharmaceutical hygienic piping material selection is not just a corrosion decision. It is a cleanability, weldability, compliance, documentation, and lifecycle reliability decision. In GMP projects, the material that touches product, process fluids, high-purity water, or cleaning media must do more than “resist rust.” It must remain non-reactive, non-additive, non-absorptive, cleanable, inspectable, and support repeatable fabrication and QA release. That is why experienced engineers do not select tubing and fittings by alloy name alone.
In most pharmaceutical hygienic piping systems, 316L stainless steel is the practical baseline for critical metallic product-contact service because it balances corrosion margin, weldability, industry acceptance, and compatibility with high-purity fabrication practices. That does not mean every line, every fitting, or every assembly must be handled the same way. The correct decision depends on actual process chemistry, CIP/SIP regime, surface finish requirement, weld density, contamination sensitivity, and the documentation package required for turnover and validation.
This guide explains how engineers select materials for pharmaceutical hygienic piping, where 316L makes clear sense, where standard 316L may not be enough without the right finish and weld quality, and where documentation and traceability become just as important as the metal itself. In actual project reviews, the final material decision is rarely changed by one big failure. It is usually changed by a pattern of smaller issues: a weld that is harder to inspect than expected, a finish record that does not close cleanly with QA, a sample branch that sees harsher SIP cycling than the rest of the loop, or a local assembly that behaves like a higher-risk zone than the P&ID suggested.
What Pharmaceutical Hygienic Piping Material Selection Really Means
Material Choice Is a GMP and Contamination-Control Decision
In pharmaceutical systems, material selection is part of contamination control. A pipe, valve body, ferrule, or fitting is not accepted because it looks sanitary. It is accepted because the product-contact surface can support the intended process without altering safety, identity, strength, quality, or purity. That principle aligns with FDA 21 CFR 211.65, which requires product-contact surfaces to be non-reactive, non-additive, and non-absorptive.
In practice, that means engineers review material choice together with surface finish, weld condition, drainability, passivation, cleaning chemistry, and inspection accessibility. A correct alloy with poor weld purge, uncontrolled surface condition, or weak documentation can still become a GMP problem. One common field lesson is that a piping package can look technically acceptable at fabrication release and still become difficult during qualification because the material decision was treated as a purchasing item rather than a contamination-control decision.
Engineering note: in GMP work, the question is not only “Will this alloy survive?” but also “Will this finished, welded, documented surface behave predictably in inspection, cleaning, and QA release?”
Why Pharma Piping Selection Is Different from General Sanitary Service
Pharmaceutical hygienic piping is usually held to a tighter standard than general food or utility sanitary service. The reason is not only product value. It is the consequence of contamination, residue retention, undocumented substitutions, inconsistent fabrication, and delayed QA release in validated environments. The same stainless steel grade may be acceptable in a general sanitary application but still fall short in a GMP system if finish consistency, weld records, or traceability are not controlled to the level the project requires.
That is why material review in pharma projects should sit beside connection selection, dead-leg control, weld acceptance, and QA turnover planning. A correct alloy in the wrong assembly is still the wrong engineering outcome.
Which Standards Actually Drive the Decision
The most useful standards are the ones that change what you specify, inspect, and accept. In pharmaceutical hygienic piping, the most relevant references usually include:
- ASME BPE for materials, design, fabrication, inspection, testing, and certification logic in bioprocessing and pharmaceutical equipment.
- FDA 21 CFR 211.65 for equipment construction and product-contact surface suitability.
- FDA 21 CFR 211.67 for cleaning and maintenance expectations.
- ASTM A270/A270M for sanitary tubing, including the option to specify pharmaceutical quality as a supplementary requirement.
- EU GMP Annex 1 for contamination control, quality risk management, and sterile-manufacturing expectations.
The engineer’s job is to turn those requirements into actual material, finish, weld, and documentation decisions that can survive procurement, fabrication, inspection, startup, and QA review. In strong projects, standards are not quoted at the end to justify a finished choice. They are translated at the beginning into RFQ language, inspection criteria, turnover requirements, and change-control boundaries.
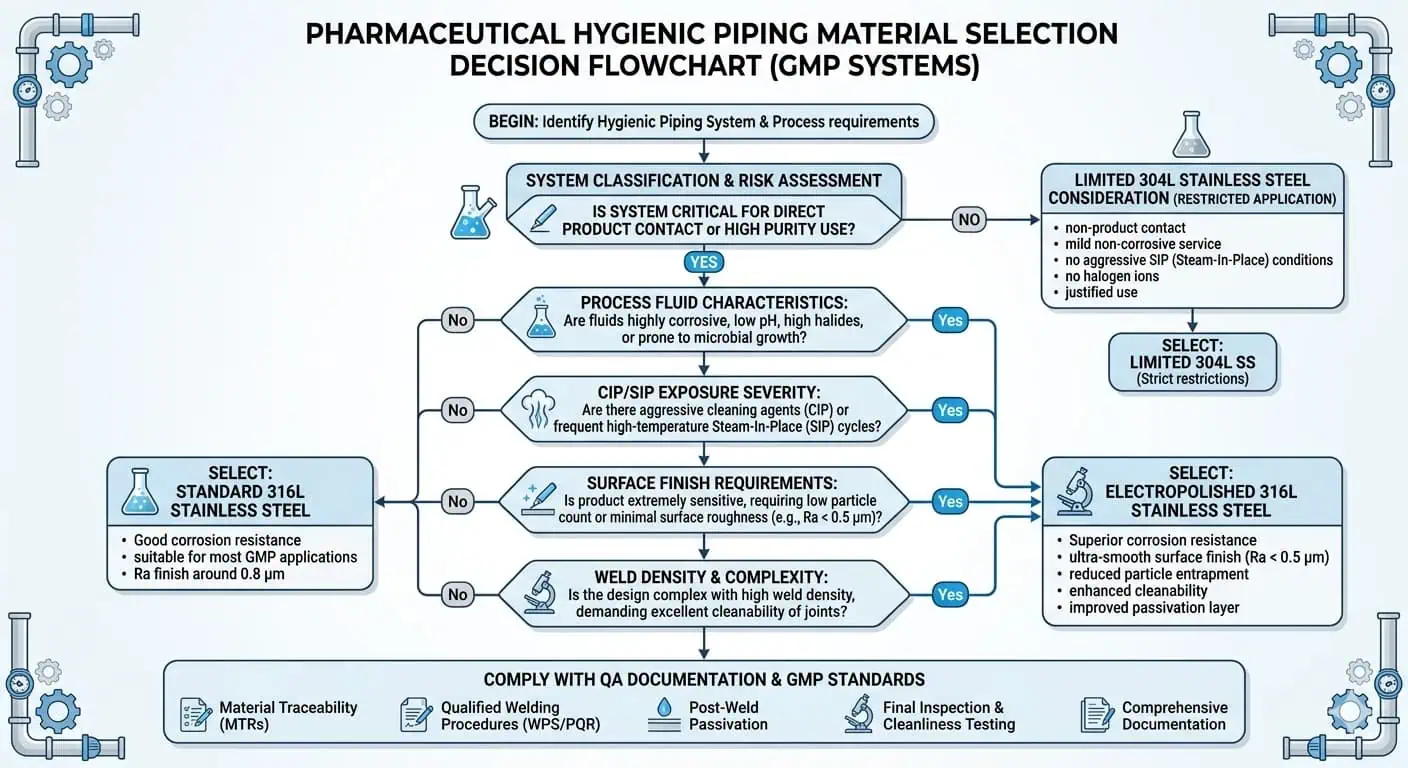
The Core Material Families Used in Pharmaceutical Hygienic Piping
Why 316L Is the Practical Baseline for Many GMP Hygienic Systems
316L is commonly treated as the baseline metallic material for critical pharmaceutical hygienic piping because it offers a practical combination of corrosion resistance, weldability, and broad industry acceptance. The low-carbon grade supports welded hygienic fabrication more reliably than standard 316 in many process conditions, and the molybdenum-bearing chemistry improves resistance to localized corrosion compared with 304/304L in more demanding cleaning and service environments.
That does not make 316L a magic answer. It means 316L is often the most defensible starting point for product-contact metallic piping when the system must also support high-purity fabrication and documentation expectations. In real projects, engineers often choose 316L not because every section of the system is chemically severe, but because the total project risk is carried by welded assemblies, local cleanability, SIP/CIP exposure, and validation expectations together.
When 304/304L May Still Appear—and Why It Is Often Limited
304 or 304L may still appear in some pharmaceutical projects, but usually in more limited roles. Lower-risk auxiliary service, legacy systems, noncritical support areas, or cost-driven historical designs may still use 304-family materials. But in critical hygienic product-contact piping, the industry usually moves toward 316L because the total risk is broader than bulk corrosion alone.
This is one of the most common specification mistakes in early-stage projects: treating 304 vs 316L as a simple price choice instead of a cleanability, fabrication, and QA-risk choice. In practice, when a team asks whether 304L can still be used, the better follow-up question is usually: used where, under which cleaning regime, with what finish requirement, and under what documentation standard?
Where Non-Metallic Materials Still Matter
Pharmaceutical piping material selection does not stop with the tube wall. Diaphragms, valve seats, gaskets, hoses, and other non-metallic wetted parts often determine whether the final assembly performs as intended. PTFE, modified PTFE, EPDM, FKM, silicone, or application-specific compounds may all be involved depending on chemistry, temperature, SIP exposure, and validation requirements.
In many GMP deviations, the weak point is not the stainless tubing. It is the interface material that was treated as a minor consumable instead of a controlled wetted component. A common example is a chemically compatible metal system paired with a seal material that ages faster under repeated SIP than the original risk review assumed. The lesson is not that the metal selection was wrong in isolation; it is that the wetted system was not reviewed as a whole.
How Engineers Decide Between Standard 316L, Electropolished 316L, and Higher-Spec Options
Standard 316L vs Electropolished 316L
Standard 316L and electropolished 316L are not competing alloy families. They are different surface-condition decisions built on the same base material family. Standard 316L may be fully acceptable where the required finish, cleanability, and inspection expectations are moderate. Electropolished 316L becomes more attractive where surface consistency, residue release behavior, inspectability, and high-purity cleanability requirements are tighter.
The better question is not “Is electropolished better?” but “Does this process need the finish-related advantages strongly enough to justify the tighter requirement?” In real engineering review, electropolish is often selected not because standard 316L failed everywhere, but because the process demanded a more controlled surface for inspection confidence, residue response, or high-purity operating discipline. For the detailed finish discussion, connect this article with Electropolished 316L vs Standard 316L Stainless Steel.
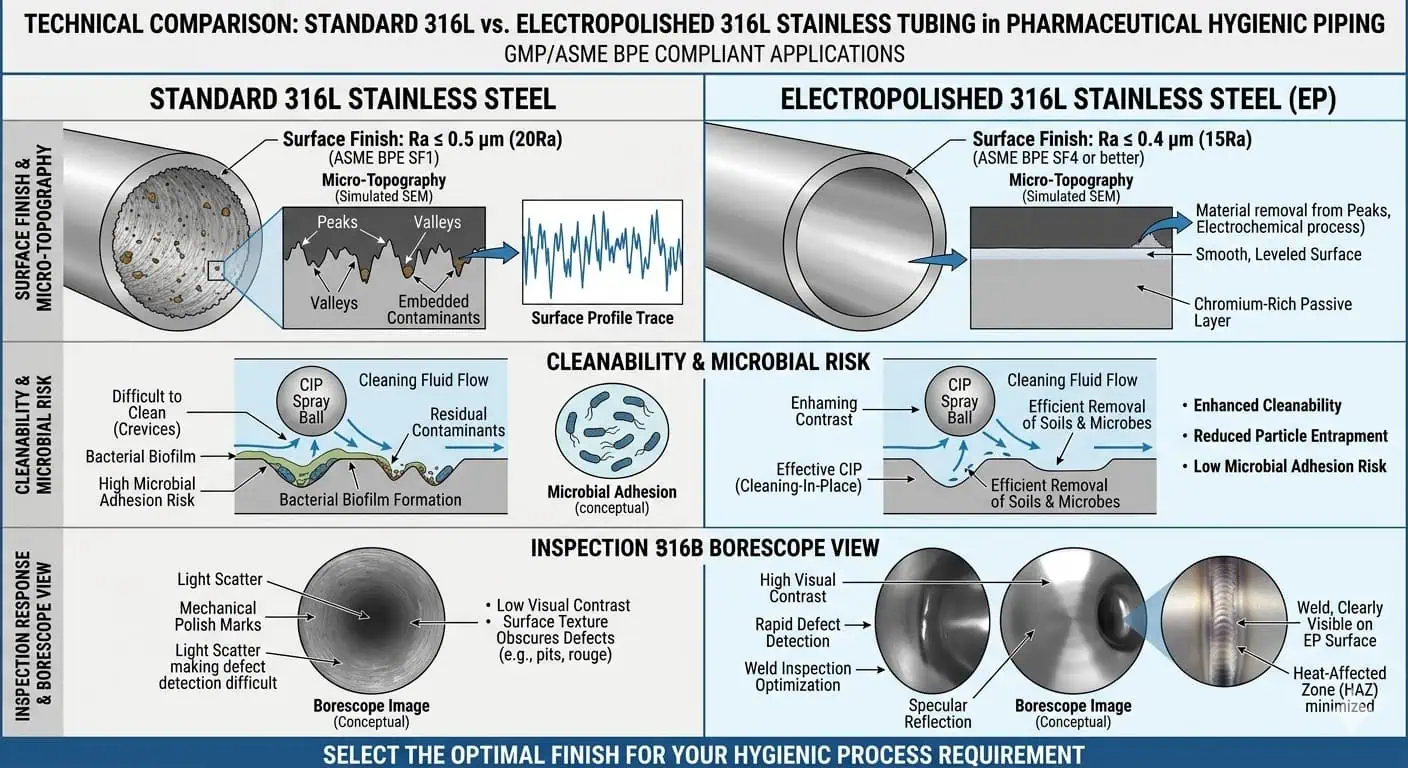
When Surface Finish Changes the Real Hygienic Performance
Surface finish is not a cosmetic upgrade in pharmaceutical service. It changes cleanability, inspection response, and residue-retention behavior. A finish requirement matters most where the process is sensitive to residue, where SIP/CIP repeatability is critical, where product-contact inspection is strict, or where local geometry makes any surface defect more consequential.
In high-purity and biopharmaceutical environments, engineers do not separate alloy selection from finish selection because the hygienic result is created by both together. One repeated lesson from field investigations is that a system can use the correct alloy and still underperform if the local finish condition varies more than the risk model allowed.
When a Higher Alloy or Special Material Review Is Justified
Sometimes 316L is still not enough, but that decision should come from chemistry and failure mechanism, not habit. More aggressive halide exposure, unusual solvent systems, strong process chemistry, or repeated failure history may justify a higher-alloy review. That step should be based on actual service data, corrosion mechanism review, and process chemistry, not on a generic assumption that “pharma always uses the highest grade available.”
Expert advice here is straightforward: do not upscale the alloy because the system is “important.” Upscale it only when you can define the failure mode you are trying to prevent and the reason the baseline option does not provide enough margin.
Material Selection by Process Area in Pharmaceutical Facilities
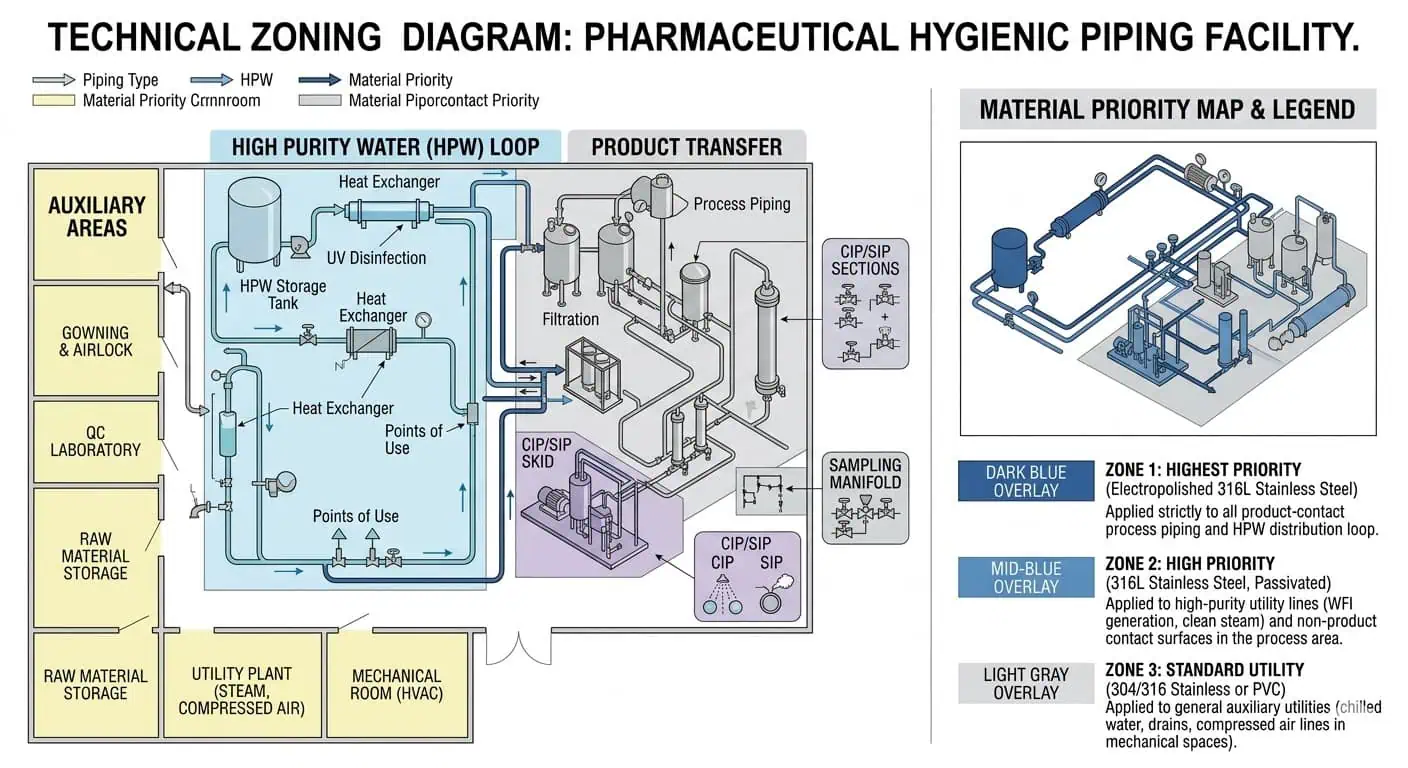
High-Purity Water and Clean Utility Related Systems
High-purity water and related clean utility loops put pressure on material consistency, weld quality, and long-term surface stability. In these systems, the material decision is tied closely to rouging tendency, finish consistency, weld acceptance, and sanitization compatibility. The strongest specifications usually focus less on the alloy name alone and more on whether the complete tubing-and-weld package can support stable, repeatable hygienic performance over time.
Typical engineering note: in high-purity water loops, the first concern is often not bulk wall corrosion. It is local finish inconsistency, weld condition, or incomplete restoration of the product-contact surface after fabrication and rework. That is why engineers who have seen repeated loop investigations tend to focus early on weld execution and finish evidence, not only on alloy designation.
Product-Contact Process Lines and Transfer Assemblies
Product-contact process lines usually justify the most conservative material thinking. This is where corrosion, surface change, extractables concern, or fabrication inconsistency can have the most direct effect on product quality and batch confidence. The material grade, finish, fitting interfaces, and documentation package should all be defined before RFQ, not clarified after fabrication.
In project terms, this is where “acceptable for general sanitary use” and “acceptable for validated product-contact use” separate most clearly. The penalty for late clarification is often not only technical rework, but delayed FAT, SAT, or QA closeout.
CIP/SIP-Exposed Sections
CIP/SIP-exposed sections often reveal material weakness faster than the process itself. Repeated hot water, clean steam, caustic, acid, sanitizer, and cooling cycles change the service environment significantly. In practice, many material-related problems in pharmaceutical piping are not first revealed by the product; they are revealed by repeated cleaning and sterilization cycles.
This is one reason why an engineer should review metallic and non-metallic wetted materials together whenever the system sees repeated SIP/CIP duty. A common field pattern is that the tubing remains acceptable while a local seal, seat, or branch assembly ages faster than expected under the real thermal and chemical cycle.
Sampling, Valve Manifolds, and Other High-Risk Local Assemblies
The most difficult material decisions are often at local assemblies, not in straight runs of tubing. Sample valves, distribution manifolds, branch connections, diaphragm valves, and instrument tees combine higher weld density, more interfaces, more dead-leg risk, and more inspection sensitivity. This is where the total hygienic package matters most.
That is why this article should connect directly with Sanitary Fitting Selection for GMP Facilities. In pharma, the correct alloy in the wrong assembly is still the wrong outcome. Engineers who troubleshoot these systems regularly know that the line rarely fails “everywhere.” It fails first at the local detail that was treated as normal when it was actually the highest-risk point in the loop.
Weldability, Surface Condition, and Post-Fabrication Treatment
Why Weld Density Changes Material Risk
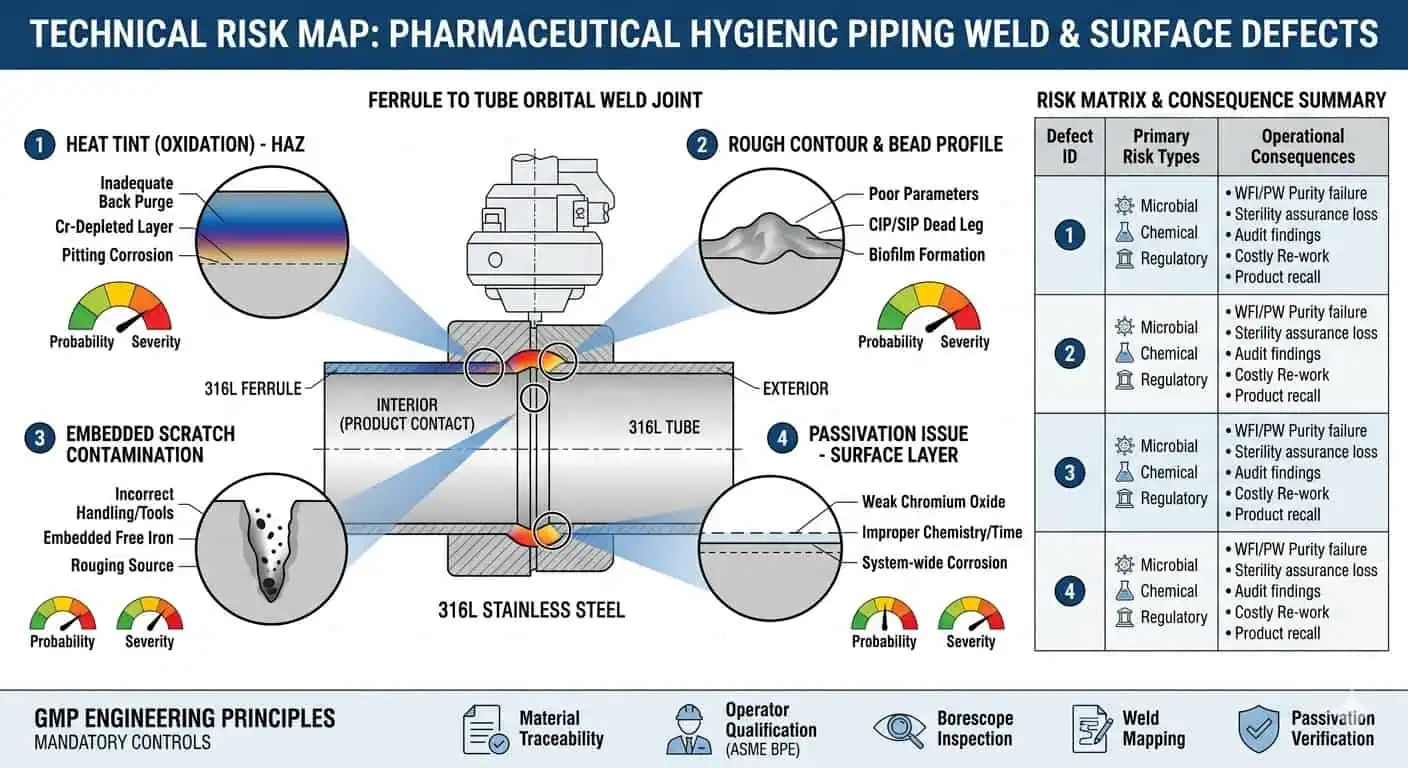
The more welds a hygienic system contains, the less useful it is to talk about material choice as though only the base tube matters. Weld heat-affected zones, purge quality, internal bead condition, orbital-weld consistency, and rework history all influence whether the final system behaves like a cleanable GMP asset or a recurring deviation source.
A system with excellent alloy choice but poor weld execution is still a weak pharmaceutical system. In practical terms, every added weld increases the importance of fabrication discipline, not just the importance of the alloy certificate.
Why Surface Defects Can Defeat a Good Material Choice
A correct alloy does not rescue a poor product-contact surface. Surface irregularities, weld discoloration, poor contour, scratches, contamination from fabrication handling, or uncontrolled rework can all reduce the hygienic value of a good material selection. This is where engineers often see the first gap between specification intent and installed reality.
Typical industry case: the project correctly specified 316L for a critical product-contact loop, but the turnover package still triggered concern because local weld appearance, finish consistency, and boroscope review did not support the claimed hygienic standard. The alloy was not the problem. The fabricated surface condition was.
That case is common enough to matter. It is one reason experienced reviewers do not sign off mentally on a loop just because the material test certificate looks right.
Passivation, Cleaning Compatibility, and Long-Term Stability
Passivation and post-fabrication treatment are part of the material strategy, not an optional finishing touch. A material that is correct on paper but poorly finished, poorly cleaned after fabrication, or inadequately restored after weld work will not perform like the specified material was intended to perform. That matters even more in systems with repeated cleaning, sanitization, and validation scrutiny.
Expert advice here is simple: never treat post-fabrication surface condition as a cosmetic closeout item. In hygienic pharmaceutical service, it is part of the technical performance of the material decision itself.
Documentation and Traceability: Why Material Selection Is Also a QA Decision
What QA Usually Wants to See
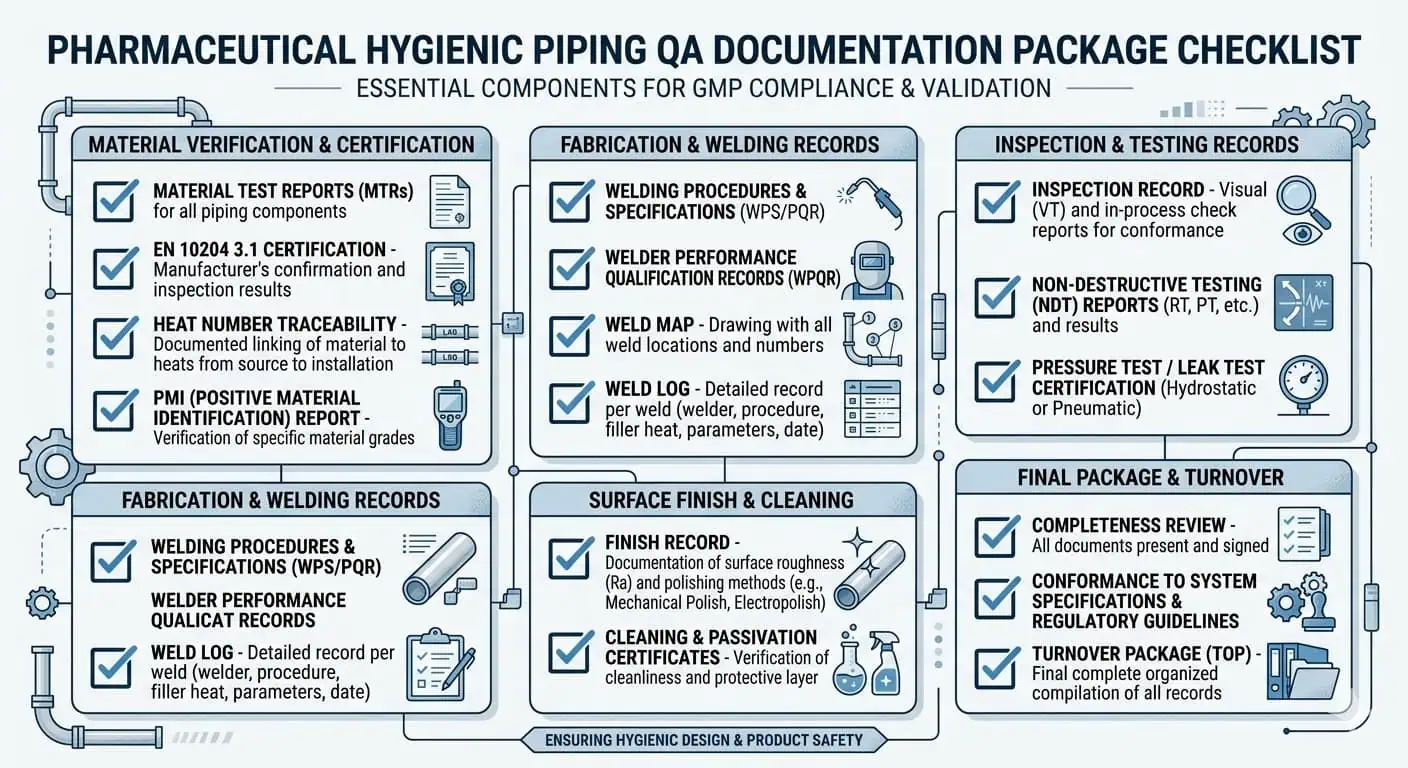
In GMP piping projects, the correct material is only half the answer. QA usually needs proof. Depending on project scope, that may include heat numbers, material test certificates, EN 10204 3.1 certificates, PMI records where required, surface finish records, weld maps, weld logs, inspection records, and turnover documentation that ties the installed assembly back to the approved specification.
The exact package changes by project, but the pattern is consistent: if the material choice cannot be traced and verified, it remains a project risk. This is where many technically capable packages lose time during turnover: not because the metallurgy was wrong, but because the evidence chain was thinner than the GMP environment required.
Why “Correct Material” Without Records Is Still a Project Risk
One of the most common pharma project failures is not wrong alloy selection. It is incomplete closure of the evidence chain. The tubing may be correct. The fittings may be correct. The welds may even be acceptable. But if traceability, finish records, or inspection documentation are incomplete, QA release can still stall.
Typical industry case: a GMP piping package passed fabrication review but failed turnover readiness because heat-traceability and finish records for a group of hygienic spool pieces were incomplete. The problem was not technical metallurgy. It was documentation control.
This is why experienced engineers treat documentation as part of material selection from the beginning, not as a paperwork exercise for the end of the project.
How to Define Material Requirements Before RFQ
The best time to define material expectations is before RFQ, not during punch-list review. A strong RFQ usually defines the alloy grade, tubing standard, finish target, weld requirements, passivation expectations, documentation deliverables, and any specific inspection requirements for acceptance.
For the full QA side of that topic, connect this article with Documentation and Traceability in Pharma Piping Projects.
Research Examples, Industry Cases, and Expert Advice
Typical Engineering Case 1: 316L Was Correct, but the Weld Zone Failed the Hygienic Intent
This is one of the most realistic pharmaceutical piping failures. The design team chose the right alloy family, but the installed weld zone did not preserve the hygienic intent of the material choice. The resulting concern may show up as finish inconsistency, inspection failure, difficult cleaning response, or QA hesitation about local product-contact quality.
The lesson is direct: in pharma, material selection and weld execution cannot be separated. The strongest systems are not the ones with the most expensive material description. They are the ones where the material, finish, fabrication, and inspection logic all support the same hygienic target.
Typical Engineering Case 2: The QA Problem Was Documentation, Not the Alloy
Another common failure mode appears during turnover. The project used the correct material grade, but the package lacked the records needed to prove compliance. Missing certificates, unclear traceability, incomplete finish records, or weak weld documentation can delay approval even when the installed metal is technically right.
This is why experienced engineers treat documentation as part of material selection from the beginning. In practical GMP work, an undocumented correct choice is still an incomplete choice.
Typical Engineering Case 3: Electropolish Was Needed Because of Cleanability, Not Because 316L Was “Wrong”
Sometimes standard 316L is not rejected because the alloy is inadequate, but because the finish requirement changed the hygienic target. In those cases, the better answer is not to say the project chose the wrong metal family. The better answer is that the project eventually recognized a tighter cleanability and inspectability requirement than the original finish specification supported.
That is a normal engineering refinement, not an embarrassment. It is exactly how a strong process team improves specification accuracy over time.
Expert Advice: Ask What Failure You Are Actually Trying to Prevent
The best material selection question is not “What grade do most people use?” It is “What failure are we trying to prevent in this specific system?” The answer may be corrosion. It may be rouging risk. It may be extractables concern. It may be finish-driven cleanability. It may be weld inconsistency. It may be QA closure failure. Once that failure mode is clear, the material decision becomes much more rational.
That is the most consistent advice from engineers who have worked through real pharma piping deviations: define the failure mode first, and the material logic becomes clearer, narrower, and easier to defend.
Common Mistakes in Pharmaceutical Hygienic Piping Material Selection
Choosing by Alloy Name Alone
The alloy family matters, but the installed hygienic result depends on finish, welds, geometry, interfaces, and documentation too.
Separating Grade from Surface Finish
Standard 316L and electropolished 316L are not interchangeable if the hygienic target is defined by finish-related cleanability and inspection expectations.
Ignoring Weldability and Fabrication Reality
A correct base material with poor weld execution is still a weak GMP piping package.
Treating Documentation as a Post-Procurement Problem
In pharma projects, documentation is part of acceptance, not an afterthought for turnover.
Using One Material Rule for Every Process Area
High-purity water loops, product-contact transfers, CIP/SIP-exposed sections, and auxiliary areas do not all carry the same hygienic or compliance consequence.
Practical Selection Checklist for Pharmaceutical Hygienic Piping Materials
Questions to Answer Before Final Material Approval
- What process fluid or product actually contacts the surface?
- What CIP/SIP exposure will the system really see?
- What finish level is required for cleanability and inspection?
- Which local assemblies carry the highest contamination or retention risk?
- What documentation package is required for QA release?
- Is standard 316L enough, or does the finish and fabrication requirement change the answer?
What to Define in the RFQ
- Alloy grade
- Tubing standard
- Surface finish requirement
- Weld acceptance criteria
- Passivation / post-fabrication expectations
- Certificates and traceability deliverables
- Inspection requirements for release
Conclusion: Good Pharma Material Selection Is a System Decision, Not a Metal Purchase
Good pharmaceutical hygienic piping material selection is a system decision, not a metal purchase. The strongest projects choose materials by looking at corrosion resistance, finish, weldability, cleaning regime, QA documentation, and process risk together. That is why 316L is so common in critical GMP hygienic piping, but also why 316L alone is never the whole answer.
If you want a cleaner pharma topic cluster, connect this article with Electropolished 316L vs Standard 316L Stainless Steel, Sanitary Fitting Selection for GMP Facilities, and Documentation and Traceability in Pharma Piping Projects.
FAQ
Is 316L always required in pharmaceutical hygienic piping?
No. 316L is the practical baseline for many critical pharmaceutical hygienic systems, but the final choice still depends on process chemistry, finish requirement, weld density, validation expectations, and project risk.
What is the difference between electropolished 316L and standard 316L in pharma service?
The base alloy family may be the same, but the surface condition is not. Electropolished 316L is chosen where tighter finish consistency, cleanability, and inspectability are needed.
Can 304L still be used in pharmaceutical piping projects?
Sometimes. It may still appear in lower-risk or legacy applications, but critical GMP hygienic product-contact systems more often move toward 316L because the decision is broader than corrosion cost alone.
Why is documentation part of material selection in GMP projects?
Because QA release depends on proof, not only on intent. A correct material without traceability, certificates, and related inspection records is still a project risk.
What standards matter most when selecting pharma hygienic piping materials?
ASME BPE, FDA 21 CFR 211.65 and 211.67, ASTM A270, and the GMP framework including Annex 1 are among the most useful starting points. They shape how engineers define materials, fabrication quality, cleanability, and documentation expectations.
Does good alloy selection eliminate the need for weld and finish control?
No. A correct alloy with poor finish consistency, weak weld quality, or incomplete post-fabrication treatment can still fail the hygienic intent of the project.




