
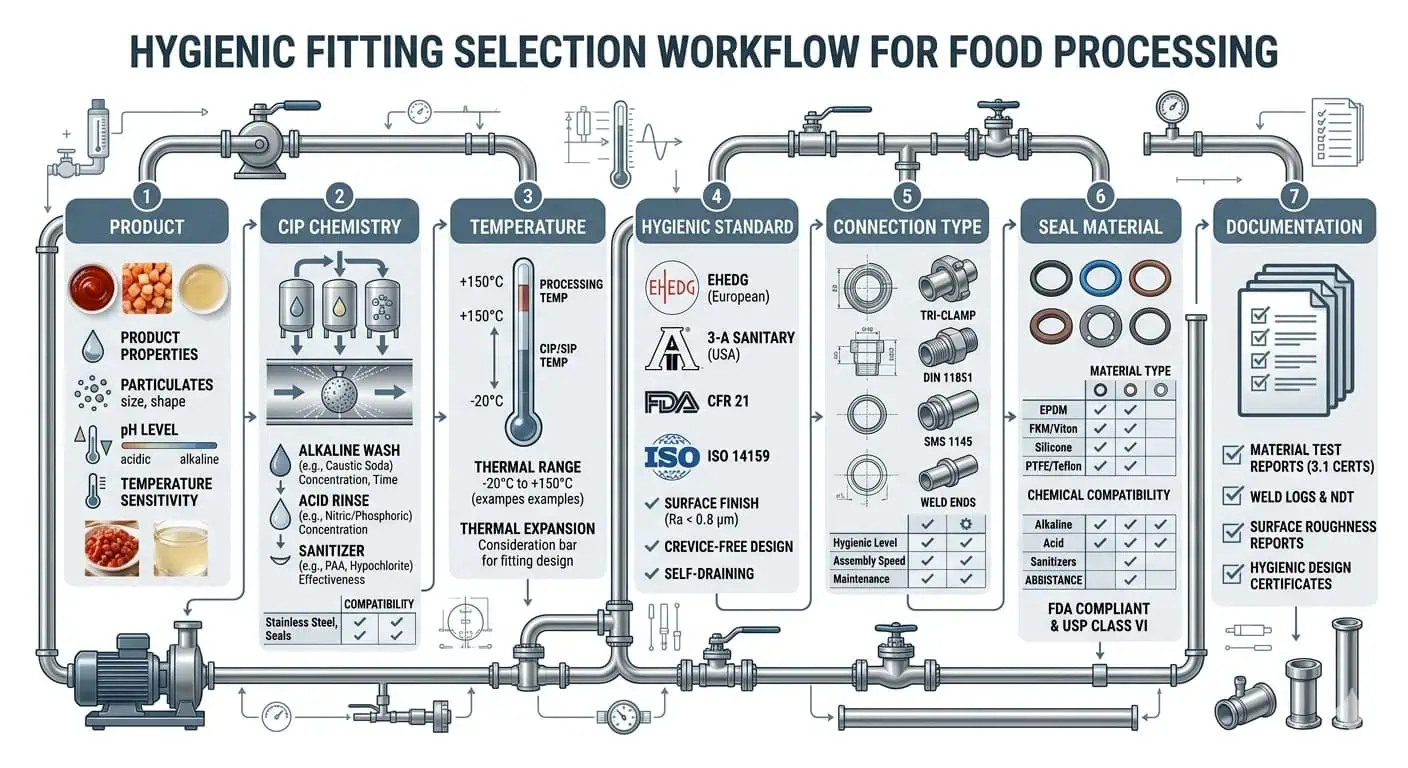
How to select hygienic fittings starts with matching your actual product, cleaning chemistry, and line geometry, then verifying hygienic design, food-contact suitability, material compatibility, and installation details. That sequence matters because many fitting failures in food plants do not begin as obvious mechanical failures. They usually start as product hold-up, poor drainability, gasket incompatibility, weld defects, or missing documentation, then turn into contamination findings, repeat CIP failures, leaks, or audit nonconformities. In food processing, the fitting-related issues that most often create trouble include:
- Missing or incomplete material traceability, gasket compound records, or surface-finish evidence for product-contact parts
- Dead-leg branches around instruments, sample points, or valve manifolds that do not clean or drain consistently
- Wrong gasket material for the actual product, caustic, acid, sanitizer, or hot-water/SIP exposure
- Ferrule mismatch, poor internal weld finish, or clamp assemblies forced into alignment during installation
You need practical selection logic, not generic catalog language. This guide focuses on what actually affects cleanability, drainage, seal life, changeover control, and audit readiness in food and beverage service. SUNHY can then support the supply side with documentation, material traceability, and application review once your technical requirements are clear.
Start with the Real Food Process, Not the Catalog
Product and Media Requirements
You must match fittings to the real product and process media, not to a general product description. In food processing, the medium is not just “liquid food.” You need to define pH, fat content, protein load, sugar concentration, chloride exposure, viscosity, particulate size, allergen profile, and whether the line sees caustic, nitric acid, peracetic acid, hot water, or steam during cleaning. Each factor changes what the fitting has to survive and how easy it will be to clean.
- Material compatibility—verify that the body alloy, gasket, seat, and any lubricant exposure are suitable for both the product and the actual CIP/SIP chemistry. A fitting that handles the product but swells or cracks under sanitizer is still the wrong fitting.
- Temperature envelope—check normal production temperature, CIP temperature, hot-water sanitizing temperature, and any upset condition. Temperature affects corrosion rate, gasket compression set, and published pressure capability.
- Cleaning exposure—confirm whether the assembly must tolerate repeated chemical cleaning, high-flow CIP, foam cleaning, manual teardown, or steam sterilization. Cleaning method changes both design preference and maintenance burden.
- Surface finish and weld condition—smooth, correctly finished product-contact surfaces reduce residue retention and make validation easier, but only if the internal welds, ferrule alignment, and heat tint condition are also controlled.
If you process dairy, beverages, or high-protein foods, this is where many avoidable mistakes begin. A common field example is a sauce or brine line specified in 304L for general service, then exposed to more aggressive chloride-bearing cleaning or product residue at clamp crevices. The visible symptom may start as tea staining or local pitting near the seal land, but the root cause is usually a chemistry-and-crevice mismatch rather than “bad stainless” alone.
Process Mode and Cleaning Strategy
Your cleaning strategy determines which fitting design is practical and which one only looks acceptable on paper. A line designed for closed, repeatable CIP does not want the same connection strategy as a short batch skid that is frequently opened for changeovers. You need to decide early whether the system is mainly closed and automated, routinely disassembled, or a hybrid of both.
- The equipment layout and whether the process is fully closed, partially open, or frequently broken for inspection
- The level of automation in cleaning, validation, and changeover control
- The trade-off between quick disassembly, seal life, downtime, and cleanability consistency
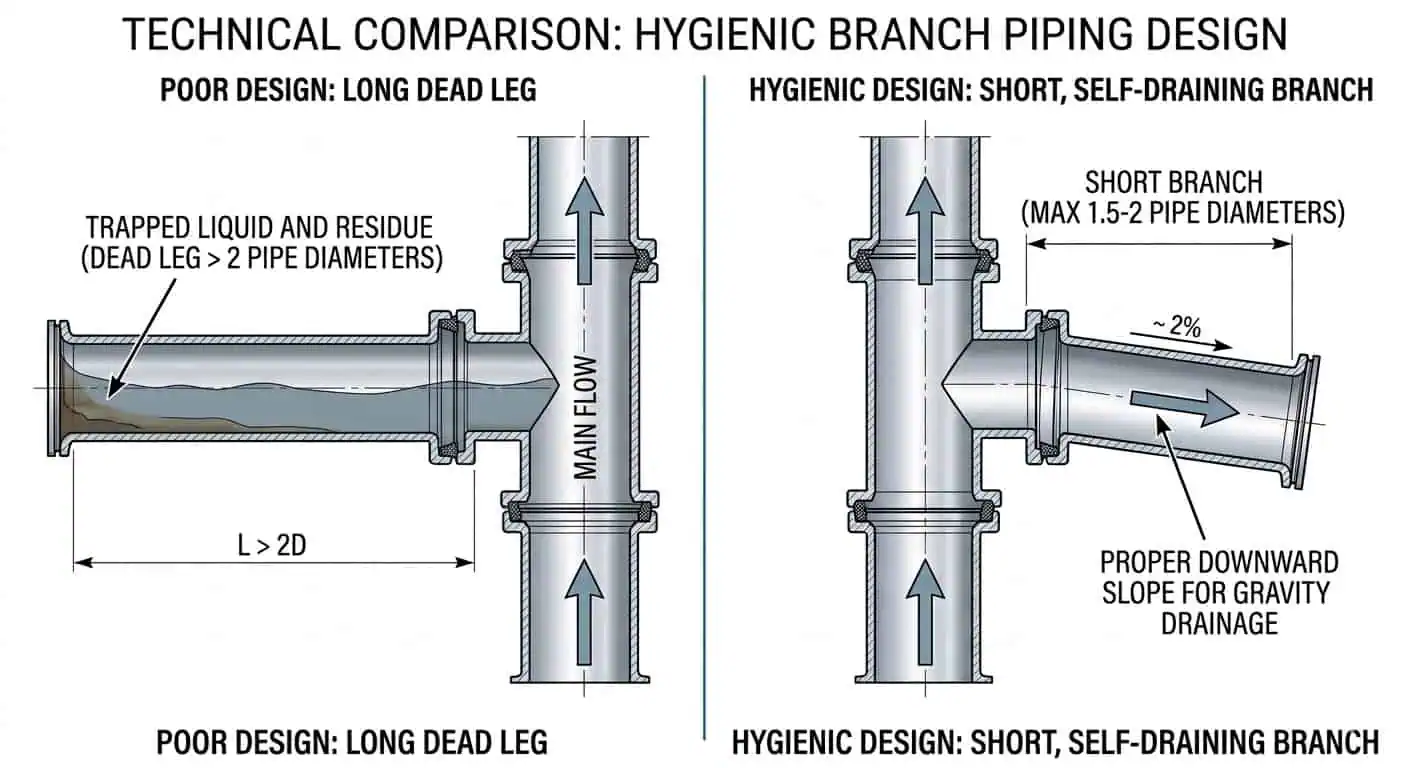
CIP service usually favors fittings and valves with predictable internal geometry, good drainability, and minimal product traps. Manual cleaning can tolerate more operator intervention, but it also raises variation between shifts. One practical example is a dairy sample point installed on a long branch off the main line: the line may pass a normal CIP cycle, yet the sample branch can still retain residue because velocity, wetting, and drainage are poor in that pocket. The fix is usually geometric—shorter branch, better orientation, or a cleaner sample-valve design—not stronger chemicals.
Size, Flow, and Connection Compatibility
You must ensure fittings match the hygienic tube standard, actual flow duty, and connection system used on the line. This is not only a size question. In hygienic service, the wrong standard family can create tolerance mismatch, weld mismatch, seal problems, poor drainability, or replacement confusion during maintenance. Use the table below as a practical reference framework:
| Attribute | Specification |
|---|---|
| Reference standards | Common hygienic projects may involve BS 4825, EN 10357 / DIN 11850-family tubing, DIN 32676 clamp connections, DIN 11864 aseptic connections, or ASME BPE on higher-purity lines; the line standard must match the fitting standard. |
| Surface finish roughness (typical project requirement) | Ra ≤ 0.8 μm is a common baseline for many hygienic product-contact surfaces, but final acceptance should follow the applicable customer specification, tube/fitting standard, and weld requirement. |
| Common body materials | 304L and 316L are the most common stainless choices for hygienic fittings; 316L is often preferred when chloride exposure, aggressive cleaning, or higher corrosion margin is part of the duty. |
| Compatibility check | Verify OD, wall thickness, tolerance, ferrule profile, gasket geometry, and clamp series together. “Same nominal size” does not guarantee interchangeable hygienic parts. |
| Typical applications | Dairy, beverage, brewing, liquid foods, ingredient transfer, biotech-adjacent food systems, and higher-cleanability utility/process interfaces. |
| Installation implication | Correct standard matching reduces forced fit-up, internal step formation, gasket damage, and future spare-parts confusion. |
| Maintenance implication | Standardized connection families make seal replacement, stock control, and audit traceability much easier over the life of the line. |
By starting with the real process instead of the catalog, you avoid a common purchasing mistake: selecting by price and nominal size first, then discovering later that the fitting family, gasket compound, or internal geometry does not match the plant’s cleaning reality.
Identify the Hygienic Risk Before Choosing the Fitting Type
Where Contamination Usually Starts
Contamination in food lines often starts at transitions, seals, and low-flow details—not in the middle of a straight tube. That is why connection points deserve more attention than the catalog usually gives them. In real plants, the main contamination or cleaning-failure triggers around fittings are:
- Particles—weld slag, rouge, metal fines, damaged gasket fragments, and installation debris left in the line after fabrication or maintenance
- Water and condensate—poor drainage at horizontal take-offs, low points, valve bodies, or improperly sloped spool pieces
- Lubricant or utility carryover—especially where compressed air or gas is introduced near product-contact equipment and has not been treated appropriately
- Microorganisms and residue films—most commonly where there are dead spaces, rough welds, damaged seals, shadow areas, or incomplete CIP wetting
A practical troubleshooting pattern is this: when the same connection keeps showing ATP hits, micro positives, or odor retention after an otherwise acceptable wash, the cause is often local geometry, seal condition, or drainage orientation rather than inadequate detergent concentration across the whole plant.
Cleanability and Drainability
Cleanability and drainability are the two design filters that separate “food-grade looking” fittings from fittings that actually behave hygienically in service. You need fittings designed to minimize residue retention and to empty consistently after product transfer and after CIP. Look for these features:
- Avoiding crevices—product-contact joints should not create narrow, stagnant pockets where residue survives chemical contact but remains difficult to flush
- Smooth, continuous internal surfaces—surface finish only helps when it is paired with flush alignment, clean welds, and intact seal lands
- Self-draining geometry—branches, valve bodies, and spool orientation should allow full drainage rather than leaving a heel of product or rinse water
- Rounded transitions and no sharp internal shoulders—abrupt internal steps increase product hold-up and cleaning shadow zones
- Controlled seals and shaft passages—elastomers must seal without extrusion, tearing, or chronic over-compression, because damaged seals quickly become hygienic weak points
One repeated field issue is clamp joints that look fine from outside but are assembled across slightly mismatched ferrules. Operators tighten the clamp until the leak stops, but the gasket is then pinched unevenly, creating an internal lip and shortening seal life. The short-term result is a dry line that “holds pressure.” The long-term result is harder cleaning, repeat leak calls, and premature gasket replacement.
Risk-Based Selection by Product Sensitivity
Product sensitivity should change your selection threshold. Not every food line carries the same hygienic consequence if a fitting traps residue or a seal starts to degrade. Low-acid dairy, high-protein beverages, allergen changeover systems, infant-nutrition ingredients, aseptic interfaces, and microbiologically sensitive products deserve a more conservative selection approach than low-risk utility transfer or short-run noncritical service. In higher-risk lines, you should favor 316L where chemistry justifies it, minimize branch dead spaces, tighten weld and finish control, and use connection systems that are easier to inspect and document. On lower-risk lines, the right answer may still be a simpler connection—but it must still be cleanable, drainable, and chemically compatible with the actual process.
Compliance and Standards for Hygienic Fittings
Food Safety and Food-Contact Requirements
You must select sanitary fittings that satisfy both hygienic design expectations and food-contact material requirements under the market you serve. In practice, that means you should stop using blanket language like “FDA approved fitting” unless you can point to the exact material or component scope. For hygienic fittings, the correct approach is to verify the assembly through material grade, elastomer declaration, finish, weld quality, intended conditions of use, and traceability records.
| Requirement Type | Description |
|---|---|
| 3-A Sanitary Standards | Widely used hygienic design criteria for food and dairy equipment. Useful when the processor, specifier, or regulator expects 3-A design language or a 3-A Symbol-covered equipment model. |
| FDA 21 CFR 117.40 | Requires food-contact surfaces to be adequately cleanable, corrosion-resistant when in contact with food, made of nontoxic materials, and able to withstand intended food and cleaning exposure. |
| EU Regulation (EC) No 1935/2004 | Requires food-contact materials and articles to avoid harmful migration and unacceptable changes to food composition or organoleptic properties under normal or foreseeable conditions of use. |
- Food-contact suitability must be checked for the actual body alloy, elastomer, seat, and any ancillary material that may contact product.
- Compliance evidence is strongest when it is compound-specific and condition-of-use-specific, especially for seals and gaskets.
Tip: In hygienic projects, ask for evidence at assembly level whenever possible—body material, seal compound, finish condition, and traceability—not just a generic statement copied from a catalog footer.
Hygienic Design Standards and Accepted Practices
You need to understand which standards govern design, which govern food-contact safety, and which govern plant food-safety systems. These are not interchangeable. For most food and beverage projects, the core references usually include:
- 3-A SSI standards for hygienic equipment design criteria and, where specified, 3-A Symbol authorization with third-party verification
- EHEDG hygienic design principles for risk-based hygienic design and cleanability thinking
- FDA CGMP / Preventive Controls framework for sanitation, plant, process, and contamination-control obligations in the U.S.
Food-safety system standards such as ISO 22000, FSSC 22000, BRCGS, or IFS matter too, but they do not replace the need to select a fitting geometry that actually drains and cleans. Documentation and HACCP review do not rescue a poor branch design.
Certifications and Documentation
You must treat documentation as part of the hygienic specification, not as an afterthought after purchase. For critical food-contact fittings and valves, the minimum useful document pack should be defined before the RFQ is sent.
- Identify the applicable standard family and customer requirement first—3-A, EHEDG-based internal spec, FDA/U.S. food-contact expectation, EU food-contact requirement, or a plant-specific hygienic standard.
- Confirm what evidence is needed for the exact component: material certificate, gasket declaration, finish statement, pressure-temperature data, weld or passivation record, and traceability marking.
- Where 3-A Symbol coverage is required, verify that the exact equipment model or component scope is actually covered and not assumed by brand reputation alone.
- Ask for sample documents before approval, including EN 10204 3.1 mill certificates where relevant, seal material declarations, and any cleanability-relevant product data.
- Make sure the document trail can still connect the installed fitting to the purchase record, lot, heat, or component batch during an audit or product investigation.

A common audit problem is not that the fitting itself is wrong, but that the plant cannot quickly prove what was installed after maintenance. When a gasket is replaced with a visually similar but different compound, the hygienic risk may stay hidden until swelling, odor retention, or chemical attack appears in service.
Note: For hygienic fittings, the most useful records are the ones that answer four questions immediately: what material, what seal compound, what finish condition, and what lot or heat was installed.
Material and Design Selection for Food Processing
Stainless Steel and Non-Metal Materials
You should choose materials that resist the actual product, the actual cleaning chemistry, and the real maintenance pattern of the line. Stainless steel remains the standard body material for most hygienic fittings, but hygienic performance depends on more than grade name alone. Finish condition, weld quality, passivation, crevice design, and the seal compound usually decide whether the fitting stays trouble-free in service.
| Material Choice | Corrosion / Service Consideration | Best Use Case |
|---|---|---|
| 304L stainless steel | Common and economical for many neutral food and beverage duties, but less forgiving where chlorides, aggressive cleaners, or persistent crevice exposure are part of the service. | General hygienic service where chemistry is moderate and the corrosion margin is well understood. |
| 316L stainless steel | Usually preferred where chloride-bearing product, salt, acidic cleaning, or higher corrosion margin is expected; also widely specified for more demanding hygienic environments. | Dairy, brine, sauce, ingredient systems, more aggressive CIP regimes, and higher-risk hygienic circuits. |
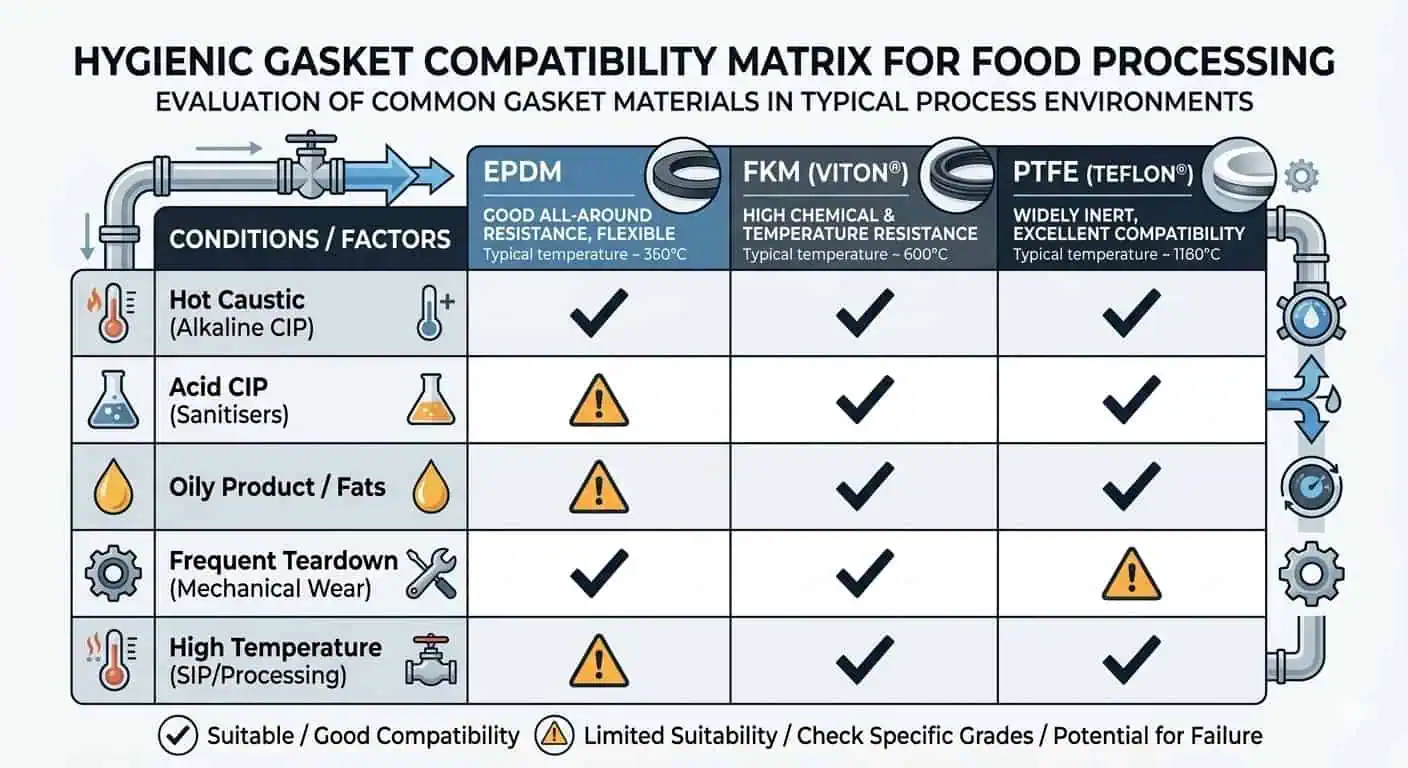
| Seal materials such as EPDM, FKM, PTFE, or others | Selection depends on temperature, fat/oil exposure, sanitizer chemistry, compression set risk, and whether frequent disassembly is expected. | Choose by compound compatibility and operating envelope, not by habit or what was already in stock. |
In practice, seal selection causes almost as many hygienic problems as metal selection. One common example is an elastomer gasket that survives the product but swells after repeated hot caustic and acid cycles. The visible failure may appear as clamp leakage or a gasket that “walks” during reassembly, but the root cause is chemical compatibility plus compression history.
You may also see PTFE-faced or PTFE-based sealing components used where chemical resistance is the dominant driver. That can work well, but seal resiliency, seating behavior, and assembly handling must still be considered. A chemically resistant seal that does not seat reliably in the real clamp assembly is not a hygienic upgrade.
Hygienic Fitting and Valve Types
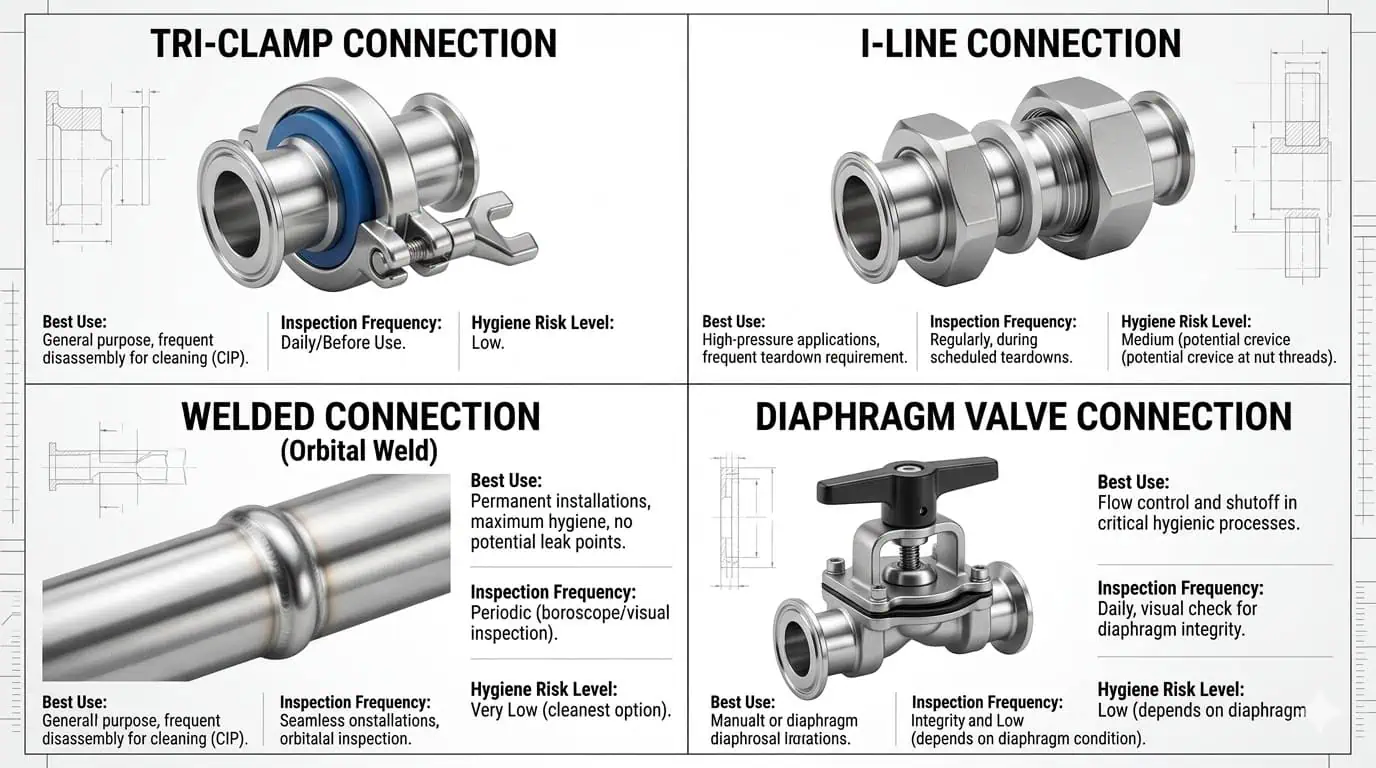
You need to select the fitting and valve type according to the process step, inspection frequency, and hygienic consequence of failure. The most common hygienic connection and valve choices include:
- Tri-clamp / clamp connections: Fast to open and widely used. Best where inspection and routine seal replacement matter, but only when ferrule alignment, gasket choice, and clamp series are correct.
- I-Line or heavy-duty hygienic mechanical connections: Useful where higher mechanical robustness, vibration resistance, or pressure capability is needed compared with light clamp service.
- Welded hygienic connections: Preferred where you want the least disassembly and the fewest mechanical joints, but they demand stronger fabrication and borescope-quality discipline.
- Butterfly valves: Common for on/off isolation in hygienic lines and relatively easy to maintain, but seat wear and body drainability still matter.
- Ball valves: Can work in some food duties, especially where shutoff is important, but internal cavity behavior and cleanability must be reviewed carefully for hygienic service.
- Diaphragm valves: Often favored where cleanability, sterility, or product segregation are more critical.
- Check valves, sample valves, and relief devices: Necessary for flow protection and sampling, but these are frequent dead-space creators when branch length and installation angle are not controlled.
You should match each valve or fitting type to the product behavior, cleaning method, opening frequency, and consequence of internal residue. A fitting that is technically hygienic but awkward to inspect will often become a maintenance blind spot.
Hygienic Design Features
You must prioritize design details that directly influence residue retention, inspection access, and seal reliability. The table below highlights the design features that matter most in daily operation:
| Feature | Description |
|---|---|
| Drainability | Connections, branches, and valve bodies should empty consistently after production and after CIP. Residual heel volume is a recurring contamination risk. |
| Crevice and weld control | Flush alignment, full-penetration hygienic welds, and minimal internal steps reduce residue retention far more effectively than appearance-only polishing. |
| Seal control | Correct gasket profile, compound, and compression prevent extrusion, tearing, and chronic rework at clamp joints. |
Key requirements for every hygienic valve and fitting:
- Assemblies should be self-draining or intentionally oriented so that product and rinse water do not stagnate.
- Product-contact surfaces should remain smooth, continuous, and free from pits, folds, sharp shoulders, and misalignment.
- Construction materials must be suitable for food contact and able to withstand product and cleaning exposure without becoming a contamination source.
Tip: The best hygienic fitting is not the one with the most certificates on the brochure. It is the one whose geometry, material, seal, and weld condition still clean reliably after repeated production and maintenance cycles.
Performance, Installation, and Maintenance
Pressure and Temperature Ratings
You must select hygienic fittings and valves against the real pressure-temperature-duty combination, not against a single headline pressure value. Published ratings vary by connection family, size, clamp type, gasket material, ferrule alignment, assembly torque, and temperature. That is why flange class tables are not a substitute for hygienic fitting data, and why one clamp series cannot be used as a universal rule for all sanitary connections.
You can use published sanitary clamp pressure tables as a practical illustration of how much ratings vary even within hygienic clamp families:
| Published Clamp Example | 70°F Pressure Rating | 250°F Pressure Rating |
|---|---|---|
| Heavy-duty sanitary clamp, 1″–1 1/2″ | 500 psi | 300 psi |
| High-pressure sanitary clamp, 1″–1 1/2″ | 1000 psi | 800 psi |
| Heavy-duty sanitary clamp, 4″ | 300 psi | 150 psi |
| High-pressure sanitary clamp, 4″ | 800 psi | 600 psi |
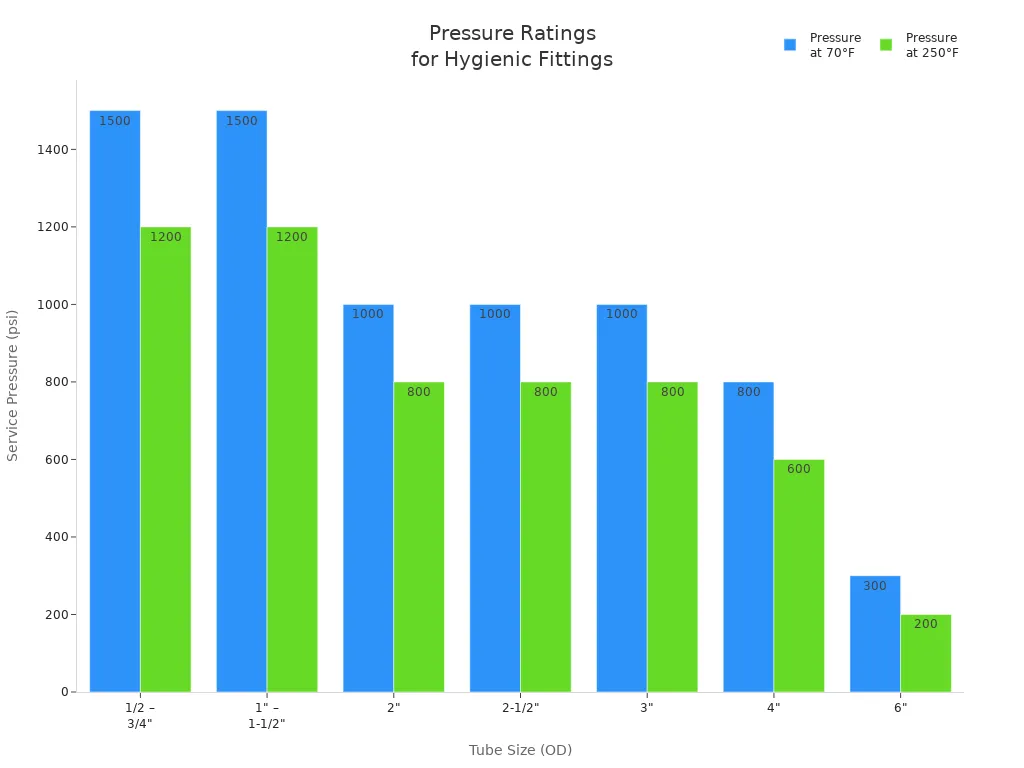
For tri-clamp valves and fittings, always check the rating of the complete assembled connection—ferrules, gasket, clamp, and temperature—not just the body material. In real troubleshooting, “the fitting failed at low pressure” often means the joint was misassembled, over-compressed, or operated outside the published assembly assumptions.
Cleaning and Maintenance Routines
You need a maintenance routine based on hygienic risk and opening frequency, not a generic mechanical calendar. Regular inspection prevents buildup, seal degradation, and unnoticed geometry problems from becoming contamination events. A practical routine looks like this:
| Frequency | Maintenance Tasks |
|---|---|
| Per changeover / per opening event | Inspect product-contact gaskets, ferrule faces, and visible seal lands; remove damaged seals; confirm that no residue, nicks, or misalignment remain before reassembly. |
| Weekly or risk-based routine | Check chronic leak points, clamp condition, threaded external hardware, valve seats, and sample points; review whether any area repeatedly retains product or rinse water. |
| Periodic deep inspection | Open representative critical assemblies, inspect for pitting, weld discoloration, roughness, compression set, mineral buildup, and evidence of incomplete drainage; verify replacement parts match the approved compound and size. |
You should choose valves and fittings that can be inspected without turning every maintenance event into a production delay. A design that is theoretically cleanable but rarely opened in practice often becomes the same kind of hidden hygienic risk as a badly designed branch.
Installation Practicality
You must install hygienic fittings correctly or you will build contamination risk into the line on day one. Common installation errors include ferrules pulled together by clamp force, branches installed without drain-friendly orientation, unsupported piping that distorts valve bodies, and internal welds that were never inspected after fabrication. Address these issues with the following best practices:
- Train installers to align ferrules before closure rather than using the clamp to force components into position.
- Design branch points, instruments, and sample valves to minimize stagnant zones and improve drainage.
- Use the specified hygienic material grade and do not mix tube/fitting standard families unless compatibility has been checked dimensionally.
- Select assemblies that can be opened, cleaned, and reassembled repeatably without damaging the product-contact seal.
- For critical circuits, verify cleanability through review of branch geometry, drainability, CIP coverage, and—where justified—challenge testing or more detailed hygienic design review.
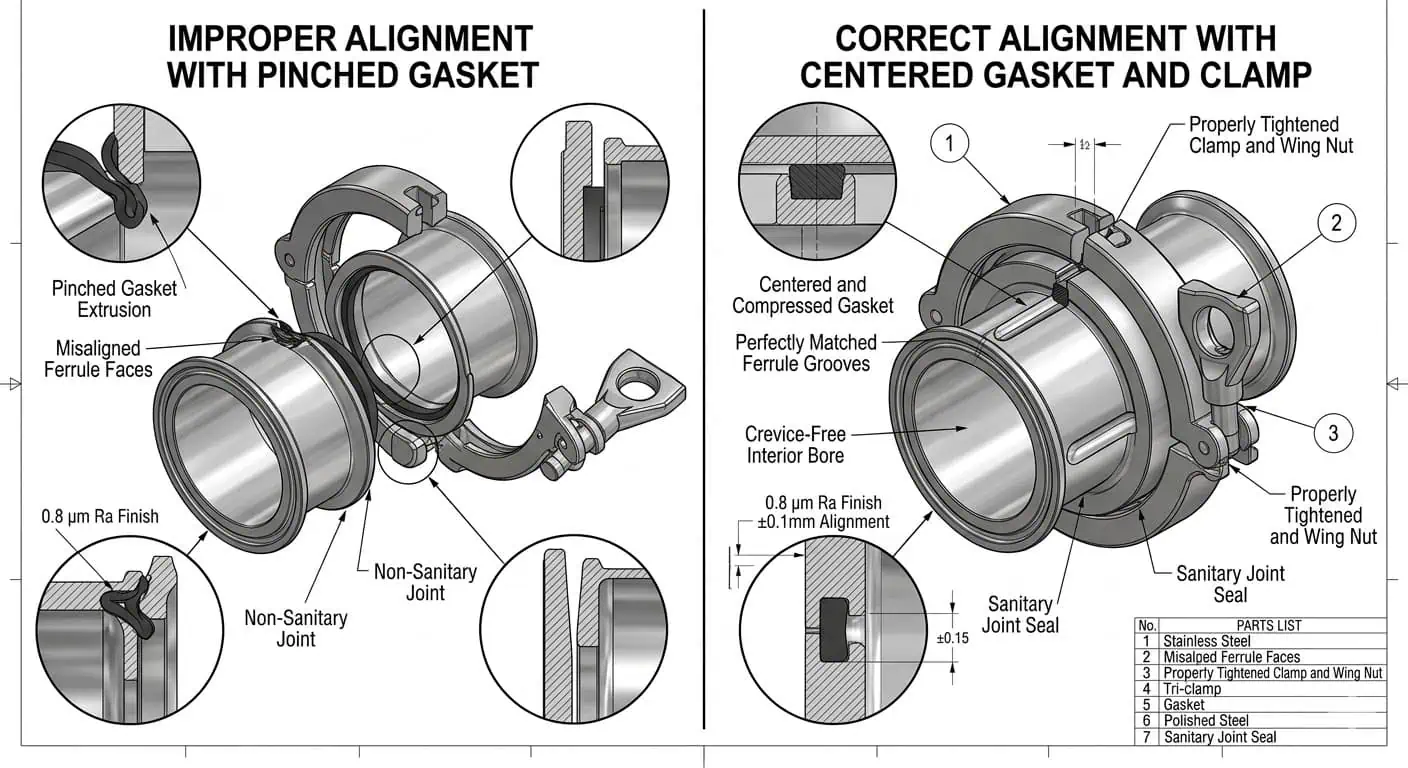
Tip: A leak at a hygienic clamp after startup is often an installation signal, not just a gasket problem. Check ferrule alignment, seal seating, clamp series, and whether the joint was closed under piping stress.
When these details are handled well, hygienic fittings support stable cleaning, faster maintenance, and fewer repeat deviations. When they are ignored, the same connection can become a permanent weak point in an otherwise well-designed line.
How to Select a Hygienic Fittings Supplier
Supplier Reputation and Technical Support
You should choose a supplier that can answer hygienic questions with drawings, tolerances, and records—not only with a sales sheet. A capable supplier understands why seal compression, internal weld finish, dimensional control, and traceability matter in food service. When you evaluate suppliers, use the following criteria:
| Criteria | Description |
|---|---|
| Technical capability | The supplier should understand hygienic standards, tolerances, weldability, seal selection, and pressure-temperature limits for the exact connection family offered. |
| Quality systems | You need controlled documentation, lot traceability, deviation handling, and a consistent method for identifying revisions and approved materials. |
| Regulatory and hygienic support | The supplier should be able to explain what is covered by standards, what is covered by declarations, and what must still be verified at the assembly or application level. |
You should still ask for references from similar hygienic projects, but make the references technical. Ask how the supplier handled weld finish, material traceability, seal compatibility, and replacement-part control in comparable food and beverage applications. That tells you much more than a generic customer list.
Supplier Certifications and Traceability
You must verify supplier documentation and traceability before approval, especially for product-contact fittings and replacement seals. Traceability is not only for the initial delivery. It must still work after maintenance, gasket changes, and part substitution. Review the following evidence types:
| Evidence Type | Description |
|---|---|
| Material certificates | Ask for EN 10204 3.1 mill test certificates or equivalent traceability evidence for critical stainless components where project scope requires it. |
| Seal and food-contact declarations | The supplier should identify the exact gasket or seat compound, not just “food grade” as a vague claim. |
| Surface and fabrication records | Where finish, weld quality, passivation, or special fabrication matters, ask what evidence is available and how it maps to the delivered component. |
| Pressure-temperature data | You need product data for the actual connection type, size, seal, and temperature range—not a borrowed chart from a different series. |
| Traceability response capability | The supplier should be able to connect the delivered fitting or seal lot back to source documents without guesswork. |
You should also confirm how replacement parts are controlled. Many hygienic deviations start when a correct original assembly is later maintained with an undocumented substitute gasket, seat, or clamp. A supplier that supports hygienic service well will understand that the spare-parts trail is part of the food-safety trail.
Practical Selection Checklist for Hygienic Fittings
Questions to Answer Before Requesting a Quote
Before you request a quote, define the process conditions that actually drive hygienic selection. Use this checklist to guide your first technical discussion:
- What exactly flows through the line, and what cleans the line?
You should define product chemistry, viscosity, solids, allergen status, and the full CIP/SIP chemical and temperature profile. - Which hygienic standard family must the fitting match?
You need to confirm whether the line is built around BS 4825, EN/DIN hygienic tubing and clamp families, 3-A expectations, or a plant-specific standard. - What are the real operating limits?
You should provide design pressure, vacuum requirement, operating temperature, cleaning temperature, and whether shock, pulsation, or vibration is present. - How often will the connection be opened?
The answer affects whether quick-disconnect convenience or a more permanent welded approach is better for your hygiene and maintenance model. - What document package is required with delivery?
You need to specify material traceability, seal declarations, finish information, rating data, and any special quality documentation before ordering.
Tip: The clearest RFQs for hygienic fittings describe media, cleaning chemistry, operating limits, connection standard, and required documents in the first page—not after the supplier has already quoted the wrong series.
Questions to Ask Before Final Approval
Before you approve any hygienic fitting for production use, confirm that the installed assembly matches the intended hygienic duty. Ask these questions:
- Do the body material, seal compound, and connection standard match the approved specification?
You must confirm what is actually installed, not what was originally quoted. - Are the ferrules, branches, and welds aligned and drainable in the as-built condition?
You need to inspect the real installation because good components can still create a bad hygienic geometry. - Has the supplier provided the correct pressure-temperature information for this exact assembly?
You should confirm the rating by size, series, gasket, and temperature—not by catalog family alone. - Can the plant trace this fitting and its replacement seals back to supporting documents?
You need records that still work after commissioning and future maintenance. - Has the line been reviewed for likely hygienic weak points such as dead spaces, poor drainage, and repeat-open joints?
You want to identify cleaning failures before the first deviation does it for you.
Note: Final approval should close both the technical loop and the traceability loop. If you cannot prove what the fitting is, what seal is in it, and how it should be cleaned, approval is incomplete.
You should always start with the actual process, then review hygienic risk, compliance scope, material suitability, cleanability, pressure-temperature limits, and installation practicality. Selecting hygienic fittings is not about buying a polished part with a familiar label. It is about matching geometry, material, seal, and documentation to the real line. Before you request quotes or approve fittings, review the common mistakes below:
| Mistake | Impact |
|---|---|
| Assuming all “sanitary” fittings of the same size are interchangeable | Dimensional mismatch, seal problems, and spare-parts confusion |
| Ignoring gasket compound compatibility | Swelling, leakage, odor retention, and short seal life |
| Installing branches or valves without checking drainage | Residue retention, inconsistent CIP, and higher microbial risk |
| Using catalog pressure data from the wrong connection series | Unsafe service assumptions and avoidable startup failures |
| Treating documentation as optional | Weak audit defense and poor traceability after maintenance |
Work with suppliers that can support both the hardware and the evidence chain. In hygienic systems, a fitting is only as reliable as the geometry, seal, and records behind it.
FAQ
What is the most important factor when selecting hygienic fittings?
You must match the fitting to the real product, cleaning chemistry, and line geometry.
Nominal size alone is not enough. Review product sensitivity, CIP/SIP exposure, seal compound, drainability, and the applicable hygienic standard before approval.
How do I ensure fittings are easy to clean?
Choose fittings with smooth internal transitions, minimal dead spaces, and reliable drainage.
Also inspect weld quality, ferrule alignment, and seal condition. A smooth catalog finish does not help much if the installed joint creates a pocket or internal step.
Why is documentation important for hygienic fittings?
Documentation proves what was actually installed and helps preserve traceability after maintenance.
You need material certificates where required, seal declarations, rating data, and records that connect installed parts to approved specifications. Without that trail, audits and deviation investigations become much harder.
Can I use the same fittings for all food and beverage applications?
No, you must select fittings according to the specific process, hygiene risk, and cleaning regime.
Different products and cleaning agents change corrosion risk, seal life, and cleanability. A fitting that works well on one beverage line may be the wrong choice for dairy, sauce, allergen changeover, or higher-temperature cleaning duty.
Is 304L enough, or should I use 316L for hygienic fittings?
304L is often acceptable for moderate hygienic service, but 316L is usually the safer choice when chlorides, aggressive cleaning, or a higher corrosion margin are involved.
The correct choice depends on the actual product, CIP chemistry, temperature, crevice exposure, and customer specification. Grade selection should be confirmed with the real service envelope, not copied from another line.




