CIP design for hygienic piping is not just about choosing the right detergent, temperature, and cycle time. In real food, beverage, dairy, brewing, and hygienic process systems, the success of cleaning-in-place depends first on whether the piping is actually designed to be cleaned in place. Drainability, dead-leg control, branch geometry, valve integration, weld quality, seal compatibility, and verification access usually determine whether the cleaning program works consistently or keeps compensating for a design weakness.
That is why good CIP results often start with piping geometry rather than chemistry alone. A line can have a well-written CIP recipe and still show retained liquid, repeat ATP deviations, odor carryover, or local product buildup if the hardest parts of the system are not truly cleanable in place. This guide explains the design considerations engineers should review before approving a hygienic piping CIP layout, and how those decisions connect to fittings, materials, valves, and long-term maintenance performance.
What CIP Design Really Means in Hygienic Piping
CIP Design Is a Geometry Problem Before It Becomes a Chemistry Problem
A hygienic line becomes easy or difficult to clean largely because of its geometry before cleaning chemistry ever enters the system. If the piping does not drain, if branch connections create stagnant pockets, or if valves and instruments interrupt the flow path in hard-to-clean ways, stronger chemistry usually becomes a workaround rather than a durable solution. In practical plant work, this is one of the most common misunderstandings around CIP design: people adjust cycle parameters when the more basic problem is that the line is not behaving like a cleanable circuit.
EHEDG hygienic design principles make this point clearly by stating that equipment and facilities with poor hygienic design are difficult to clean. That principle applies directly to hygienic piping systems. If the local geometry is weak, cleaning consistency usually becomes fragile even when the overall system looks sanitary on paper.
Why “Sanitary-Looking” Piping Is Not Always CIP-Cleanable
Not every hygienic-looking pipe run is truly suitable for full CIP. Stainless steel tube, polished ferrules, and sanitary fittings help, but they do not automatically make the installed system easy to clean without breakdown. 3-A’s cleanability guidance directly addresses this misconception: not all 3-A conforming equipment is automatically suitable for full CIP, because standards and accepted practices can cover both manual cleaning and cleaning-in-place designs. For engineers, the practical meaning is simple: never approve a line as CIP-ready just because the hardware style looks hygienic.
If you need the upstream fitting logic behind that distinction, connect this article with how to select hygienic fittings for food processing and sanitary vs industrial pipe fittings.
Where CIP Design Usually Fails in Real Plants
CIP design failures usually appear first at details, not at the center of a straight tube. The repeated weak points are dead legs, poorly drained low spots, long sample branches, horizontal instrument take-offs, valve clusters, and assemblies that look accessible from the outside but are hard to flush internally. A common field finding is that the main loop data looks acceptable while one local branch keeps showing retained residue or repeat hygiene deviations.
| Typical Weak Point | Why It Matters in CIP | Common Plant Symptom |
|---|---|---|
| Dead-leg branch | Weak fluid exchange and poor local flushing | Residue or odor at one repeated location |
| Low point or poor drainback | Retained rinse water or chemistry | Carryover, dilution, or microbial risk |
| Valve or sample assembly | Internal cavities and shadow zones | Line looks clean but assembly needs repeated attention |
| Poor weld transition | Roughness and film retention | Slow cleaning response or hard-to-explain buildup |
| Wrong seal material | Chemical and temperature incompatibility | Leak recurrence, swelling, odor retention |
Start with the Real Process: Product, Soil, and Cleaning Regime
What Product Characteristics Change the CIP Design
Different hygienic systems foul differently, and that changes what the piping has to do during CIP. Sugar-based residues behave differently from protein soils. Fat-bearing products behave differently from yeast-heavy suspensions or viscous concentrates. Syrups, sauces, cultured products, flavor lines, beverage blends, and brewery yeast-bearing circuits all create different cleaning challenges, especially in branches, valves, and locally slow-moving areas.
This matters because the hardest soil to remove is usually not spread evenly across the system. The more useful design question is where the most persistent soil will accumulate and whether that location receives enough flow, coverage, and drainback to be cleaned without guesswork. In plant retrofits, the biggest cleaning problem is often not the main line product load, but one local feature that accumulates the most stubborn residue.
How Cleaning Chemistry and Temperature Affect Design Choices
Cleaning chemistry and thermal cycling are design inputs, not only operating parameters. Caustic, acid, sanitizer, hot water, and repeated heating and cooling affect not just the product soil, but also weld zones, seals, valve seats, branch geometry, and corrosion margin. That is why food-contact equipment rules require surfaces to withstand the intended environment, including food, cleaning compounds, sanitizing agents, and cleaning procedures. FDA 21 CFR 117.40 frames this as a design requirement, not merely an operations preference.
If material selection is part of the design review, connect this article with 316L Stainless Steel for Dairy and Brewery Applications.
Why the Same CIP Program Does Not Suit Every Hygienic Line
A CIP program that works on one hygienic circuit may not work on another circuit that looks similar from the outside. Hot-side, cold-side, dairy, ingredient, beverage, and brewery lines differ in soil type, consequence of residue, local branch complexity, and acceptable verification strategy. The same overall plant can have one loop that behaves predictably and another that repeatedly needs operator intervention because the geometry and local assemblies respond differently to the cleaning program.
A dairy loop with protein and fat carryover, for example, will often stress drainability and seal compatibility differently than a brewery line where yeast and biofilm risk dominate sampling branches and valve clusters. Good CIP design starts by accepting that “same plant” does not mean “same cleanability problem.”
Drainability: The First Design Requirement Most Teams Underestimate
Why Residual Liquid Is a CIP Design Failure, Not Just a Maintenance Issue
Retained liquid after cleaning is usually a design problem first and a maintenance problem second. Residual rinse water, diluted chemistry, or product heel left in a low point can change the next cleaning cycle, support growth, create carryover, and distort troubleshooting. In real systems, the repeated question should not be “Can maintenance drain this manually?” but “Why is the piping retaining liquid at all after a supposed self-draining cleaning cycle?”
This is one of the clearest points where design and regulation overlap. FDA equipment requirements expect food-contact surfaces to be adequately cleanable and maintained in a condition that protects food. A piping circuit that predictably traps liquid after cleaning is already underperforming from that point of view.
A common commissioning example is a line that drains correctly on paper but develops a persistent low point after support adjustment or field routing changes. The CIP program is then blamed for dilution or carryover, even though the real issue was installation geometry.
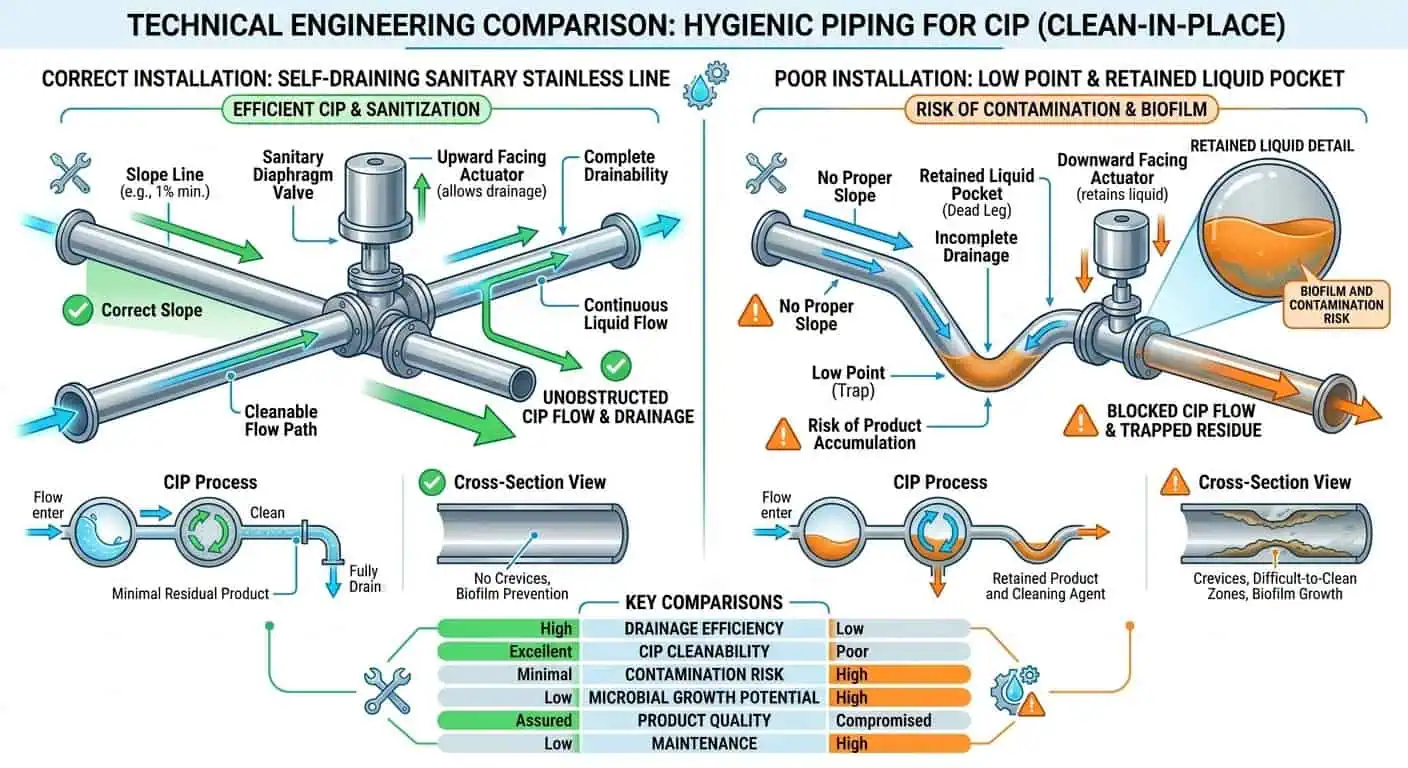
Typical Low-Drain Locations in Hygienic Piping
Low-drain risk often develops at places teams do not initially treat as “major” piping features. Unsupported spans, valve bodies that do not empty well, dead-end sensor tees, sample points, reducer transitions, and poorly oriented branches are typical examples. In practice, one small local low point can cause more cleaning instability than a much longer straight run of otherwise acceptable pipe.
What Engineers Should Review for True Self-Drainability
Drainability has to be checked as an installed reality, not assumed from a drawing symbol. Review line slope, support spacing, valve orientation, branch take-off angle, drain point location, and whether the worst local assembly still empties after the CIP sequence. One recurring commissioning case is a line that was theoretically sloped but changed behavior after support adjustment, resulting in a repeated low point that only became obvious after startup.
Dead Legs, Branches, and Flow Shadows
Why Dead Legs Remain One of the Most Common CIP Design Mistakes
Dead legs remain one of the most common CIP design errors because they are easy to create and easy to underestimate. They appear not only in original design, but also during plant modifications when an instrument, sample point, temporary tie-in, or utility connection is added without enough attention to local cleanability. In hygienic piping, a dead leg is not just an inefficient branch. It is a repeated challenge to fluid exchange, residue removal, and verification confidence.
EHEDG’s FAQ on cleanability and CIP testing is useful here because it emphasizes that internal wetted surfaces and critical areas must be evaluated for cleanability, and that different sizes or configurations are not always equally cleanable just because they share the same design family. That logic applies directly to branches and dead-leg features.
How Branch Geometry Changes Cleaning Effectiveness
Branch geometry changes cleaning effectiveness by changing local fluid exchange, not only by changing pipe layout. A short, well-oriented branch may be manageable. A longer or poorly positioned branch may become a local flow shadow that never receives the same wall shear, fluid replacement, or drainback as the main line. In practical engineering terms, the branch is no longer just a connection detail. It becomes the dominant cleaning problem in that zone.
A Common Industry Case: The Main Loop Cleans, the Branch Does Not
One of the most common hygienic piping cases is a branch that fails repeatedly even though the main loop appears to clean well. The return-side temperature, conductivity, and time data look acceptable. Operators assume the cleaning cycle has succeeded. Later, one sample branch, instrument connection, or valve assembly still shows retained residue or repeat ATP concern. The root cause is usually not insufficient detergent strength. It is weak local flow, branch geometry, or poor drainability.
The more durable correction is usually to shorten, reorient, simplify, or redesign the branch instead of endlessly escalating cleaning parameters.
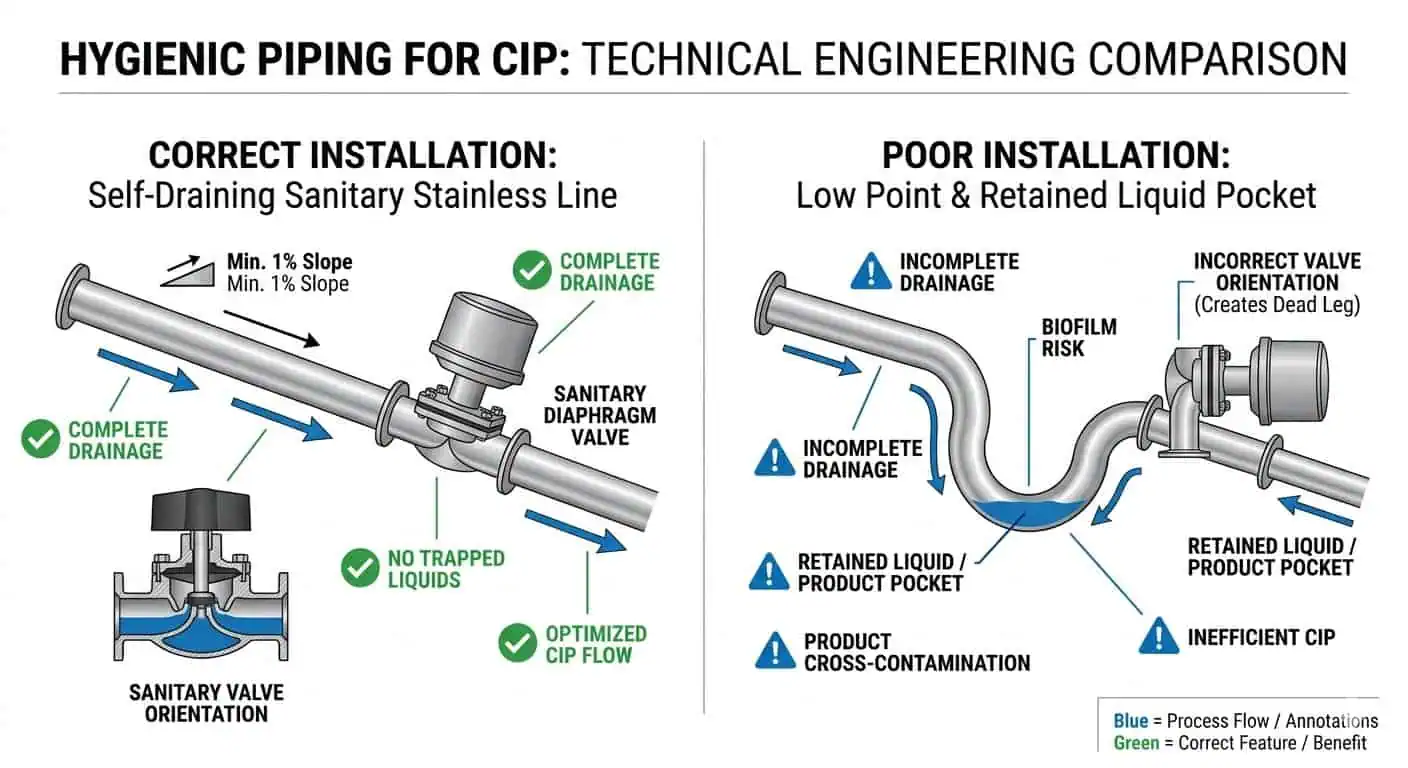
Flow Path Design: Velocity, Coverage, Circuit Logic, and Return Behavior
Design the Circuit for Cleaning, Not Only for Production Flow
A line that transfers product well does not automatically clean well. Production flow and cleaning flow are not the same design problem. A line can meet process throughput and still create weak local flushing during CIP because valves are sequenced poorly, branches are not balanced, or the cleaning circuit was never designed around the hardest-to-clean feature in the first place.
Why Return-Side Data Can Be Misleading
Return-side CIP data is useful, but it can create false confidence if engineers stop there. Temperature, conductivity, and cycle time may look acceptable at the return point while one local assembly remains under-cleaned. This is why plants sometimes experience a repeated issue at one branch or valve while the larger circuit keeps “passing” operational checks.
Engineering note: return-side stability proves that the overall circuit is running. It does not prove that every critical local assembly is exchanging fluid effectively enough to clean.
What to Review in CIP Circuit Layout
Review the circuit as a cleaning path, not just as a piping map. Check supply and return logic, valve sequencing, branch isolation, segmentation between loops, potential stagnant pockets, and whether local assemblies are truly included in the effective cleaning path. The better review question is not “Can the liquid reach this area?” but “Can the liquid clean and replace itself in this area with enough consistency to support repeatable verification?”
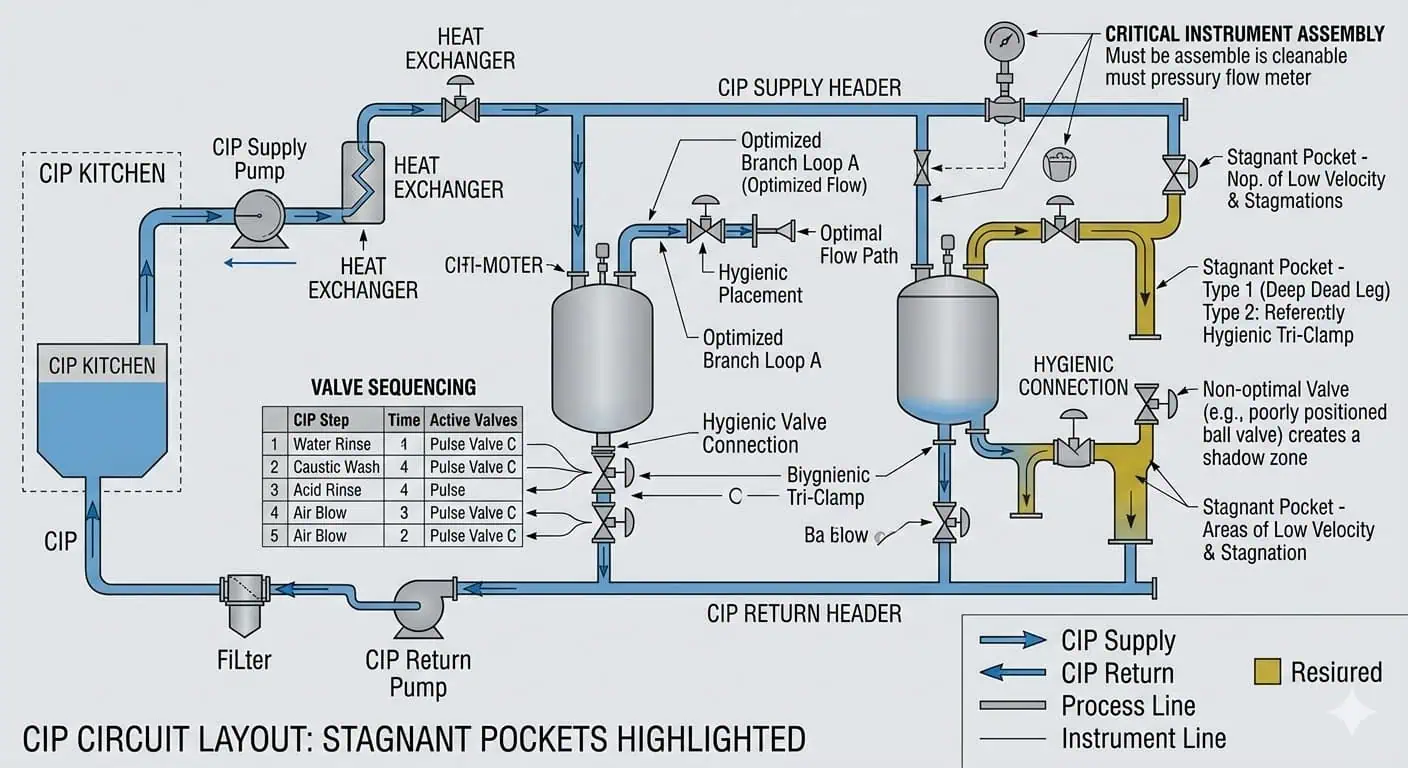
Valves, Instruments, and Sample Points: The Real Difficulty in Hygienic Piping CIP
Why Valves Are Harder to Clean Than Straight Tube
Valves are harder to clean because they contain internal details that straight tubes do not. Seats, cavities, internal transitions, and seal surfaces make them more sensitive to flow path design, cleaning sequence, and local drainback. In hygienic piping, a valve is often the first place where cleanability assumptions become visible as actual cleaning instability.
Instrument Tees, Sensors, and Sample Assemblies
Instrument tees, hygienic sensors, and sample assemblies deserve more CIP scrutiny than their size suggests. They are small enough to be overlooked and complex enough to create local retention or weak flushing. In practice, many repeat hygiene findings originate at these points rather than at the main pipe run.
If your process uses modular brewery or beverage connections with frequent sample access, connect this section with Tri-Clamp Fittings in Brewing Systems.
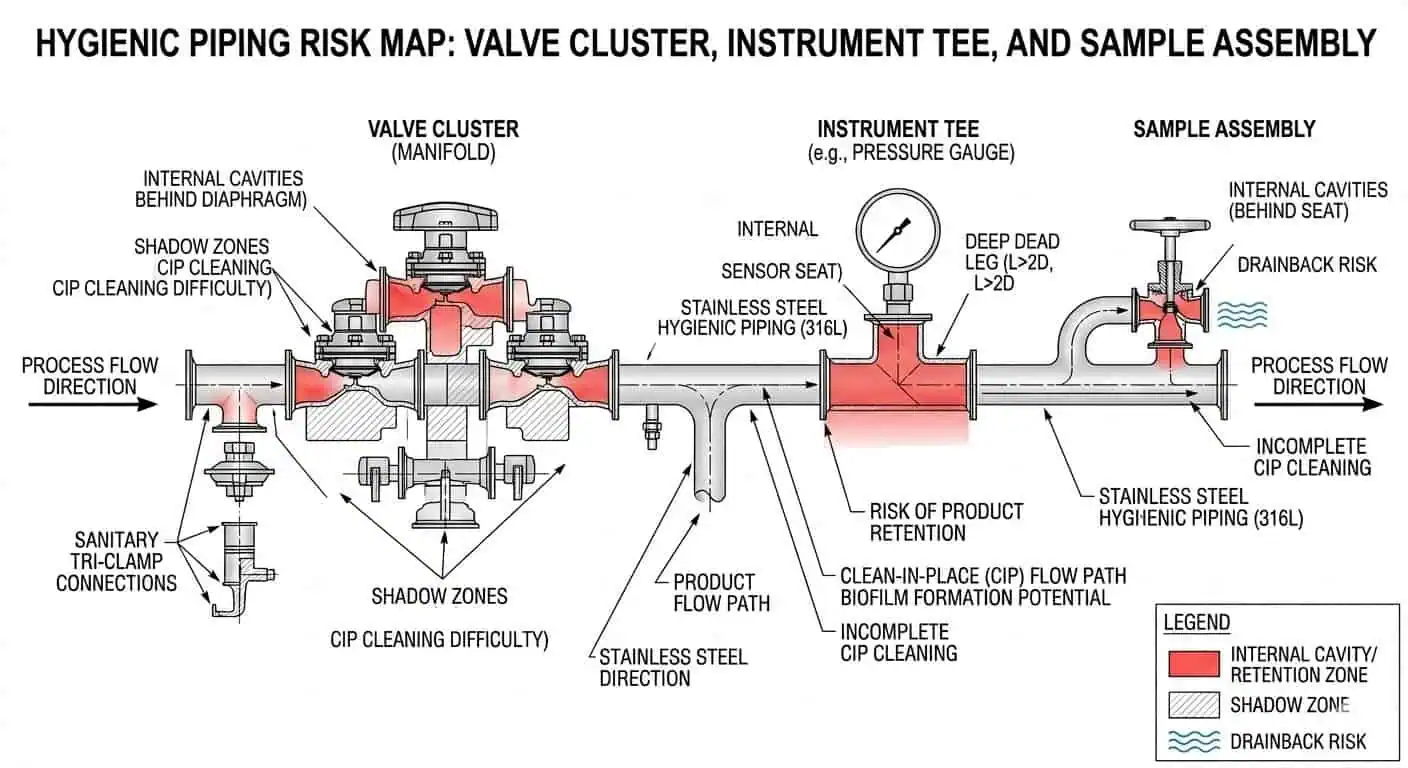
Design for Access, Verification, and Controlled Disassembly
Not every local assembly should be treated as permanently full-CIP without inspection. Some systems need targeted review, occasional breakdown, or specific verification around valves, sample points, and branches. That is not necessarily a design failure. It becomes a design failure only when the system is presented as fully in-place cleanable without providing realistic access or verification logic where it is actually needed.
For post-startup discipline, link this article with Cleaning and Maintenance of Sanitary Piping Systems.
A common packaging-line example is a sample assembly that stays acceptable during routine operation but begins showing repeat odor or ATP concern after product changes. The issue often turns out to be not the main cleaning circuit, but a small, hard-to-verify local assembly that was assumed to be “covered” by the overall CIP loop.
Materials, Surface Finish, Welds, and Seals
Material Selection Must Match Product and Cleaning Environment
Material selection in hygienic piping should be tied to both product exposure and cleaning exposure. Corrosion margin, temperature resistance, and seal compatibility all affect how consistently the line can be cleaned over time. The correct material choice is the one that remains stable under the actual food, beverage, cleaning compound, and sanitizing conditions the circuit will see in service.
Why Surface Finish and Weld Quality Directly Affect CIP
Surface finish and weld quality directly affect CIP because they change residue retention and cleanability at the product-contact surface. Rough internal welds, oxidation, poor contour, or hard transitions can make one local section slower to clean than the rest of the circuit. This is why hygienic weld quality should be reviewed as part of CIP design approval, not treated as a separate fabrication detail that has no cleaning consequence.
EHEDG’s hygienic welding guidance is useful here because it frames weld quality as a hygienic performance issue, including how to achieve and verify product-contact welds in stainless process tubing.
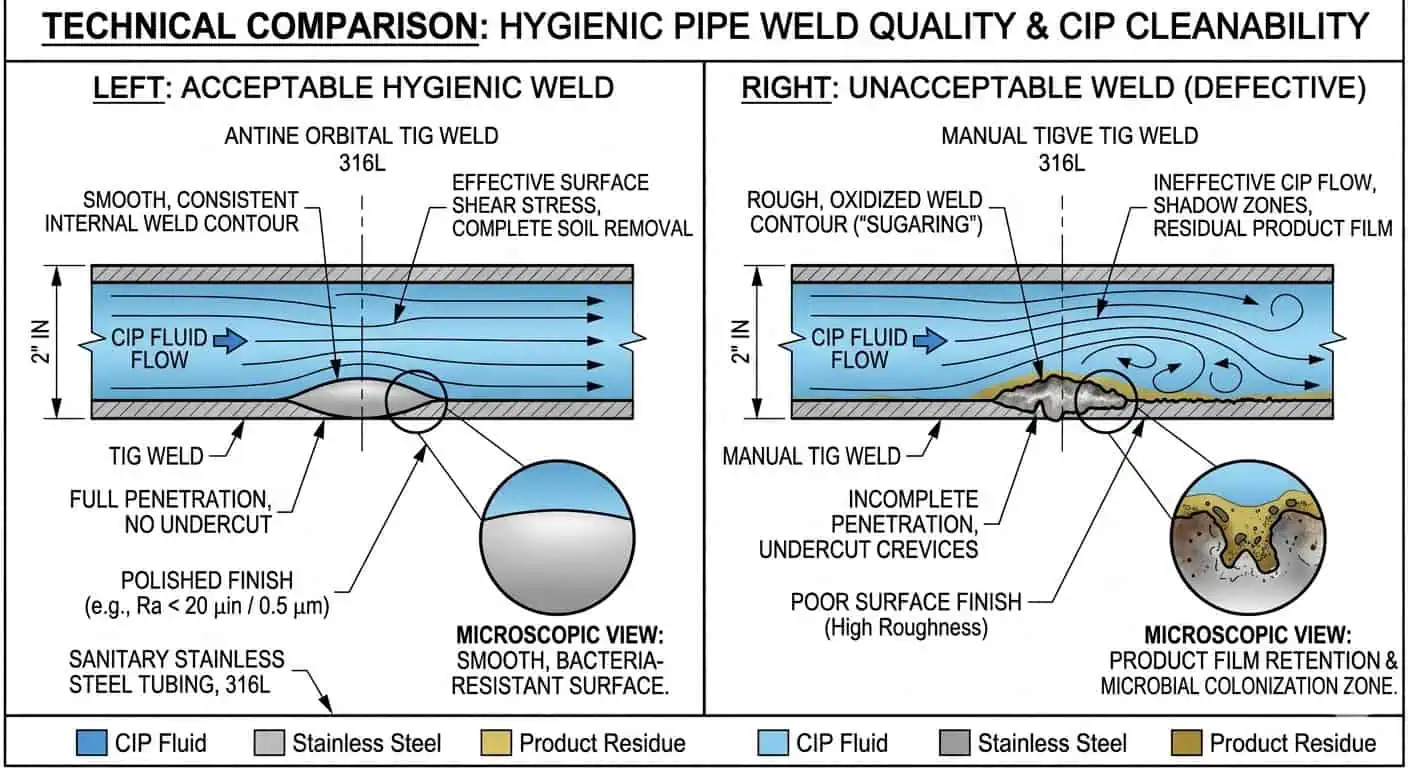
Gasket and Seal Compatibility Is Part of CIP Design
Gaskets and seals are part of CIP design because they shape the real boundary between chemistry, temperature, and hygiene. Swelling, loss of recovery, odor retention, chemical incompatibility, and repeated compression damage can all reduce both seal reliability and cleanability. This is one reason why seal material cannot be left to last-minute purchasing substitution in a hygienic piping project.
A realistic field example is a line that shows repeated odor retention or unstable sealing only after several cleaning cycles. The metal is usually not the first thing that failed. It is often the seal material interacting with chemistry, heat, compression history, and reassembly practice.
Validation, Verification, and Proof That the Piping Is Actually CIP-Cleanable
Design Review Is Not Enough—You Need Verification Logic
A clean design review does not prove cleanability by itself. It reduces risk, but verification is still needed to show that the installed system behaves as intended. This is especially important where branches, valves, different equipment sizes, or complex local assemblies are involved.
What Cleanability Testing Actually Tries to Prove
Cleanability testing tries to prove that an item or internal wetted surface can actually be cleaned in place to an acceptable level, not merely that it looks hygienic. EHEDG’s FAQ and the updated Guideline 2 release note both reinforce that CIP suitability is tied to evaluation and testing of in-place cleanability for internal wetted parts, and that different sizes or configurations are not always equally cleanable without review.
What Plants Commonly Use for Verification
Plants commonly rely on a mix of methods to verify CIP performance. Depending on the process and risk level, this can include visual inspection, targeted swab work, ATP where justified, local microbiological verification, or specific cleanability testing during qualification. The best approach is not the one that generates the most paperwork. It is the one that confirms the hardest local assembly is performing as intended.
There is also a practical business case for getting this right earlier. In its 2025 introduction, EHEDG cites case examples such as a 30% reduction in CIP water use at a Nestlé dairy facility, a 2–5% product yield increase in a brewery implementation, and a 15–20% reduction in downtime between production runs on a beverage line when hygienic design and CIP performance improved. Those examples are not a guarantee for every plant, but they show why CIP-ready piping design is both a hygiene issue and an operating-cost issue.
Practical CIP Design Checklist for Hygienic Piping
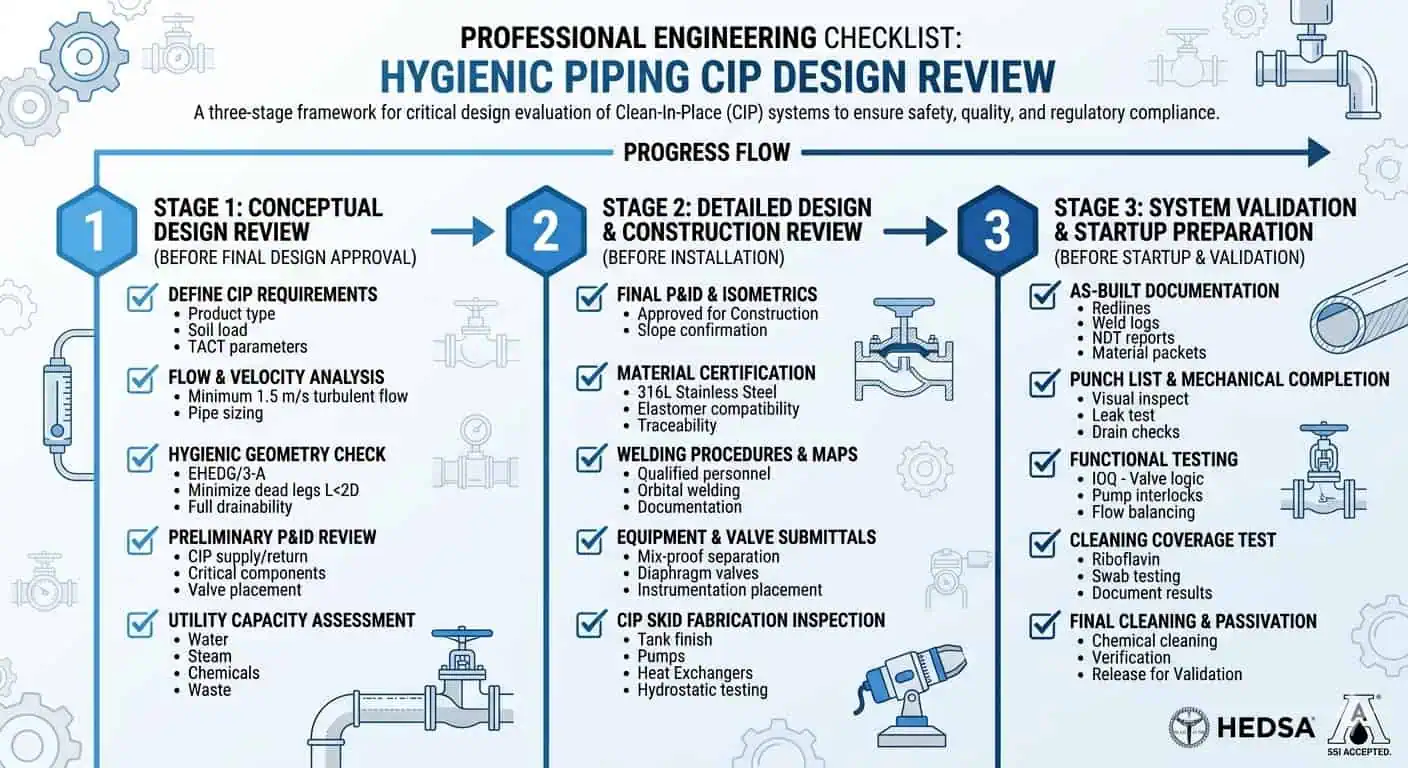
Before Final Design Approval
- Is the line genuinely self-drainable under installed conditions?
- Are dead legs, sample branches, and low-flow features controlled?
- Are valves, instruments, and local assemblies integrated with CIP in mind?
- Do materials and seals match product and cleaning exposure?
- Is the hardest local assembly still cleanable, not just the main line?
Before Installation
- Confirm support spacing and slope behavior
- Review ferrule and weld quality at hygienic connections
- Check valve orientation and branch take-off direction
- Confirm access for inspection or controlled breakdown where needed
- Verify that no obvious low-point traps were introduced during fabrication
Before Startup and Validation
- Confirm supply and return circuit logic
- Identify the hardest-to-clean local assemblies
- Define verification points before production pressure begins
- Document what “acceptable cleanability” means for the circuit
- Make sure the CIP program is matched to the real installed geometry
Conclusion: Good CIP Performance Starts with Good Hygienic Piping Design
Good CIP performance starts with good hygienic piping design, not with stronger chemistry alone. Stable CIP results depend on drainability, dead-leg control, correct branch and valve integration, compatible materials and seals, and a verification method that proves the hardest local assembly is actually cleaning as intended. In most plants, repeated cleaning trouble is a sign that one of those fundamentals was missed or weakened.
For a full hygienic piping topic cluster, connect this article with how to select hygienic fittings for food processing, sanitary vs industrial pipe fittings, Tri-Clamp Fittings in Brewing Systems, 316L Stainless Steel for Dairy and Brewery Applications, and Cleaning and Maintenance of Sanitary Piping Systems.
FAQ
What is the most important CIP design factor in hygienic piping?
The most important factor is usually drainable, cleanable geometry. If the system does not drain well, contains dead legs, or creates poorly flushed local assemblies, stronger chemistry and longer cycle time rarely solve the problem permanently.
Can a hygienic-looking pipe system still be hard to clean?
Yes. Hygienic-looking hardware does not guarantee real CIP cleanability. Local geometry, valve details, weld quality, seal choice, and branch orientation still determine whether the installed system is actually easy to clean in place.
Why do dead legs matter so much in CIP design?
Dead legs reduce local fluid exchange and make residue removal less reliable. They are one of the most common reasons a branch or sample point keeps failing even when the larger CIP loop appears to perform normally.
Does higher cleaning chemical concentration solve poor piping design?
Not reliably. It may reduce symptoms in some cases, but if the real problem is poor drainability, weak branch flow, or a hard-to-clean local assembly, design correction is usually more effective than endlessly increasing chemistry.
How should valves and sample points be handled in CIP design?
They should be treated as higher-risk assemblies than straight tube. Review their orientation, local flow path, internal cleanability, drainback behavior, and whether they need targeted inspection or controlled disassembly rather than blanket full-CIP assumptions.
What is the difference between CIP design review and CIP validation?
CIP design review asks whether the piping should be cleanable in place. CIP validation or verification asks whether the installed system actually performs that way. Both are necessary if the circuit includes critical branches, valves, instruments, or high-consequence hygienic zones.




