Die Auswahl von Hygienefittings für Fermentationssysteme sollte auf Reinigungsfähigkeit, Entleerbarkeit, Dichtungskontrolle, Sterilisationskompatibilität und lokalen Montagerisiken basieren – nicht nur auf der Anschlussart. Im Fermentationsbetrieb ist ein Fitting nicht nur ein Verbinder zwischen Rohrabschnitten. Es wird Teil der aseptischen Grenze, Teil des CIP/SIP-Pfads und Teil der lokalen Geometrie, die bestimmt, ob der schwierigste Punkt im System tatsächlich gereinigt, sterilisiert, inspiziert und gewartet werden kann, ohne unnötige Kontaminationsrisiken zu schaffen.
Deshalb sind Fermentationssysteme weniger nachsichtig als allgemeine Hygienerohrleitungen. Inokulation, sterile Übertragung, Gaszugabe, Schaumkontrolle, Probenentnahme, Ernteleitungen und wiederholte Reinigungs- oder Dampfzyklen belasten lokale Montagen stärker, als viele Teams erwarten. Ein Fitting, das im Katalog hygienisch aussieht, kann dennoch einen Totraum, eine schlecht entleerbare Tasche, eine wiederholte Dichtungsbelastung oder einen schwer zu validierenden Abzweig verursachen, wenn es am falschen Ort installiert oder für die falsche Lebenszykluslogik ausgewählt wird.
Dieser Leitfaden erklärt, wie Ingenieure Hygienefittings für Fermentationssysteme auswählen sollten, wo Klemmenfittings sinnvoll sind, wo geschweißte Verbindungen in der Regel sicherer sind, wie die Abzweiggeometrie die hygienische Leistung verändert und warum die Fittingauswahl immer an Material-, Oberflächen-, Dichtungs- und Dokumentationsanforderungen gebunden sein sollte, anstatt als isolierte Kaufentscheidung behandelt zu werden.
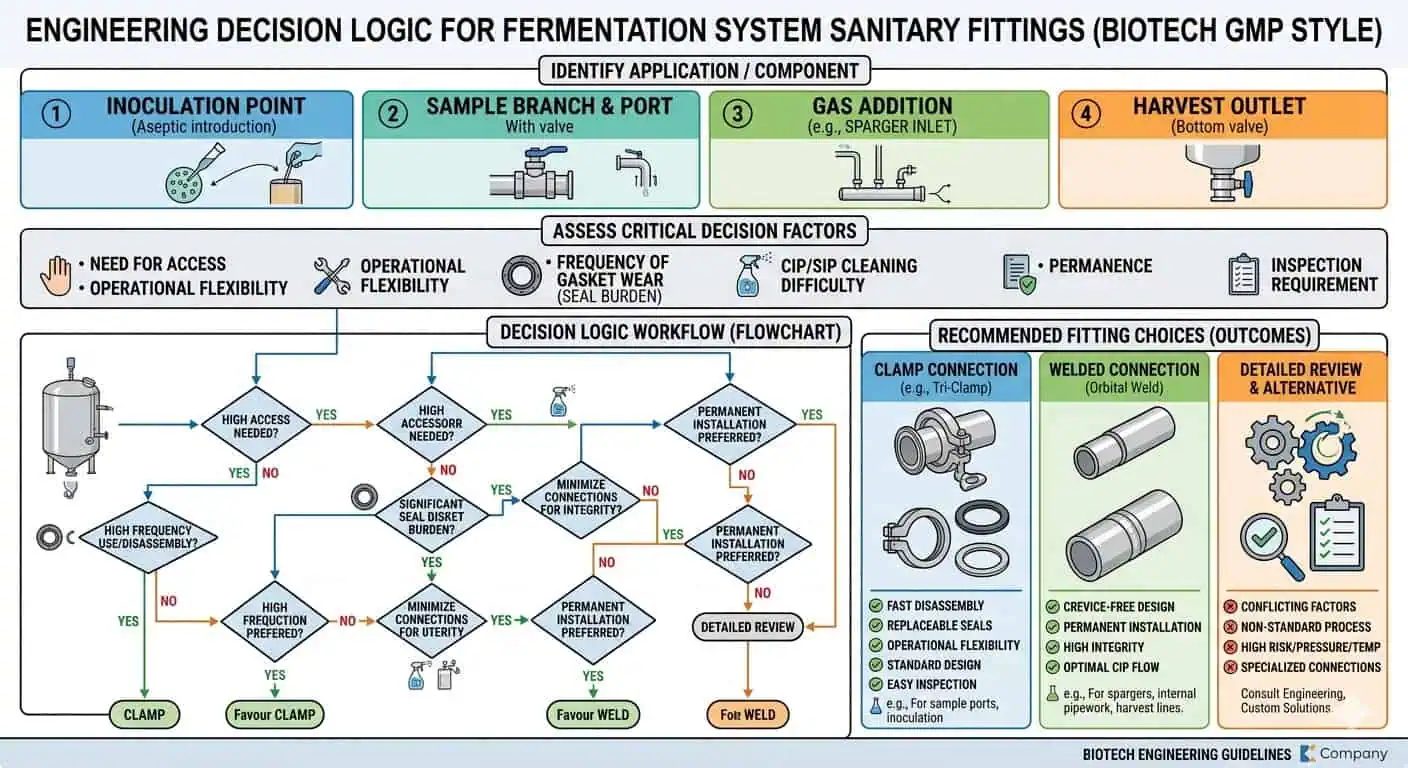
Warum die Fittingauswahl in Fermentationssystemen wichtiger ist
Fermentationssysteme sind empfindlich gegenüber lokalen Montagen
Das erste Hygienefehler in einem Fermentationssystem tritt normalerweise an einer lokalen Montage auf, nicht in der Mitte eines geraden Laufs. Probenahmepunkte, Gaszugabeanschlüsse, Sensort-Stücke, Ernteabzweige, Membranventilblöcke, Bodenauslässe und lokale Übergangsschnittstellen kombinieren mehr Variablen als ein einfacher Rohrabschnitt. Sie vereinen Abzweiggeometrie, Dichtungsschnittstellen, Bedienerinteraktion, Schweißdichte und lokales Entleerungsverhalten. Diese Kombination ist der Grund, warum die Fittingauswahl in Fermentationssystemen viel ortspezifischer sein muss als im allgemeinen Hygienebetrieb.
In der praktischen Ingenieurarbeit ist die sicherste Regel, die Armatur für die schwierigste lokale Montage zu wählen, nicht für die einfachste gerade Strecke. Eine Verbindungsart, die auf einem einfachen Transferabschnitt effizient erscheint, kann am Probenahmepunkt, Gaszuführungsabzweig, Ventilblock oder Behälterstutzen, wo die hygienischen Konsequenzen höher und der Prozess weniger nachsichtig ist, die falsche Wahl sein.
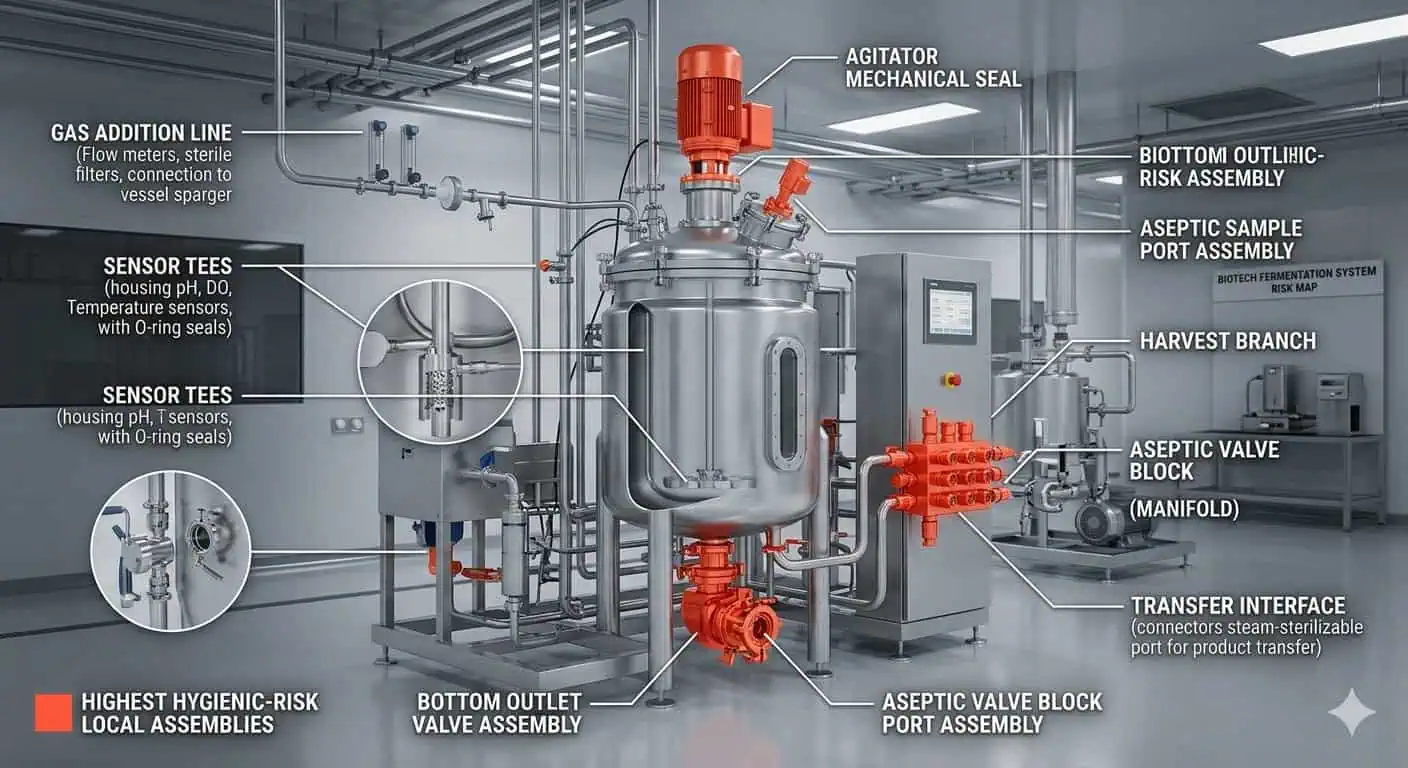
Steriler Transfer und Dichtungsintegrität erhöhen die Anforderungen
In Fermentationssystemen sitzen Armaturen oft direkt innerhalb der aseptischen Kontrollgrenze. Der FDA Biotechnology Inspection Guide besagt, dass Bioreaktor-Impfung, Transfer und Erntevorgänge mit validierten aseptischen Techniken durchgeführt werden müssen und dass Zugaben oder Entnahmen aus industriellen Bioreaktoren im Allgemeinen über dampfsterilisierte Leitungen und Dampfsperrenbaugruppen erfolgen. Derselbe Leitfaden stellt auch fest, dass Faktoren, die das Endprodukt beeinflussen, die Wirksamkeit von Dichtungen und Dichtungen einschließen. Das ist eine starke Erinnerung, dass die Armaturenauswahl nicht nur um Metallgeometrie geht. Es geht auch darum, ob die lokale Dichtungsgrenzfläche die Sterilitätslast des tatsächlichen Prozesses unterstützen kann. FDA Biotechnology Inspection Guide
Typische Ingenieurrealität: Ein Dichtungsproblem an einem Abzweig oder Probenahmegrenzfläche zeigt sich oft nicht zuerst als dramatisches Leck. Es zeigt sich als ein Ort, der wiederholte Eingriffe, zusätzliche Überprüfung nach SIP oder mehr Vorsicht während des aseptischen Gebrauchs als der Rest des Systems erfordert.
Schlechte hygienische Konstruktion schafft Reinigungs- und Validierungsaufwand
Eine Armatur, die eine schlechte lokale Geometrie erzeugt, erhöht normalerweise sowohl den Wartungsaufwand als auch den Validierungsaufwand später. EHEDG-Hygienekonstruktionsprinzipien stellen klar, dass Einheiten mit schlechter hygienischer Gestaltung schwer zu reinigen sind. In Fermentationssystemen bedeutet dies normalerweise, dass das Problem nicht “die gesamte Leitung” ist. Es ist ein lokaler Abzweig, eine Probenentnahmestelle, ein Reduzierstück oder ein Ventilbaugruppe, die Flüssigkeit nicht austauscht oder abläuft wie der Rest des Kreislaufs. EHEDG-Hygienekonstruktionsprinzipien
Typischer Branchenfall: Die Hauptfermentationsübertragungsleitung sah bei der allgemeinen Überprüfung wiederholt akzeptabel aus, aber ein Probenblock erforderte ständig zusätzliche Aufmerksamkeit, weil die lokale Baugruppe nach SIP-Abkühlung Kondensat zurückhielt. Das Problem war nicht “schlechter Edelstahl”. Es war eine Fitting- und Abzweiganordnung, die die lokale Baugruppe schwieriger zu spülen und zu überprüfen machte als den Rest des Systems.
Was Ingenieure tatsächlich bei der Auswahl von Fermentationsfittings bewerten
Reinigungseigenschaften
Die erste Frage ist, ob das Fitting die Reinigungsfähigkeit am eingebauten Standort fördert oder beeinträchtigt. Innere Übergänge, Körpergeometrie, angrenzendes Schweißverhalten, Dichtungsposition und Abzweigform beeinflussen alle, ob ein Fitting zuverlässig vor Ort gereinigt werden kann. In Fermentationssystemen wird das Fitting anhand der Leistung der schwierigsten lokalen Oberfläche beurteilt, nicht daran, wie poliert die Hardware von außen aussieht.
Deshalb ist ASME BPE bei der Fittingauswahl nützlich. Es behandelt Materialien, Konstruktion, Fertigung, Inspektionen, Prüfungen und Zertifizierung nicht als isolierte Themen. Im Bioprozesseinsatz ist ein Fitting nur wirklich akzeptabel, wenn die installierte Baugruppe das beabsichtigte hygienische Verhalten über Fertigung, Betrieb, Reinigung und Inspektion hinweg unterstützt – nicht nur, wenn die Komponente in einer Teileliste korrekt aussieht. ASME BPE offizielle Übersicht
Entleerbarkeit
Ein Fitting, das nicht gut abläuft, wird normalerweise ein wiederkehrendes hygienisches Problem, selbst wenn der Rest des Kreislaufs akzeptabel erscheint. Fermentationssysteme erleben Brüheübertragung, Reinigungslösungsexposition, Dampfsterilisation, Kondensatbildung und Stillstandszeiten. Wenn ein Abzweig, Ventilauslass oder Reduzierstück nach diesen Schritten Flüssigkeit zurückhält, kann die Belastung dieser lokalen Retention das hygienische Risiko einer viel größeren Leitung dominieren.
Expertenrat: Beurteilen Sie die Ablaufbarkeit niemals allein anhand der Zeichnungsabsicht. Überprüfen Sie die endgültige Ausrichtung, Stützeffekt, Tiefpunktverhalten und Nach-SIP-/Nach-CIP-Rückfluss wie installiert.
Dichtungskontrolle
Viele Risiken bei Armaturen in Fermentationssystemen sind tatsächlich Dichtungsrisiken. Eine Klemmenverbindung kann mechanisch einwandfrei und einfach zu öffnen sein, führt aber auch eine kontrollierte Dichtungsschnittstelle ein, die Dampf, chemische Reinigung, wiederholte Kompression und manchmal wiederholte Handhabung durch Bediener tolerieren muss. In einer Fermentationsumgebung kann diese Schnittstelle nicht wie eine gewöhnliche verbrauchbare Grenze behandelt werden.
Typisches Feldmuster: die Metallhardware bleibt akzeptabel, aber der Standort wird wartungsempfindlich, weil die Dichtung mehr Dampf, mehr Chemikalien oder mehr Montagevariationen überstehen muss, als die ursprüngliche Lebenszyklusannahme zuließ.
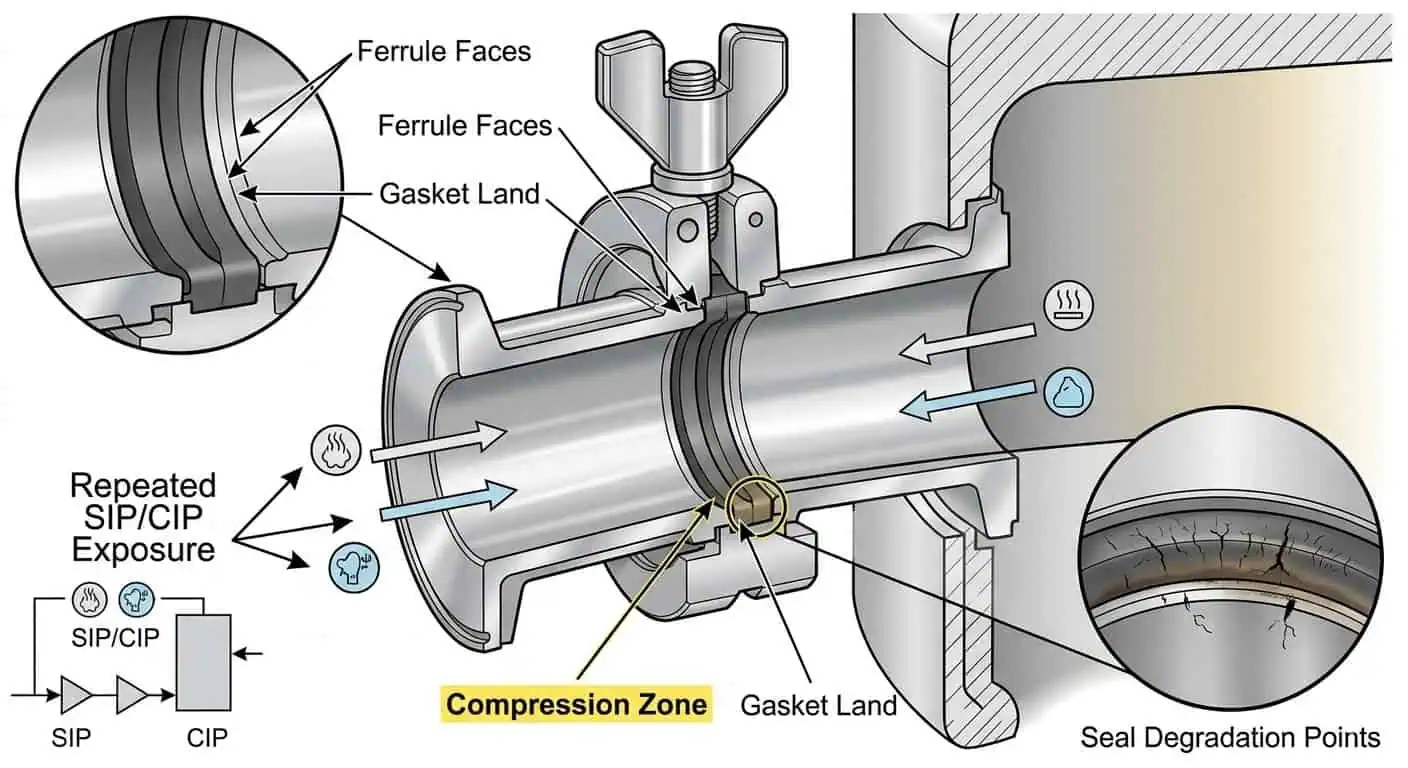
CIP- und SIP-Kompatibilität
Die Armaturenauswahl muss der tatsächlichen thermischen und chemischen Belastung des Systems entsprechen. Der FDA Biotechnology Inspection Guide besagt, dass Medien üblicherweise durch SIP oder ein kontinuierliches Sterilisationssystem sterilisiert werden, dass alle nach der Sterilisation hinzugefügten Nährstoffe oder Chemikalien steril sein müssen und dass Luftleitungen sterile Filter enthalten müssen. In technischer Hinsicht bedeutet dies, dass die Armatur nicht nur unter Prozessübertragungsbedingungen bewertet werden kann. Sie muss auch die Sterilisations- und Reinigungslogik überstehen, die den wahren Lebenszyklus des Fermentationssystems definiert. FDA Biotechnology Inspection Guide
Diese gleiche Lebenszykluslogik passt auch zu FDA 21 CFR 211.67, das vorschreibt, dass Geräte nach schriftlichen Verfahren gereinigt, gewartet, hygienisiert oder sterilisiert werden müssen, wo dies angemessen ist. FDA 21 CFR 211.67
Inspektions- und Wartungszugang
Die richtige Armatur hängt teilweise davon ab, ob der Standort mehr von Zugänglichkeit oder von einer geringeren Schnittstellenanzahl profitiert. Einige Baugruppen benötigen tatsächlich kontrolliertes Öffnen, Dichtungswechsel, lokale Wartung von Sensoren oder Wartung von Probenahmestellen. Andere sind sicherer, wenn die Anzahl der Demontageschnittstellen minimiert wird. Dies ist die eigentliche technische Frage hinter der Entscheidung zwischen Klemmen und Schweißen im Fermentationsbetrieb.
Die Hauptanschlusstypen, die in Fermentationssystemen verwendet werden
Klemmtyp-Hygieneverbindungen
Klemmartige hygienische Anschlüsse sind dort nützlich, wo kontrollierter Zugang, Dichtungswechsel, Modularität oder Instrumentenwartung echte Lebenszyklusanforderungen sind. Ihr Wert liegt nicht nur in der Installationsgeschwindigkeit. Es ist, dass sie lokalen Zugang ermöglichen, ohne die Leitung zu schneiden und neu zu schweißen. In Fermentationssystemen kann das an Instrumentenpunkten, einigen Probenahmebaugruppen und bestimmten modularen Verbindungen, bei denen Wartungszugang Teil des tatsächlichen Betriebsplans ist, sehr nützlich sein.
Aber Klemmschnittstellen schaffen auch eine kontrollierte Belastung. Jede Klemmbaugruppe fügt eine Dichtungsschnittstelle, eine Ausrichtungsanforderung und eine Frage zur Wiederzusammenbauqualität hinzu. In Bereichen mit geringen Konsequenzen mag das akzeptabel sein. In hochkonsequenten aseptischen oder schwer zu entleerenden Zonen kann derselbe Komfort zu einem wiederholten hygienischen Risiko werden.
Geschweißte hygienische Verbindungen
Geschweißte hygienische Verbindungen werden oft bevorzugt, wo der Prozess wiederholte Demontageschnittstellen minimieren und das Risiko durch Dichtungsanzahl reduzieren sollte. In festen produktberührenden Fermentationsleitungen entfernt eine gute geschweißte Verbindung eine Ebene der Baugruppenvariabilität, die eine Klemmverbindung immer beibehält. Das macht Schweißen nicht automatisch an jedem Ort besser. Es bedeutet, dass der Kompromiss normalerweise zwischen Zugang und Kontrolle liegt, nicht zwischen “hygienisch” und “unhygienisch”.”
Expertenrat: Verwenden Sie die dauerhaftere geschweißte Option, wenn der Ort hochkonsequent, relativ stabil ist und nicht genug von routinemäßigem Öffnen profitiert, um die zusätzliche Dichtungsschnittstelle zu rechtfertigen.
Membranventil- und Probenahmebaugruppen-Schnittstellen
Einige der risikoreichsten Armaturenentscheidungen in Fermentationssystemen betreffen Membranventile, Probenahmevorrichtungen und verzweigungsreiche lokale Verteiler. Diese Bereiche vereinen Dichtungsanzahl, Bedienerinteraktion, Empfindlichkeit der Verzweigungsgeometrie und starke hygienische Konsequenzen. In der Praxis führt die falsche Armaturenwahl hier oft nicht zu dramatischen Ausfällen. Sie scheitert, indem sie zur lokalen Baugruppe wird, die wiederholt Reinigungs-, Sterilitäts- oder Wartungsfragen aufwirft.
Instrumenten- und Gasleitungsanschlüsse
Kleine Armaturen können unverhältnismäßig hohes hygienisches Risiko in Fermentationssystemen bergen. pH-Sonden, gelöste Sauerstoffpunkte, Druck- und Temperaturschnittstellen, Luft- oder Sauerstoffzugaben sowie Entschäumer- oder Nährstoffdosierpunkte hängen alle vom lokalen hygienischen Verhalten ab, das leicht unterschätzt wird. Ein kleines Instrumenten-T-Stück oder ein Gasleitungsanschluss in falscher Ausrichtung kann ein größeres reales hygienisches Risiko darstellen als ein viel längerer gerader Abschnitt gut gestalteter Transferleitung.
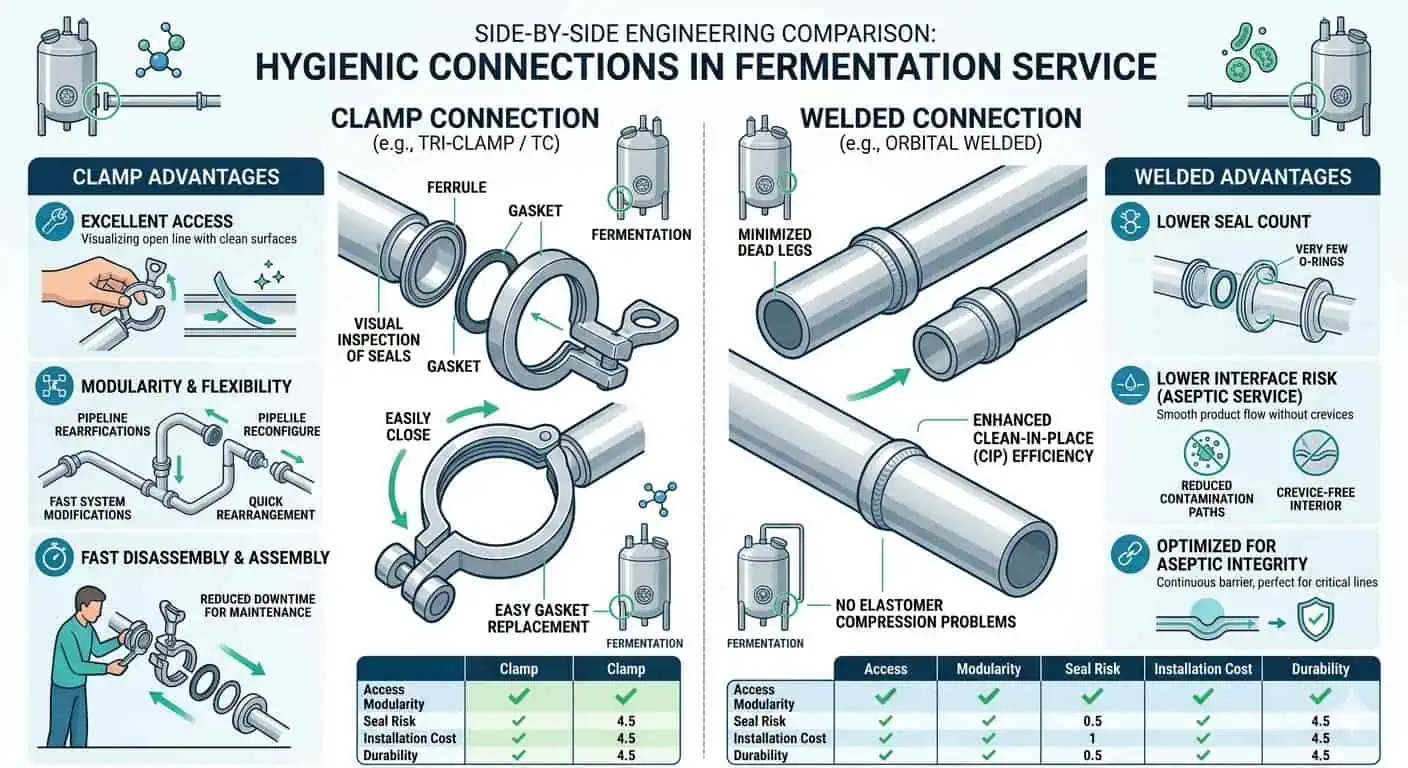
Wie Armaturen nach Fermentationsprozessbereich auswählen
Inokulations- und aseptische Transferpunkte
Inokulations- und aseptische Transferpunkte sollten primär nach Sterilitätserhalt und sekundär nach Bequemlichkeit ausgewählt werden. Der FDA-Biotechnologieleitfaden betont validierte aseptische Techniken und dampfsterilisierte Transferlogik an diesen Standorten. Das bedeutet, unnötige Schnittstellen, schwer zu sterilisierende Verzweigungen oder schwach kontrollierte Dichtungen erzeugen hier eine unverhältnismäßige Belastung. In vielen Projekten verlagert dies die Entscheidung hin zu einfacheren, besser kontrollierten lokalen Baugruppen mit den geringsten vermeidbaren hygienischen Unsicherheiten.
Fermenterbehälter-Stutzen und Deckelanschlüsse
Deckel- und Behälterstutzen-Fittings müssen anhand der gesamten umgebenden Baugruppe beurteilt werden. Gaszugabe, Sprühvorrichtungen, Druckregelung, Sensoreinführung, Probenahme und entlastungsbezogene Funktionen häufen sich oft am Behälterdeckel. Dies sind keine guten Orte für eine generische Fitting-Entscheidung. Sie erfordern eine lokale Überprüfung von Neigung, Kondensatverhalten, Sterilitätspfad und Wartungszugang gemeinsam.
Typische Ingenieurrealität: Deckelanschlüsse, die während der Layout-Überprüfung akzeptabel erscheinen, können nach SIP viel schwerer zu verteidigen werden, wenn Kondensatverhalten, Dampferreichbarkeit oder Zugang zur Verifizierung nie als installiertes Baugruppenproblem geprüft wurden.
Bodenauslass, Ernte- und Transferleitungen
Bei Bodenauslässen und Ernteleitungen wird die Entleerbarkeit zu einem der entscheidendsten Fitting-Kriterien. Eine Verbindung, die Brühe, Kondensat oder Reinigungslösung nach der Verwendung zurückhält, kann zum eigentlichen hygienischen Schwachpunkt werden, selbst wenn der größere Transferkreislauf gut gestaltet ist. In diesen Bereichen sollte die Fitting-Auswahl gemeinsam mit Ausrichtung, Reduziererstrategie und Nachnutzungs-Rücklaufverhalten überprüft werden.
Probenahmepunkte und Seitenabzweigungen
Probenahmepunkte und Seitenabzweigungen sind Orte, an denen Fitting-Auswahlfehler normalerweise am schnellsten sichtbar werden. Sie kombinieren lokale Strömungsschwäche, höhere Bedienerinteraktion, Dichtungsbelastung und starke Kontaminationsfolgen.
Ein häufiger Fall in Fermentationssystemen ist eine Schleife, die im Hauptlauf akzeptabel funktioniert, während eine lokale Probe- oder Abzweigbaugruppe ständig zusätzliche Überprüfungen erfordert. Der Grund ist in der Regel nicht mysteriös. Der Abzweig weist ein anderes Austauschverhalten, ein anderes Kondensatverhalten und eine andere lokale Dichtungsbelastung als die Hauptleitung auf. Deshalb sollte die Auswahl der Fittings auf dem schwierigsten lokalen Standort basieren, nicht auf dem einfachsten zu entwerfenden Abschnitt.
Typischer Branchenfall: Ein Fermentationssystem zeigte akzeptables Verhalten in der Hauptschleife, aber derselbe Probenabzweig benötigte wiederholt zusätzliche hygienische Aufmerksamkeit. Die Ursache war nicht das Hauptrohrmaterial oder das CIP-Rezept. Es war die lokale Abzweiglänge, die Fitting-Geometrie und das Rücklaufverhalten um die Probenbaugruppe herum.
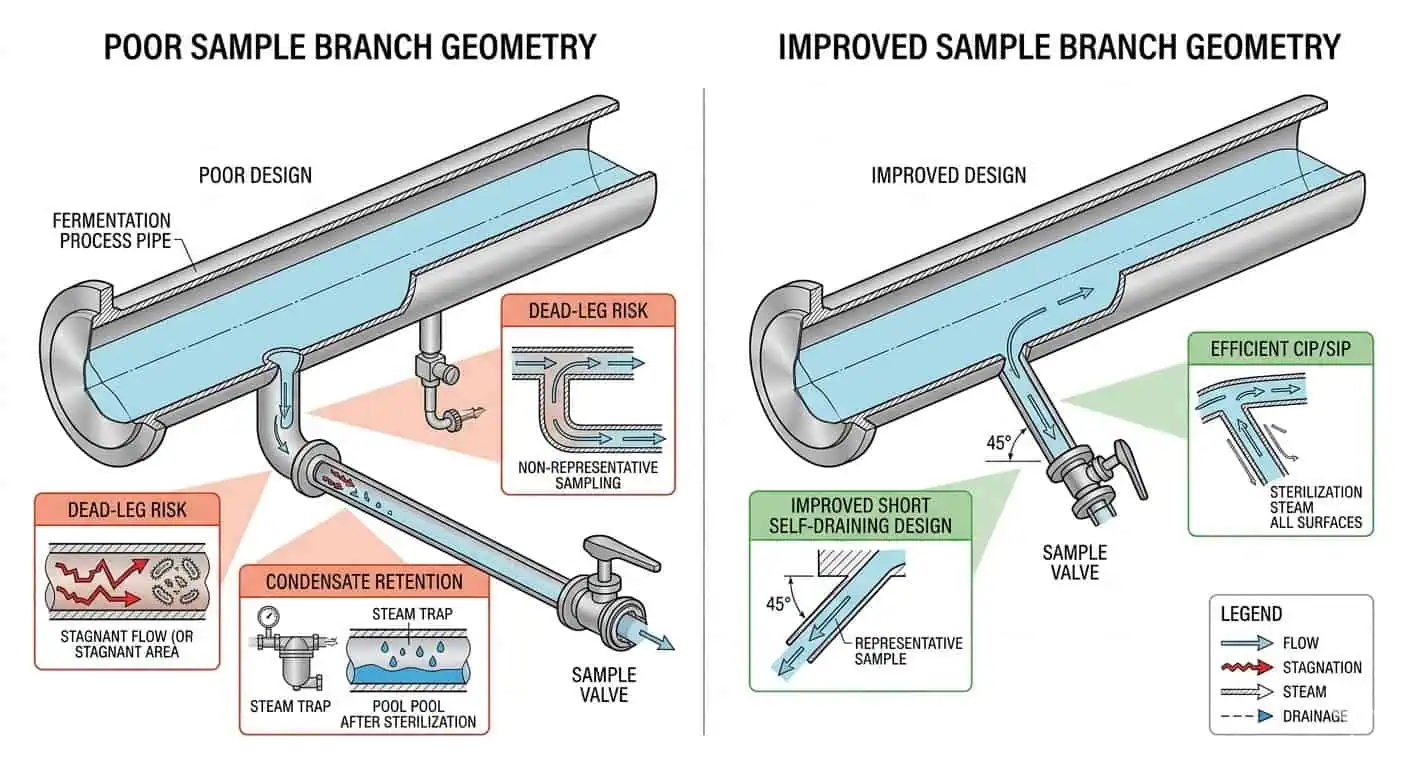
CIP/SIP-Rücklauf- und Reinigungskreisläufe
Fittings in CIP/SIP-Rücklaufbereichen müssen als Teil des Reinigungskreislaufs bewertet werden, nicht nur als Produkttransporthardware. Ihre Geometrie beeinflusst, wie effektiv die lokale Baugruppe ausgetauscht wird, wie leicht Kondensat abläuft und ob die Verifizierung weiterhin unkompliziert bleibt. Hier verbindet sich die Fitting-Auswahl direkt mit Hochreinheits-Rohrleitungsdesign für Biotechnologieanlagen und Hygienische Rohrleitungswartung und Validierungsüberlegungen.
Wo die Fitting-Auswahl normalerweise schiefgeht
Tote Zonen versteckt in “korrekten” Baugruppen
Einer der häufigsten Fitting-Fehler ist nicht eine falsche Katalogfamilie. Es ist eine lokale Baugruppe, die nach der Installation totes Verhalten erzeugt. Ein Fitting kann dimensionell korrekt und materialmäßig korrekt sein und dennoch eine schlechte Austauschzone erzeugen, aufgrund von Abzweiglänge, Abzweigrichtung, Körpergeometrie oder der Art, wie es in die Schleife integriert wurde.
Zu viele Klemmenverbindungen in Hochrisikobereichen
Klemmenverbindungen werden oft übermäßig eingesetzt, weil sie bei der Installation und Inbetriebnahme praktisch sind. Das Problem ist nicht, dass Klemmenverbindungen grundsätzlich falsch sind. Das Problem ist, dass sie in Hochrisikobereichen eine Dichtungsbelastung und Montagebelastung verursachen können, die der Lebenszyklus des Fermentationssystems eigentlich nie benötigt hat.
Gutes Material, falsche Montage
Ein 316L-Fitting kann für die installierte Baugruppe trotzdem falsch sein. Materialkorrektheit übertrumpft nicht schlechte lokale Geometrie, schwache Rücklaufentwässerung, zu viele Schnittstellen oder einen Fitting-Stil, der Reinigung und Verifizierung unnötig erschwert. In Fermentationssystemen ist das Montageverhalten wichtiger als die Katalogbeschreibung.
Beispielbaugruppen, die leicht erreichbar, aber schwer zu reinigen sind
Dies ist eines der realistischsten Feldprobleme in der Fermentationsarbeit. Ein Beispiel-Fitting kann zugänglich und praktisch sein, aber wenn es einen schlechten Austauschzweig oder eine schwierige Dichtungsschnittstelle erzeugt, kann es zur Baugruppe werden, die während des Betriebs, der Wartung oder der Validierungsprüfung wiederholt hygienische Fragen aufwirft.
Materialien, Oberfläche und Dichtungsstrategie
Warum 316L verbreitet ist, aber keine vollständige Lösung darstellt
316L ist eine gängige Basis in Bioprozess-Fermentationssystemen, da sie eine praktische Kombination aus Korrosionsbeständigkeit, Schweißbarkeit und hygienischer Akzeptanz bietet. Aber die Auswahl der Fittings endet nicht mit der Grundlegierung. Dasselbe 316L kann je nach Oberflächenbeschaffenheit, Schweißintegration, Dichtungswahl und lokaler Geometrie unterschiedliche hygienische Ergebnisse erzielen. Für die umfassendere Materialentscheidung verknüpfen Sie diese Seite mit 316L Edelstahl in Bioprozessanwendungen.
Oberflächenbeschaffenheit und Schweißintegration sind wichtiger, als Käufer erwarten
Fittings befinden sich oft genau dort, wo der Oberflächenzustand entscheidender wird. Abzweigungen, Ferrule, kleine lokale Baugruppen und schweißnahe Bereiche sind die Stellen, an denen die hygienische Leistung am empfindlichsten auf Oberflächenbeschaffenheit und Fertigungsqualität reagiert. Eine korrekte Fitting-Familie mit schwacher lokaler Schweißausführung ist immer noch eine schwache Fermentationssystem-Baugruppe.
Dichtungsmaterial muss der Fermentationsrealität entsprechen
Die Dichtungsfamilie muss dem tatsächlichen Prozess und der Lebenszyklusbelastung entsprechen. Dampf, Laugen, Säuren, Desinfektionsmittel, wiederholte Kompression, wiederholtes Öffnen und lokale Temperaturzyklen beeinflussen alle, ob eine Dichtung oder Membran zuverlässig bleibt. In Fermentationssystemen ist eine Fitting-Entscheidung erst abgeschlossen, wenn auch die Dichtungsstrategie definiert ist.
Klemm- vs. geschweißt: Die eigentliche Entscheidung für Fermentationssysteme
Wenn die Klemme die bessere technische Wahl ist
Klemm-Hygienikverschraubungen sind die bessere Wahl, wenn ein kontrollierter modularer Zugang tatsächlich erforderlich ist. Dies umfasst Standorte, an denen Inspektion, routinemäßiger lokaler Service oder Dichtungsersatz Teil des Lebenszyklus sind und wo die lokale Geometrie auch mit der zusätzlichen Dichtungsschnittstelle hygienisch vertretbar bleibt.
Wenn die Schweißverbindung die bessere technische Wahl ist
Geschweißte Verbindungen sind in der Regel die bessere Wahl, wenn die hygienischen Auswirkungen zusätzlicher Schnittstellen hoch sind und die Baugruppe relativ stabil ist. In diesen Bereichen ist die Reduzierung der Dichtungsanzahl und der Variabilität beim Wiederzusammenbau meist wichtiger als einfaches Öffnen.
Eine bessere Frage als “Welches ist hygienischer?”
Die bessere technische Frage lautet: welche Option reduziert das dominierende hygienische Risiko in dieser spezifischen lokalen Baugruppe besser? Diese Frage zwingt das Designteam, Reinigbarkeit, Entwässerbarkeit, Sterilität, Dichtungsbelastung und Wartungsrealität gemeinsam zu betrachten, anstatt Klemme und Schweißung als abstrakte Produktkategorien zu behandeln.
Praktische Auswahlmethode
Schritt 1: Definieren Sie die Prozessbelastung
Beginnen Sie mit dem tatsächlichen Fluid, dem Reinigungsregime, der Sterilisationsbelastung und den hygienischen Folgen eines Ausfalls. Eine Armatur an einem SIP-belasteten Probenahmepunkt ist nicht die gleiche Entscheidung wie eine Armatur an einer Hilfsverbindung mit geringerem Risiko.
Schritt 2: Identifizieren Sie die anspruchsvollsten lokalen Baugruppen
Identifizieren Sie dann, wo sich die tatsächliche hygienische Belastung konzentriert. In Fermentationssystemen ist dies normalerweise nicht die gerade Transferleitung. Es ist die Abzweigung, der Probenahmepunkt, die Gas-Schnittstelle, das Ventilblock oder der Behälterstutzen.
Schritt 3: Entscheiden Sie, ob der Standort Zugang oder eine geringere Schnittstellenanzahl benötigt
Dies ist die eigentliche Entscheidung zwischen Klemme und Schweißung. Wenn der Standort stark von einer routinemäßigen kontrollierten Öffnung profitiert, kann eine Klemme gerechtfertigt sein. Wenn er mehr von einer reduzierten Dichtungsanzahl und reduzierter Montagevariabilität profitiert, ist geschweißt normalerweise die bessere Antwort.
Schritt 4: Definieren Sie Material, Oberfläche, Dichtung und Dokumentation gemeinsam
Finalisieren Sie die Armatur nicht, bis der benetzte Zustand vollständig definiert ist. Material, Oberflächenbeschaffenheit, Dichtungs- oder Membrananforderung und Dokumentationserwartungen sollten gemeinsam festgelegt werden, nicht später als separate Projektklärungen hinzugefügt.
Schritt 5: Prüfen, ob die Armatur weiterhin die Validierungslogik unterstützt
Die letzte Frage ist einfach: Reduziert diese Armatur das dominierende hygienische Risiko in dieser exakten Montage, oder sieht sie nur passend aus, weil sie anderswo im System üblich ist?
Häufige Fehler
Auswahl nach Katalogoptik
Eine polierte oder hygienisch aussehende Armatur garantiert kein gärungsbereites hygienisches Ergebnis.
Überall Klemmen wählen, weil es bequem ist
Bequemlichkeit während der Installation ist nicht dasselbe wie langfristige hygienische Eignung.
Dichtungsbelastung ignorieren
Viele armaturbezogene Ausfälle sind tatsächlich Dichtungsgrenzflächenausfälle unter Dampf-, Chemikalien- und Wiederaufbaustress.
Verzweigungsgeometrie ignorieren
Eine korrekte Armaturfamilie kann immer noch Totstrecken- oder schlechte Entwässerungsverhalten erzeugen, wenn die lokale Verzweigungsanordnung falsch ist.
Material diskutieren, aber Montageverhalten vergessen
Materialkorrektheit gleicht keine schwache lokale hygienische Montage aus.
Dokumentation als sekundär behandeln
In regulierten Systemen kann ein technisch akzeptables Fitting ohne saubere Nachweiskette dennoch Freigabe- oder Lebenszyklusrisiken verursachen.
Praktische Einkaufs- und Design-Checkliste
Verwendung von Klemmen-Typ-Hygienefittings, wenn
- Kontrollierter modularer Zugang ist tatsächlich erforderlich.
- Inspektion und Dichtungsersatz sind Teil des normalen Lebenszyklus.
- Die lokale Geometrie bleibt reinigbar und entleerbar.
- Die Dichtungskontrolle ist stark genug, um die zusätzliche Schnittstelle zu unterstützen.
Verwenden Sie mehr permanente geschweißte Verbindungen, wenn
- Die hygienischen Konsequenzen zusätzlicher Schnittstellen sind hoch.
- Die Baugruppe ist relativ fest und profitiert nicht ausreichend von routinemäßigem Öffnen.
- Das Projekt möchte die Anzahl lokaler Dichtungen und die Montagevariabilität reduzieren.
- Der Standort ist eine kritische Produktkontakt- oder aseptische Fermentationszone.
Überprüfung eskalieren, wenn
- Probenahmestellen oder Abzweigverbindungen sind beteiligt.
- Es besteht ein Risiko für Totstrecken oder schlechte Entwässerung.
- Wiederholte SIP/CIP-Zyklen sind stark ausgeprägt.
- Der Bereich ist aseptisch, validierungsempfindlich oder inspektionsempfindlich.
- Mehrere Schweiß- und Dichtungsgrenzflächen überlappen sich in einer lokalen Baugruppe.
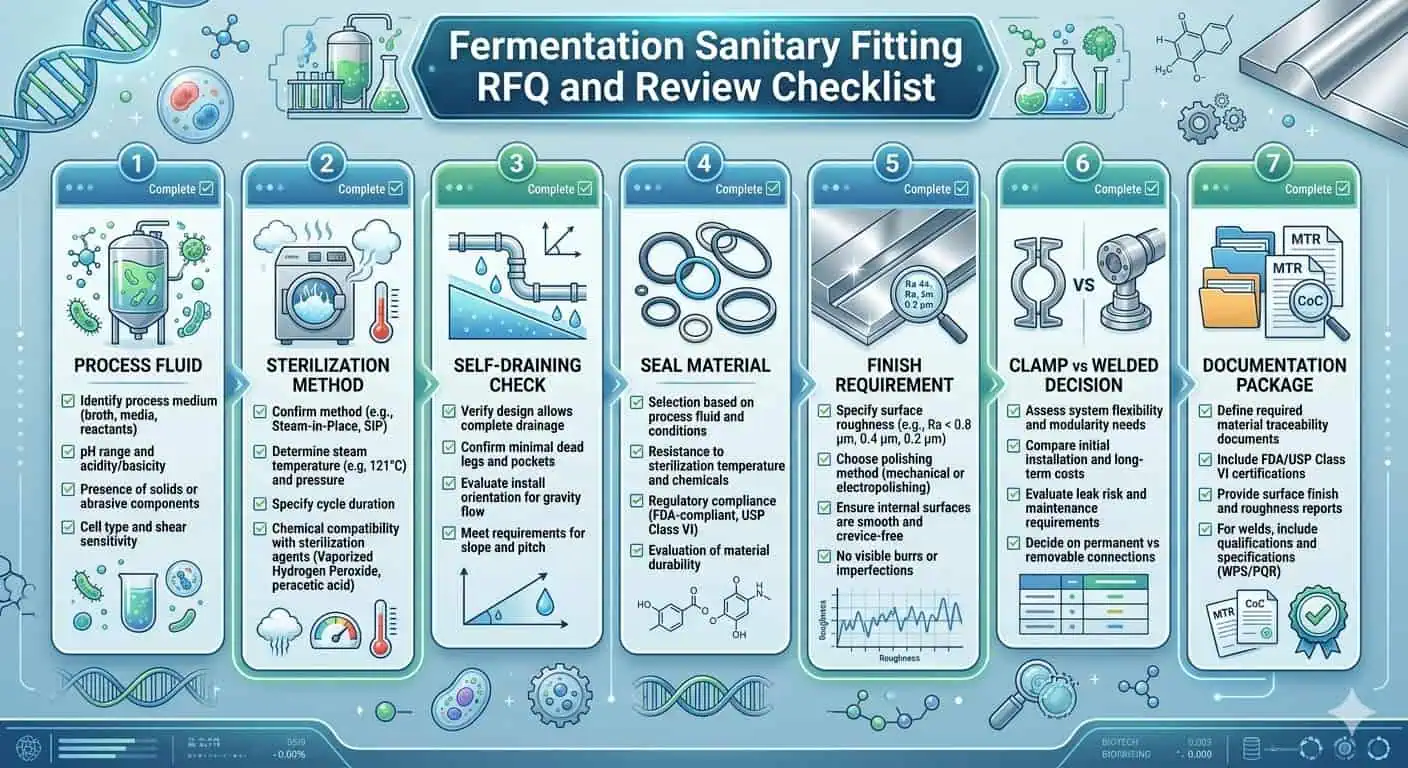
Fragen vor der Angebotsanfrage zu beantworten
- Welches Prozessmedium kommt mit diesem Fitting in Kontakt?
- Wie wird die Baugruppe gereinigt und, falls erforderlich, sterilisiert?
- Ist der Ort unter installierten Bedingungen selbstentleerend?
- Welches Dichtungsmaterial und welcher Austauschprozess gelten?
- Welche Oberflächengüte und Dokumentation sind erforderlich?
- Ist dieser Standort besser durch Zugänglichkeit oder durch eine reduzierte Anzahl von Schnittstellen bedient?
Fazit: Die richtige Fermentationsarmatur ist diejenige, die das tatsächliche hygienische Risiko reduziert.
Die beste hygienische Armatur für ein Fermentationssystem ist nicht die gängigste Katalogoption. Es ist die Armatur, die das tatsächliche hygienische Risiko der genauen lokalen Baugruppe reduziert, wo die Fermentation am wenigsten nachsichtig ist. In der Praxis bedeutet dies, Armaturen nach Reinigungsfähigkeit, Entleerbarkeit, Sterilisationskompatibilität, Dichtungsbelastung und Lebenszyklusrealität auszuwählen, anstatt nur nach Anschlussart.
Für das vollständige Biotech-Themencluster, verbinden Sie diesen Artikel mit Hochreinheits-Rohrleitungsdesign für Biotechnologieanlagen, 316L Edelstahl in Bioprozessanwendungen, Hygienische Rohrleitungswartung und Validierungsüberlegungen, Dokumentation und Rückverfolgbarkeit in Pharma-Rohrleitungsprojektenund Auswahl von Hygienefittings für GMP-Anlagen.
FAQ
Wie wählt man Hygienisch-Fittings für Fermentationssysteme aus?
Beginnen Sie mit dem tatsächlichen Prozess, der Sterilisationsbelastung, dem Reinigungsregime und dem lokalen hygienischen Risiko. Wählen Sie dann die Armatur, die Reinigungsfähigkeit, Entleerbarkeit, Dichtungskontrolle und Lebenszykluswartbarkeit in dieser genauen Baugruppe am besten unterstützt.
Sind Klemmverschraubungen immer für Fermenterrohrleitungen geeignet?
Nein. Klemmarmaturen sind nützlich, wo kontrollierter Zugang und Modularität tatsächlich benötigt werden, aber sie führen auch eine kontrollierte Dichtungsschnittstelle ein, die möglicherweise nicht die beste Wahl an jedem hochkritischen Ort ist.
Beeinflussen Hygienisch-Fittings die CIP- und SIP-Leistung?
Ja. Armaturengeometrie, Dichtungsposition, Abzweigkonfiguration und Entleerbarkeit können erheblich beeinflussen, wie gut eine lokale Baugruppe reinigt, sterilisiert und Kondensat oder Reinigungslösung abführt.
Wo entwickeln Fermentationssysteme am häufigsten armaturbezogene hygienische Risiken?
Am häufigsten an Probenahmestellen, Abzweigungen, Gasanschlüssen, Ventilgruppen, Bodenabläufen und anderen lokalen Baugruppen, nicht in geraden Rohrleitungsabschnitten.
Ist 316L ausreichend, um eine Fermentationsarmatur hygienisch zu machen?
Nein. 316L ist eine gängige Materialbasis, aber das endgültige hygienische Ergebnis hängt immer noch von Oberfläche, Schweißintegration, Dichtungsstrategie, Abzweiggeometrie und Dokumentationskontrolle ab.
Wann sollte eine geschweißte Verbindung einer Klemmenverbindung vorgezogen werden?
Üblicherweise, wenn der Standort hochkritisch, relativ stabil ist und mehr von einer reduzierten Anzahl von Schnittstellen als von einer routinemäßigen Öffnung profitiert.




