Hygienic piping maintenance and validation are not just about running a cleaning cycle on schedule. In biotech and pharmaceutical facilities, long-term piping reliability depends on whether the system still drains, cleans, seals, and performs the way the original design intended after startup, repeated use, maintenance intervention, and field changes. A line can remain mechanically intact and still become a hygienic risk if local assemblies start holding liquid, seal interfaces age poorly, weld-adjacent areas become harder to inspect, or documentation no longer proves the system remains in a controlled state.
That is why good hygienic piping maintenance is inseparable from validation logic. Maintenance does not only protect uptime. It protects cleanability, contamination control, change-control discipline, and the evidence chain needed to show that the system still supports its validated use. In practice, the first hygienic failure usually does not appear in the middle of a straight tube. It appears at a sample branch, a gasketed joint, a low-drain point, a valve body, or a repaired local weld where the system is least forgiving.
This guide explains how engineers maintain and validate hygienic piping systems in biotech facilities, what actually fails first in real operation, how to prioritize inspection and maintenance effort, when maintenance activity should trigger validation review, and how to connect component-level upkeep with long-term hygienic control.
What Hygienic Piping Maintenance Really Means
Maintenance Is More Than Scheduled Cleaning
In hygienic piping, maintenance is a control system, not a calendar event. Routine cleaning matters, but it is only one part of the lifecycle. Real maintenance also includes drainability review, gasket and diaphragm replacement logic, valve inspection, weld-condition follow-up, local assembly checks, and documentation of what changed. In biotech environments, a line is not well maintained just because it has been cleaned on time. It is well maintained when the highest-risk local assemblies are still behaving hygienically and the facility can prove it.
This is consistent with FDA 21 CFR 211.67, which requires written procedures for equipment cleaning and maintenance, including responsibility, schedules, methods, and protection of clean equipment after cleaning. In engineering terms, that means a hygienic maintenance program should be written as a contamination-control system, not as a generic PM sheet.
Expertenrat: if the maintenance plan treats all line sections equally, it is usually too mechanical and not hygienic enough. The system should be maintained by local risk, not by pipe length.
Why Maintenance and Validation Cannot Be Separated
A hygienic piping system is only truly maintained if it remains inside its validated operating intent. Replacing a gasket, changing a diaphragm, revising a branch, repairing a weld, or even adjusting supports can change how a local assembly drains, seals, or cleans. That may not stop production immediately, but it can change the assumptions behind cleaning verification, surface condition expectations, or change-control records. In regulated biotech work, that is why maintenance activity sometimes becomes a validation issue.
Typischer Branchenfall: a loop stayed leak-tight after a local weld repair, but validation review still had to reopen because the repaired section changed the original weld record set and raised new questions about local surface condition and inspection status. The problem was not visible leakage. The problem was that the hygienic evidence chain had changed.
This is also why EU GMP Anhang 1 places contamination control inside a broader lifecycle strategy. Preventive maintenance, investigation, root cause analysis, CAPA, and periodic CCS review all influence whether the facility remains in a controlled state.
Why Hygienic Failure Usually Starts at Local Assemblies
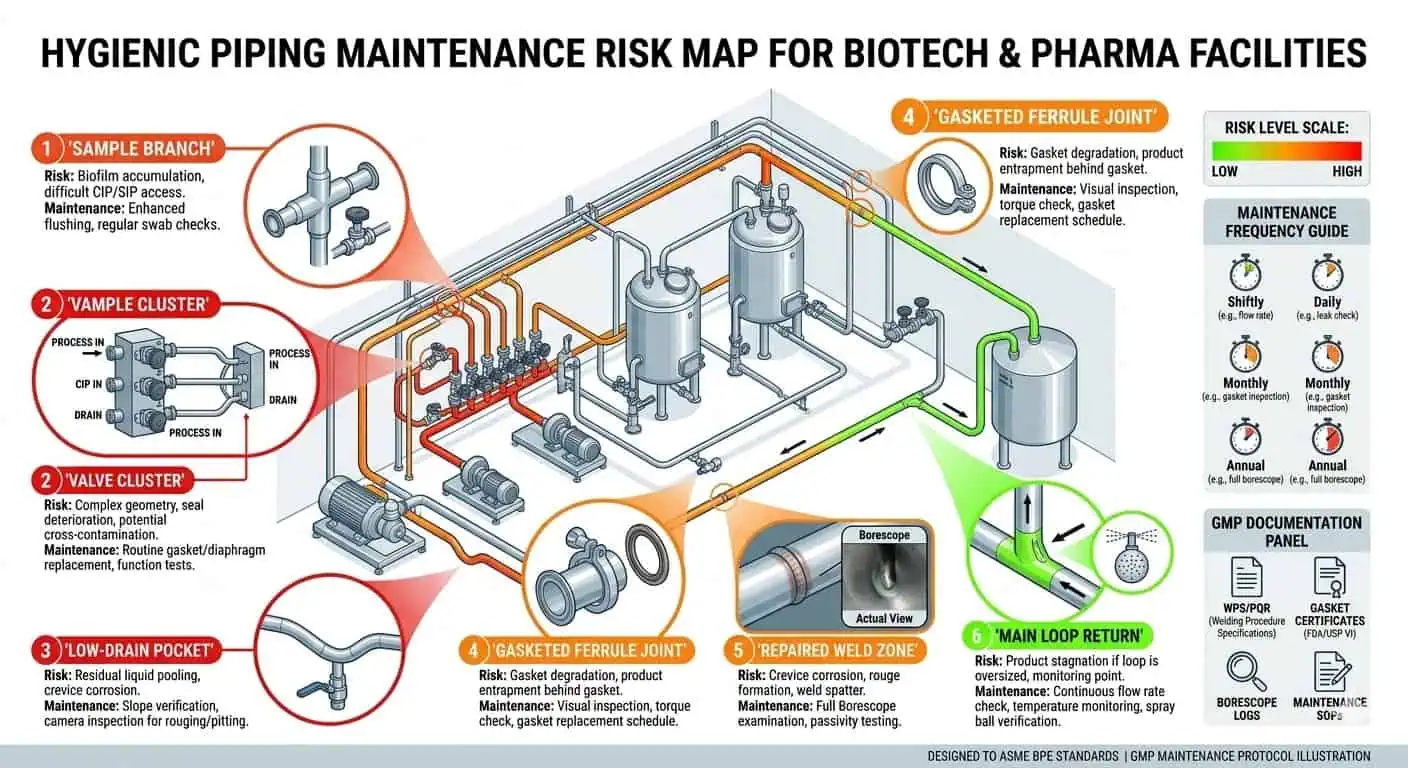
The first weak point in a hygienic piping system is rarely the straight run. It is usually the sample point, valve cluster, branch tee, gasketed ferrule, local manifold, or low-drain pocket where flow exchange is weaker and hygienic consequence is higher. One line may look stable at the main return while one sample assembly keeps creating repeat inspection findings. That is not unusual. It is one of the most common real-world maintenance patterns in hygienic systems.
That is why this article should be read together with Hochreinheits-Rohrleitungsdesign für Biotechnologieanlagen. The assemblies that are hardest to maintain are often the assemblies that were hardest to design correctly in the first place.
The Main Failure Modes Engineers Actually See
Dead Legs and Weak Branch Geometry
Dead-leg behavior remains one of the most repeated hygienic maintenance problems because it usually becomes visible only after real operation begins. A branch may look acceptable on a drawing, but once the line has seen cleaning cycles, hold-up, shutdown periods, and real operator use, one local assembly may repeatedly retain fluid or show inconsistent cleaning response. The main loop can still look healthy while the branch does not.
Typischer Branchenfall: the main line repeatedly met cleaning expectations, but one sample branch continued to generate concern because local exchange was weak and the branch geometry was never truly behaving like the rest of the circuit. The corrective action was not “clean harder.” It was to re-evaluate the local assembly as a hygienic design and maintenance problem.
Expertenrat: evaluate the hardest local branch, not the easiest straight run. If one local assembly cannot exchange fluid and drain reliably, that is the design that matters.
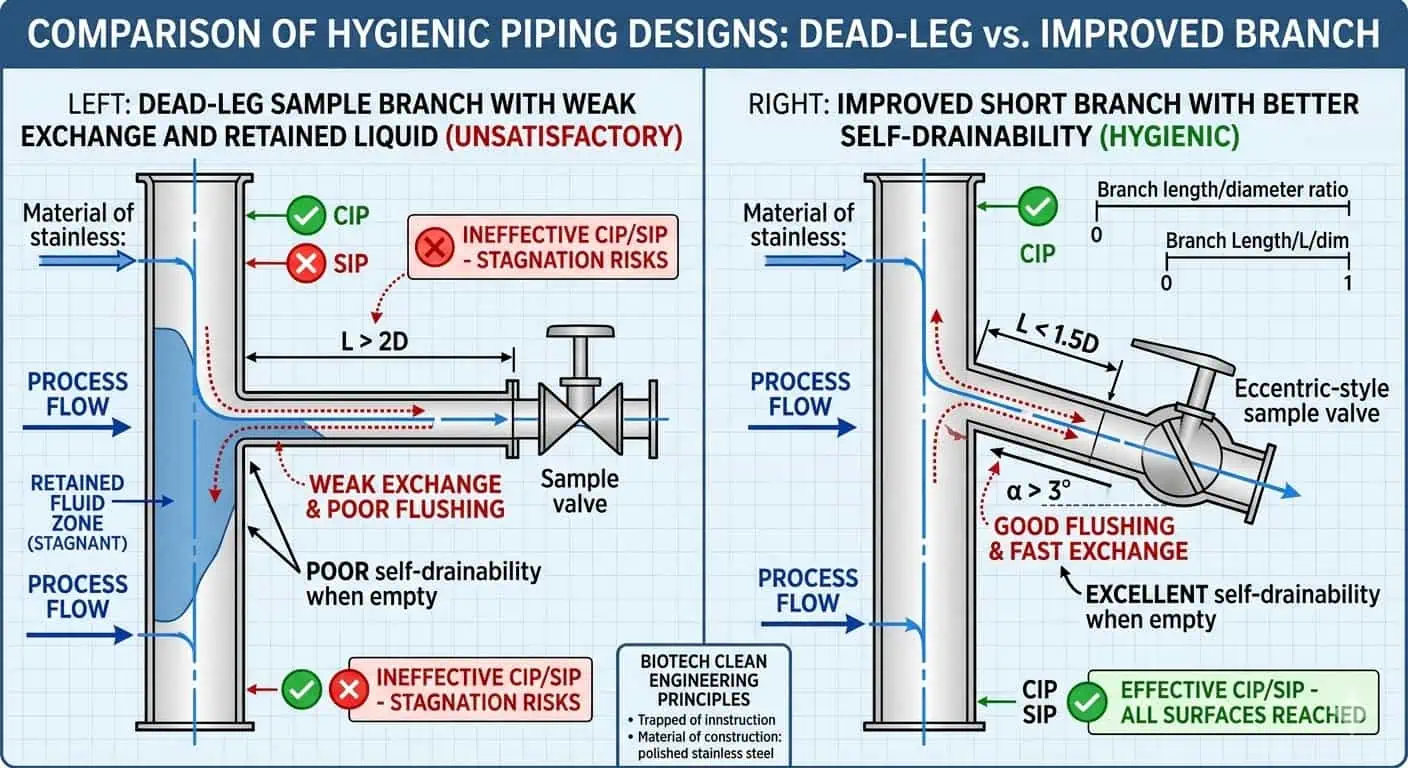
Poor Drainability and Retained Liquid
Retained liquid is one of the clearest signs that a hygienic line is no longer behaving as intended. It changes the next cleaning cycle, increases local contamination risk, weakens drying performance, and makes troubleshooting less reliable. In biotech systems, retained rinse water or condensate in a local low point is not just a nuisance. It is often the first sign that a support position changed, a valve orientation is wrong, a branch is under-reviewed, or the original drainability assumption no longer matches installed reality.
Typical engineering range: even a short retained section in a branch, sample point, or valve body can dominate the hygienic risk of a much longer loop if that location repeatedly traps fluid after CIP or SIP cooldown. The exact consequence depends on process fluid, temperature history, cleaning regime, and hold time.
Expertenrat: when a line repeatedly underperforms hygienically, check where the fluid stays first. The first useful maintenance question is usually not “Did the cycle run?” but “Did the worst local assembly actually empty?”
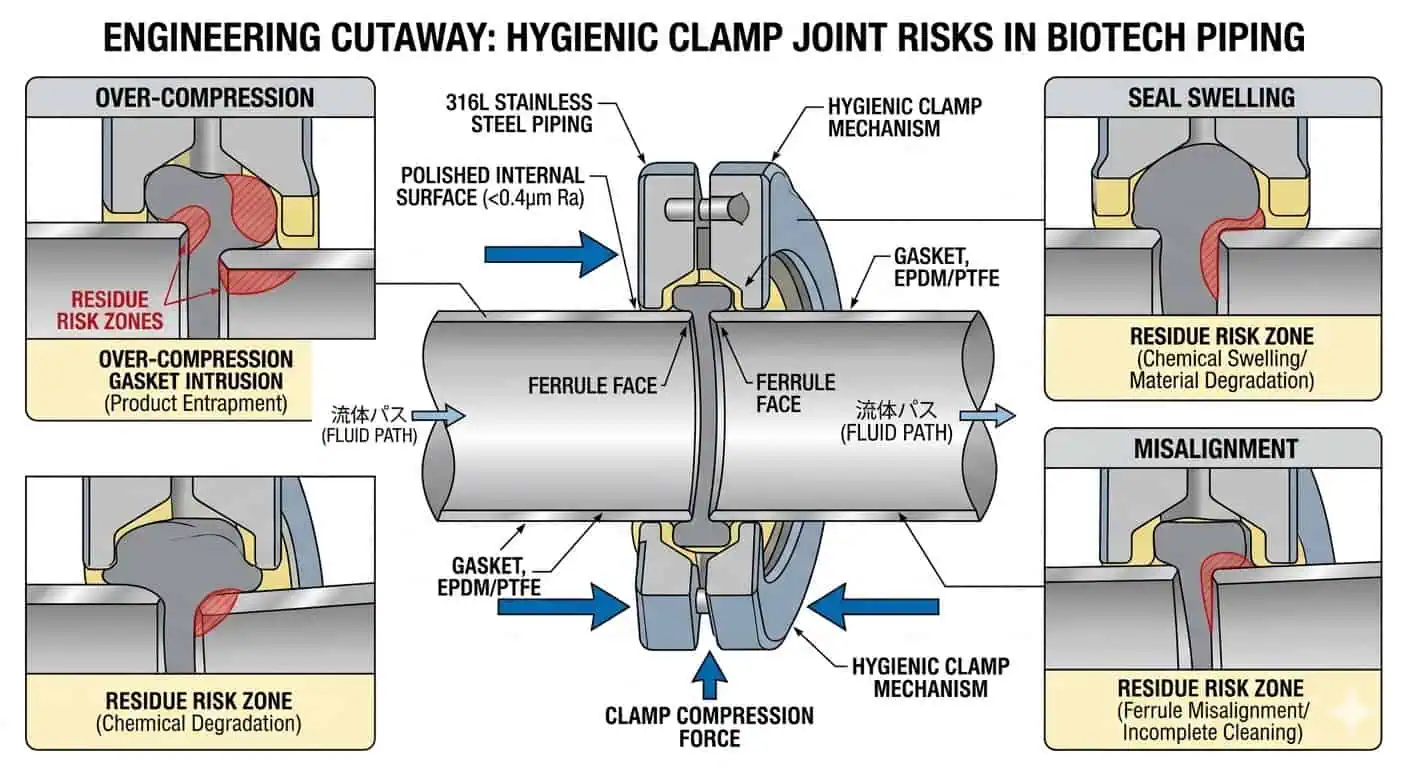
Seal and Gasket Degradation
Many “piping problems” are actually seal-interface problems. Gaskets, diaphragms, valve seats, and other non-metallic wetted parts often become the first true weak point because they experience repeated compression, thermal cycling, chemical exposure, steam exposure, and handling during reassembly. A line may keep operating while the real hygienic margin is already shrinking at the seal interface.
Typischer Branchenfall: a clamp connection that no longer behaved predictably was initially treated as a hardware nuisance. Later review showed that the ferrule alignment had become inconsistent and the gasket had been repeatedly over-compressed during interventions. The visible symptom was minor. The underlying hygienic instability was larger.
Typical engineering range: seal life is strongly influenced by chemistry, temperature, pressure cycling, compression history, and handling practice. For that reason, good sites usually combine scheduled replacement with condition-based review instead of relying on one method alone.
Weld-Zone Risk
The weld zone often dominates hygienic risk more than the general tube wall does. Surface irregularity, inconsistent purge quality, repair history, local discoloration, or finish mismatch around a weld can turn a technically acceptable system into a maintenance-sensitive one. This is one of the most realistic failure patterns in biotech piping: the base material is fine, but the local fabricated condition becomes the weak point over time.
Typischer Branchenfall: the selected material met specification, but one repaired weld in a branch-dense area repeatedly triggered additional inspection because the local surface condition no longer matched the hygienic intent of the loop. The lesson was not that the material choice was wrong. The lesson was that weld quality and maintenance evidence cannot be separated.
For the material and finish side of this logic, connect this article with 316L Edelstahl in Bioprozessanwendungen.
Valve, Sample, and Instrument Assemblies
Valves and local process interfaces require more maintenance attention because they combine more hygienic variables in less space. Seats, diaphragms, cavities, branch geometry, drainback behavior, and repeated operator interaction all converge there. In many systems, the real maintenance burden is not the main line. It is the assembly that has the highest hygienic consequence and the smallest margin for error.
What a Good Hygienic Piping Maintenance Program Looks Like
Routine Checks
Routine checks should focus on the assemblies most likely to fail hygienically, not just the easiest points to observe. This includes repeat leak locations, sample branches, low-drain points, gasketed joints, valve bodies, and assemblies with a history of intervention or difficult cleaning response. A good routine check is not a walk-by looking only for obvious damage. It is a targeted look at whether the line is still behaving like a cleanable high-purity system.
Preventive Maintenance
Preventive maintenance in hygienic piping should be component-specific, not generic. Tubing runs, clamp interfaces, diaphragms, valve clusters, sample ports, and welded assemblies do not age in the same way. A mature maintenance program reflects that difference. The most stable biotech sites usually do not rely on one blanket interval for everything. They build different review and replacement logic around different hygienic failure modes.
Expertenrat: if one local assembly has a repeat history, shorten the maintenance logic there first instead of spreading the same PM burden across the whole loop.
Trigger-Based Maintenance
Some conditions should trigger immediate review instead of waiting for the next scheduled interval. Repeat leakage, unexplained retained liquid, repeated ATP or visual cleaning concern, local odor or residue carryover, visible weld change, or recurring seal instability usually indicate that the issue is no longer routine. In a hygienic system, repeated abnormal behavior at one location is often more valuable than a long checklist showing the rest of the loop looked normal.
| Maintenance Trigger | What It Usually Means | Recommended Engineering Response |
|---|---|---|
| Repeat leak at same local joint | Seal-interface instability, misalignment, or local assembly stress | Review ferrule fit-up, gasket history, and local geometry instead of tightening only |
| Retained liquid after cleaning | Drainability or local orientation problem | Check supports, slope, valve orientation, and branch behavior |
| Repeat ATP / residue concern | Local cleanability failure | Escalate branch or assembly review, not just cycle settings |
| Local weld discoloration or finish concern | Surface-condition risk or repair-history issue | Review inspection status, repair history, and hygienic significance |
| Unexpected odor or carryover | Retention or seal-related local contamination risk | Inspect local assemblies first, especially sample and valve interfaces |
Validation Considerations After Startup
Validation Is Not Only an Initial Project Activity
Hygienic piping validation does not stop once the system first runs successfully. After startup, the system still has to remain inside the assumptions that made it acceptable in the first place. If maintenance, repair, component substitution, or field adjustment changes how a local assembly behaves, the validation burden may change too. A line that still transfers fluid is not automatically a line that still meets its original hygienic and qualification intent.
This is one reason Annex 1 matters beyond sterile filling rooms. Its CCS framework requires ongoing and periodic review of contamination-control effectiveness, not one-time design approval only.
What Maintenance Changes Should Trigger Validation Review
Not every maintenance action needs the same level of escalation, but some clearly affect validated state more than others. Local weld repairs, branch modifications, changes in seal material, valve replacement at critical hygienic points, support changes that affect drainability, and finish-related rework are all examples that can alter cleanability, inspectability, or documentation closure enough to justify review. In biotech systems, the right question is not just “Did we repair it?” It is “Did we change the assumptions under which this assembly was originally accepted?”
Typischer Branchenfall: one field repair solved a practical routing problem but forced a later re-review because the final branch configuration no longer matched the originally accepted cleanability logic. The mechanical fix was easy. Rebuilding confidence in the hygienic intent took longer.
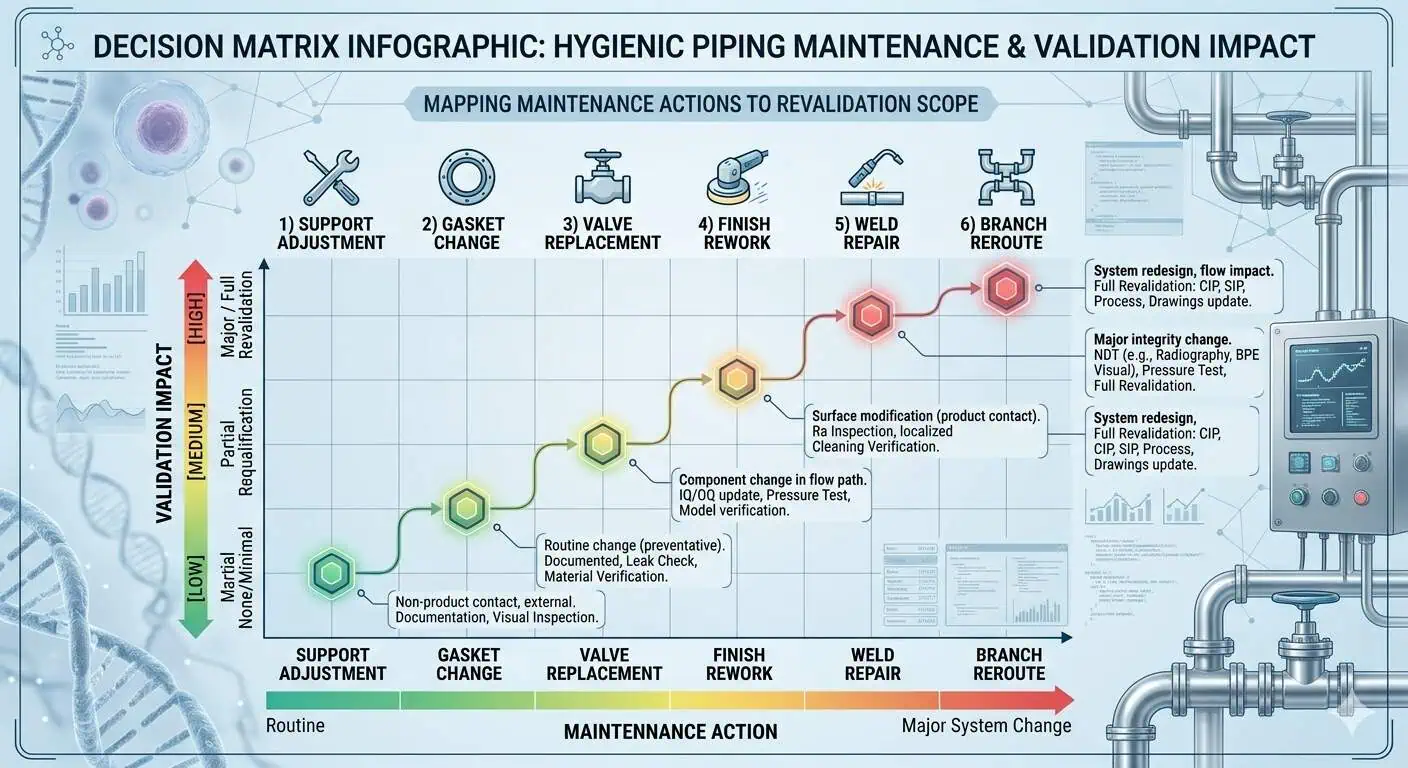
The Difference Between Operable and Still Validated
“Still operable” and “still validated” are not the same standard. A loop may be pressure-tight, visually acceptable, and able to carry process fluid, while still needing a deeper hygienic review because local cleanability, documentation status, or component traceability has changed. In real biotech facilities, that distinction is where many late-stage surprises come from.
Inspection Strategy: Where to Look First
Highest-Risk Locations
The first inspection priority should usually be local assemblies with the highest hygienic consequence. Sample points, low points, branch tees, gasketed ferrule joints, diaphragm valve interfaces, instrument connections, and repaired weld zones are commonly more informative than a general look at the main line. In many loops, one unresolved local assembly tells you more about real hygienic performance than ten clean-looking straight runs.
Visual Review, Local Opening, and Borescope Logic
Inspection depth should match hygienic risk. Some locations can be assessed visually. Others justify controlled opening, targeted local inspection, or borescope review because the real risk is internal and local rather than external and obvious. The engineering objective is not to open everything. It is to inspect the locations most likely to fail in a way that matters hygienically.
Why the Main Loop Can Look Fine While the Local Assembly Fails
This is one of the most repeated patterns in hygienic troubleshooting. The main return looks acceptable, the overall system trend looks stable, but one branch or valve assembly keeps creating repeat concern. That does not mean the system is random. It usually means the inspection strategy has to move from loop-level reassurance to local-assembly evidence.
Maintenance by Component Type
Tubing and Welded Runs
For straight tubing and welded runs, the main concern is not usually the bulk tube wall. The concern is whether the installed line still drains, whether supports or routing have changed local behavior, and whether repaired or high-density weld areas are still consistent with the intended hygienic state. Weld history matters more than outer appearance alone.
Clamp Joints and Ferrules
Clamp-fit assemblies should be maintained as controlled interfaces, not casual access points. Alignment, gasket condition, compression history, local residue behavior, and repeat reassembly quality all influence whether the connection remains hygienically stable over time.
Diaphragm Valves and Valve Blocks
Valve blocks deserve higher maintenance sensitivity because they combine seal wear, local geometry, thermal exposure, and repeated hygienic scrutiny. In many biotech systems, the valve or diaphragm interface is where maintenance and validation logic meet most directly.
Sample Assemblies and Instrument Connections
Small local interfaces often deserve the most aggressive hygienic maintenance attention. The reason is simple: they are easier to overlook, more likely to create local hold-up, and more likely to be touched by operators or modified in the field. In real operation, these locations often define the true maintenance maturity of the loop.
How Maintenance Logic Connects to Regulatory and GMP Thinking
Why Design and Maintenance Must Support Intended Use
FDA 21 CFR 211.63 is valuable because it frames equipment as something that must be appropriately designed for intended use and for cleaning and maintenance. In hygienic piping, that means the maintenance burden is part of the original engineering suitability of the system, not just a later operating inconvenience.
Why Product-Contact Condition Still Matters During Maintenance
FDA 21 CFR 211.65 matters because it requires product-contact surfaces to remain non-reactive, non-additive, and non-absorptive. In practice, maintenance decisions that alter local surface condition, seal family, or component identity should be reviewed through that same product-contact logic, not only through mechanical convenience.
Why Written Cleaning and Maintenance Control Matters
FDA 21 CFR 211.67 is important because it ties equipment cleaning and maintenance to written procedures. That means hygienic upkeep is not a matter of tribal knowledge or local habit. It has to be defined, repeatable, and reviewable. In a biotech facility, undocumented maintenance logic is weak maintenance logic.
Why Annex 1 Makes This a Contamination-Control Topic
EU GMP Annex 1 matters because it frames contamination control as a facility-wide strategy, not a single cleaning event. Its CCS logic explicitly includes design, process validation, preventive maintenance, cleaning and disinfection, monitoring, root-cause investigation, CAPA, and continuous improvement. That is exactly why hygienic piping maintenance should be treated as part of ongoing contamination control and not as a detached utility activity.
For the documentation side of this lifecycle logic, connect this article with Dokumentation und Rückverfolgbarkeit in Pharma-Rohrleitungsprojekten.
How Engineers Actually Decide What to Do Next
Step 1: Identify the Real Hygienic Failure Mode
Start with the actual failure mode, not the most visible symptom. Is the issue local retention, repeat leak, weak seal life, weld-zone concern, branch cleanability, or documentation mismatch? The wrong root-cause framing often leads to the wrong maintenance action.
Step 2: Decide Whether the Issue Is Local or Systemic
Many biotech piping problems are highly local even when the system trend looks stable. That is why one problematic branch should not be diluted by the fact that the rest of the loop appears acceptable. Local hygienic problems require local engineering attention.
Step 3: Decide Whether the Action Changes Validated State
Before acting, ask whether the proposed maintenance step changes more than mechanical condition. If it changes material identity, seal family, finish, branch configuration, weld history, or local cleanability logic, it may also change validation burden.
Step 4: Close the Evidence Chain
The technical repair is not the whole repair. In controlled hygienic systems, the evidence chain also needs to be closed so the line remains maintainable, inspectable, and releasable after the intervention.
Häufige Fehler
Treating Maintenance as a Time Interval Only
A purely time-based maintenance plan usually misses the assemblies with the highest hygienic consequence.
Over-Relying on Cleaning Cycles Without Reviewing Local Geometry
A branch that does not exchange and drain well will not become reliable just because the main cycle ran on time.
Replacing Seals Without Reviewing Compatibility and Compression History
Seal instability is often a local design-and-maintenance issue, not just a consumable issue.
Ignoring Weld-Zone Condition
A correct material with a weak weld history is still a hygienically weaker system than it looks from a distance.
Assuming “No Leak” Means “Hygienically Correct”
A dry local assembly can still be harder to clean, harder to inspect, or less stable than the design intended.
Practical Maintenance and Validation Checklist
Vor dem Anfahren
- Confirm the installed loop drains as intended.
- Check local high-risk assemblies, not only main runs.
- Verify gasket, diaphragm, and valve status at critical hygienic points.
- Confirm that documentation and release evidence match the installed system.
During Routine Operation
- Track repeat leak locations.
- Review retained liquid, odor, or carryover concerns.
- Monitor sample points, branch assemblies, and local valve interfaces closely.
- Escalate repeated local findings instead of averaging them into loop-wide normality.
Before Requalification or QA Review
- Review maintenance history at critical local assemblies.
- Check whether repairs or substitutions changed validated assumptions.
- Confirm weld-related records, component identity, and inspection closure.
- Reassess the hardest-to-clean points, not just the easiest ones to document.

| Maintenance Topic | Wie gute Kontrolle aussieht | Was normalerweise schiefgeht |
|---|---|---|
| Totwasser-Kontrolle | Local branches are treated as high-risk assemblies | Main loop looks fine while one branch keeps failing hygienically |
| Entleerbarkeit | The installed loop empties reliably after use and cleaning | Support or local geometry changes create retained liquid |
| Seal management | Gaskets and diaphragms are treated as controlled components | Repeat leakage or hygiene drift is blamed on “hardware” alone |
| Weld follow-up | Weld repairs and local condition stay linked to inspection logic | Mechanically acceptable repair changes validation burden later |
| Validation awareness | Maintenance actions are reviewed for hygienic impact | Operable system is assumed to still be fully validated |
Conclusion: Hygienic Piping Maintenance Succeeds When the System Is Managed by Local Hygienic Risk, Not by Generic PM Timing Alone
Hygienic piping maintenance and validation succeed when the system is managed by local hygienic risk rather than by generic PM timing alone. The strongest biotech facilities do not rely on main-loop reassurance. They focus on the assemblies that actually fail first: dead-leg-prone branches, low-drain points, seal interfaces, valve clusters, sample assemblies, and weld-sensitive local details.
If you want a stronger biotech topic cluster, connect this article with Hochreinheits-Rohrleitungsdesign für Biotechnologieanlagen, 316L Edelstahl in Bioprozessanwendungen, Auswahl von Hygienefittings für Fermentationssystemeund Dokumentation und Rückverfolgbarkeit in Pharma-Rohrleitungsprojekten.
FAQ
How often should hygienic piping be inspected?
Inspection frequency should follow hygienic risk, not only calendar timing. Assemblies with dead-leg risk, repeat seal intervention, sample-point complexity, or local validation sensitivity usually deserve more frequent review than simple straight runs.
What usually fails first in a hygienic piping system?
The first failure point is usually a local assembly, not the main line. Sample branches, gasketed joints, valve interfaces, low points, and weld-sensitive local details are more likely to create repeated hygienic concern than straight tubing runs.
Do dead legs always require redesign?
Not always, but repeated dead-leg-related hygiene findings usually mean the local assembly needs deeper review. If a branch repeatedly traps fluid or fails to exchange cleaning media well, cleaning harder is usually less durable than correcting the local geometry or connection strategy.
Should gasket replacement be time-based or condition-based?
Good programs usually use both approaches. Time-based control helps prevent uncontrolled aging, while condition-based review helps catch local hygienic instability earlier where thermal, chemical, and reassembly history are harsher than average.
When does maintenance work trigger validation review?
It should trigger review when the action changes more than simple mechanical condition. Weld repair, branch revision, material or seal substitution, finish-related rework, or changes that affect drainability and local cleanability can all alter validated assumptions.
Why can a main loop pass while one sample branch still fails?
Because local hygienic behavior is not always represented by loop-level results. A branch may exchange fluid, drain, or clean differently from the main run, so the local assembly can remain the real weak point even when the overall loop looks acceptable.




