Für Produktionslinien für Körperpflege und Kosmetik wird 316L-Edelstahl oft in produktberührenden Bereichen bevorzugt, die häufiger Reinigung, längere Nassbelastung, höhere Hygienekonsequenzen oder ein größeres Risiko lokaler Korrosion aufweisen. Das bedeutet nicht, dass jede Komponente in jeder Linie automatisch aufgerüstet werden muss. In realen Anlagenentscheidungen vergleichen Ingenieure 316L vs. 304 Edelstahl basierend auf Produktchemie, Reinigungsregime, Entwässerbarkeit, Schweißqualität, Oberflächenbeschaffenheit und langfristigem Wartungsrisiko.
Direkte Antwort: Wenn eine Linie Lotionen, Shampoos, Cremes, Gele, Seren, Flüssigseifen oder andere Körperpflegeprodukte verarbeitet und häufige CIP- oder COP-Reinigung, Spülpausen, nasse Abschaltungen oder wiederholte Produktwechsel sieht, wird 316L üblicherweise zuerst für produktberührende Rohrleitungen, Tanks, Ventilgruppen, Verteiler, Pumpenstränge und Füllzuführungen geprüft. Bei milder Belastung und geringerem Hygienerisiko kann 304 nach technischer Überprüfung noch akzeptabel sein.
Deshalb lautet die praktische Frage nicht einfach “Ist 316L besser als 304?” Die bessere Frage ist “Wo schafft 316L in dieser Körperpflegeproduktionslinie echten Lebenszykluswert?” Dieser Ansatz führt zu besseren Spezifikationen als pauschales Upgrading oder kostengetriebenes Downgrading.
Expertenmeinung:
In hygienischen Prozesssystemen werden Materialausfälle selten allein durch die Legierungsgüte verursacht. Die Leitungsleistung hängt normalerweise von der kombinierten Wirkung von Produktchemie, Reinigungsregime, Schweißqualität, Oberflächenbeschaffenheit, Entwässerungsfähigkeit, Dichtungsauswahl und der tatsächlichen Stilllegung, Spülung und Wiederinbetriebnahme des Systems zwischen Wechseln ab.
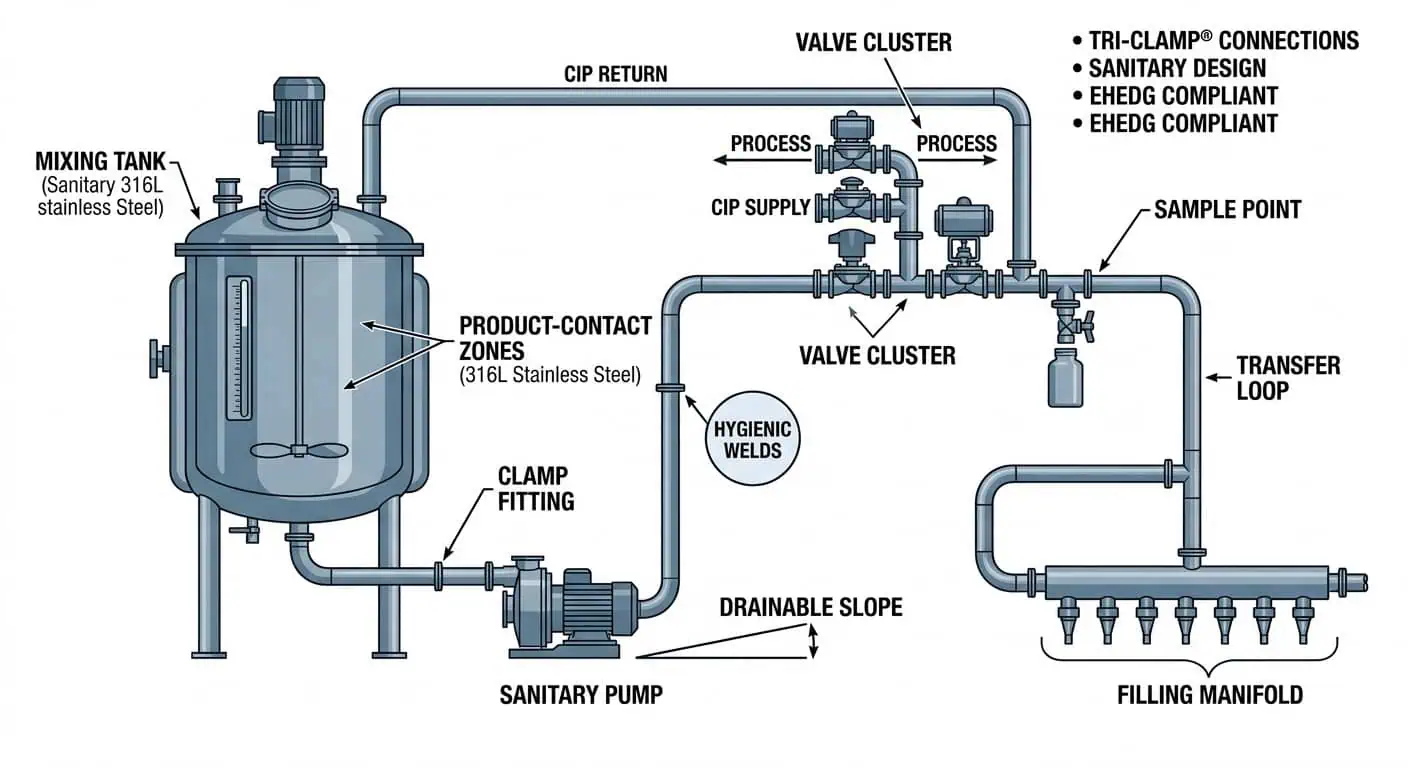
Wo 316L in Produktionslinien für Körperpflege und Kosmetik eingesetzt wird
Typische produktberührende Bereiche, in denen 316L häufig in Betracht gezogen wird
316L-Edelstahl wird am häufigsten für Produktkontaktzonen bewertet, in denen hygienische Leistung und Korrosionsbeständigkeit langfristig entscheidend sind. In Produktionslinien für Körperpflege umfassen diese Bereiche üblicherweise Mischbehälterauslässe, Umlaufschleifen, Transferverteiler, Pumpensaug- und -auslassrohre, Zuführleitungen von Abfüllmaschinen, CIP-Rücklaufabschnitte, Ventilgruppen, Probenahmepunkte und andere Baugruppen, die wiederholter Nassbeanspruchung und regelmäßiger chemischer Reinigung ausgesetzt sind.
- Misch- und Transferkreisläufe: oft viskosen Produkten, wiederholten Spülzyklen und häufigen Rezepturwechseln ausgesetzt.
- Füll- und Dosierverteiler: üblicherweise empfindlich gegenüber Rückständen, Oberflächenzustandsstabilität und Hygienerisiken beim Neustart.
- Ventilgruppen und Probenahmestellen: Orte, an denen Geometrie und Reinigungsfähigkeit oft genauso wichtig sind wie das Materialgrad.
- Tankauslassstutzen und totraumgefährdete Zonen: Bereiche, in denen Ingenieure die Materialwahl zusammen mit hygienischer Anordnung und Rücklaufverhalten überprüfen.
Nicht jeder Abschnitt der Leitung benötigt die gleiche Materialstrategie
Nicht alle Produktionslinien für Körperpflege erfordern eine vollständige 316L-Spezifikation von Anfang bis Ende. Ingenieure trennen üblicherweise Produktkontaktzonen, nicht-produktkontaktierende Stützen, Versorgungsanschlüsse, nur Spülabschnitte und niedrigrisikodienstbereiche. Die richtige Frage ist nicht “Sollte die gesamte Leitung aus 316L bestehen?” aber “Welche Abschnitte rechtfertigen den zusätzlichen Materialaufschlag basierend auf Exposition, Reinigungsfähigkeit, Wechselfrequenz und Lebenszyklusanforderungen?”
| Prozessbereich | Hauptrisiko / Exposition | Warum 316L bevorzugt werden kann | Wann 304 noch in Betracht gezogen werden kann |
|---|---|---|---|
| Mischbehälterauslass und Rückführschleife | Häufiger Nassbetrieb, Reinigungschemikalien, Produktrückstände, Neustartexposition | Bessere Korrosionsreserve und stärkere Langzeitstabilität im hygienischen Einsatz | Wo Formulierung, Reinigungsbelastung und Exposition mild und vollständig geprüft sind |
| Füllzuführungsverteiler | Hohe Reinigungsanforderungen, kurze Umrüstzeiten, geringe Toleranz für Rückstände | Unterstützt eine einfachere Oberflächenwiederherstellung und verringert das Hygienerisiko | Nur in Produktfamilien mit geringerem Risiko und gut kontrollierten Reinigungspraktiken |
| CIP-bezogene produktberührende Rohrleitungen | Wiederholte alkalische/saure Reinigung und Nass-Trocken-Zyklen | Oft gewählt, um korrosionsbedingte Wartungsprobleme zu reduzieren | Projektabhängig und sollte ohne Überprüfung nicht angenommen werden |
| Nicht-produktberührender Versorgungsbereich | Geringere Hygienefolgen | Normalerweise nicht der erste Bereich, der die 316L-Begründung vorantreibt | 304 oder andere Optionen können je nach Anwendung akzeptabel sein |
Für umfassendere Layout- und Entwässerungsentscheidungen sollte die Materialauswahl gemeinsam mit Hygienische Rohrleitungskonstruktion für Kosmetikherstellung, überprüft werden, da eine schlechte Geometrie den Wert eines höherwertigen Materials lange bevor die Legierung selbst zur tatsächlichen Grenze wird, untergraben kann.
Tipp:
Wenn ein Käufer “316L” auf einer hygienischen Rohrleitungszeichnung sieht, sollte dies nicht das Ende der Überprüfung sein. Die Materialgüte sollte gemeinsam mit Oberflächenbeschaffenheit, Schweißnahtbehandlung, Reinigungsfähigkeit und ob Verzweigungslänge, Entwässerungsneigung und Dichtungsdetails für die tatsächliche Produktfamilie geeignet sind, überprüft werden.
316L vs. 304 Edelstahl für Produktionslinien in der Körperpflege
Warum Ingenieure häufig 316L gegenüber 304 wählen
In Produktionslinien für Körperpflege und Kosmetik hängt die Entscheidung zwischen 316L und 304 normalerweise von Korrosionsreserve, Stabilität der Schweißzone, Reinigungsfähigkeit und langfristiger Betriebssicherheit ab. Ingenieure wählen 316L normalerweise nicht, weil es hochwertiger klingt. Sie wählen es, weil einige produktberührende Systeme anfälliger für lokale Lochfraß, Spaltkorrosion, wiederholte nasse Stillstände und chemische Reinigungsexposition sind, als Käufer während der anfänglichen Ausschreibungsphase erwarten.
Was Ingenieure unter “besserer Korrosionsbeständigkeit” unter realen Anlagenbedingungen verstehen
In hygienischen Systemen für Körperpflege ist “bessere Korrosionsbeständigkeit” nicht nur ein Katalogbegriff. Es bedeutet normalerweise ein stabileres Materialverhalten bei wiederholter Exposition gegenüber Produktfeuchtigkeit, Spülwasser, Reinigungschemikalien, zurückgehaltenen Rückständen und Schweißzonenspannung in einer hygienischen Umgebung. Dies ist besonders wichtig in Systemen, die häufige Wechsel durchführen oder Formulierungen mit Salzen, Tensiden, Wirkstoffen, Konservierungsmitteln, Duftstoffen oder anderen Inhaltsstoffen verarbeiten, die die praktische Korrosionsbelastung in lokalen Bereichen erhöhen, anstatt über die gesamte Leitung.
Warum die kohlenstoffarme “L”-Qualität in der geschweißten hygienischen Fertigung wichtig ist
Niedrigkohlenstoffstahl 316L wird oft bevorzugt, weil hygienische Systeme stark auf geschweißte Konstruktion und Oberflächenqualität nach dem Schweißen angewiesen sind. In der Praxis wählen Ingenieure nicht einfach zwischen zwei chemischen Bezeichnungen. Sie versuchen vielmehr, die Wahrscheinlichkeit zu verringern, dass geschweißte produktberührende Bereiche mit der Zeit, insbesondere nach wiederholten Reinigungs- und Stillstandszyklen, zum Schwachpunkt des Systems werden. Deshalb sollten die Qualität der Orbitalschweißung, die Kontrolle der Anlassfarben und die Oberflächenwiederherstellung nach der Fertigung zusammen mit der Legierungsangabe und nicht erst nach der Beschaffung überprüft werden.
Warum 316L oft eine Risikominderungsentscheidung ist und keine universelle Anforderung
Viele Anlagen spezifizieren 316L, weil es zukünftige Betriebsrisiken reduzieren kann – nicht weil jeder Prozess es unbedingt erfordert. Wo die Reinigungshäufigkeit hoch ist, die Stillstandszeiten der Leitung teuer sind und Hygieneverstöße echte Produktionskonsequenzen haben, kann der zusätzliche Materialaufwand durch geringere Wartungsempfindlichkeit und stabileren Langzeitbetrieb gerechtfertigt sein. Im Gegensatz dazu, wo die Belastung mild und die Produktchemie weniger anspruchsvoll ist, kann eine pauschale 316L-Aufrüstung nicht den gleichen Nutzen bringen.
| Bewertungsfaktor | 304 Edelstahl | 316L Edelstahl | Was dies in realen Projekten bedeutet |
|---|---|---|---|
| Allgemeine Eignung für hygienische Anwendungen | Kann bei weniger anspruchsvollen Bedingungen akzeptabel sein | Oft bevorzugt für anspruchsvollere Produktkontakt-Anwendungen | Die Wahl sollte auf Exposition und Reinigungsaufwand basieren, nicht nur auf Gewohnheit |
| Korrosionsspielraum bei lokaler Nässe / chemischer Exposition | Geringerer Spielraum unter anspruchsvolleren Bedingungen | Typischerweise stärkerer Spielraum für aggressive hygienische Anwendungen | Nützlich, wenn das Werk weniger Überraschungen bei langfristiger Nutzung wünscht |
| Geschweißte hygienische Fertigung | Verwendbar, aber anwendungsabhängig | Häufig bevorzugt in geschweißten hygienischen Anlagen | Besonders relevant in der Nähe von produktberührenden Schweißnähten und polierten Bereichen |
| Lebenszyklusstabilität | Kann für Systeme mit geringerem Risiko ausreichend sein | Oft für stärkeres langfristiges Vertrauen ausgewählt | Wichtig, wo Wechsel, Reinigung und Verfügbarkeitsziele streng sind |
Expertenmeinung:
Die bessere technische Frage ist normalerweise nicht “Welche Güteklasse ist im Allgemeinen besser?”, sondern “Wo zahlt sich der zusätzliche Spielraum von 316L in dieser Leitung tatsächlich aus?” Das führt normalerweise zu einer besseren Spezifikation, als das gesamte System als eine undifferenzierte Risikozone zu behandeln.
Was 316L in der Herstellung von Körperpflegeprodukten tatsächlich herausfordert
Chloride sind nicht das einzige Problem, das Ingenieure im Auge behalten sollten
Selbst wenn 316L ausgewählt wird, beseitigt das das hygienische Risiko nicht von selbst. Oberflächenbeschaffenheit, Geometrie, Reinigungswirkung und zurückgehaltene Feuchtigkeit können weiterhin Problembereiche verursachen. In der Herstellung von Körperpflegeprodukten ist die lokale Exposition oft wichtiger als die nominelle Materialangabe in der Zeichnung. Die hartnäckigsten Korrosions- und Hygienebefunde beginnen normalerweise dort, wo Flüssigkeiten verweilen, Feststoffe auf einer Oberfläche trocknen oder Reinigungschemikalien länger als beabsichtigt eingeschlossen bleiben.
- Zurückgehaltene Reinigungslösung: kann die lokale Exposition an schlecht entwässerten Punkten erhöhen.
- Viskose Produktrückstände: können die Kontaktzeit verlängern und die Oberflächenrückgewinnung erschweren.
- Spalten und Schattenzonen: bergen oft mehr Risiko als Käufer zunächst erwarten.
- Schweiß- und Oberflächenunregelmäßigkeiten: können den Vorteil der Spezifikation einer höherwertigen Legierung verringern.
- Feuchte Stillstandszeiten: kann das lokalisierte Risiko zwischen Produktionskampagnen erhöhen.
Geometrie und Oberflächenbeschaffenheit sind oft wichtiger als Käufer erwarten
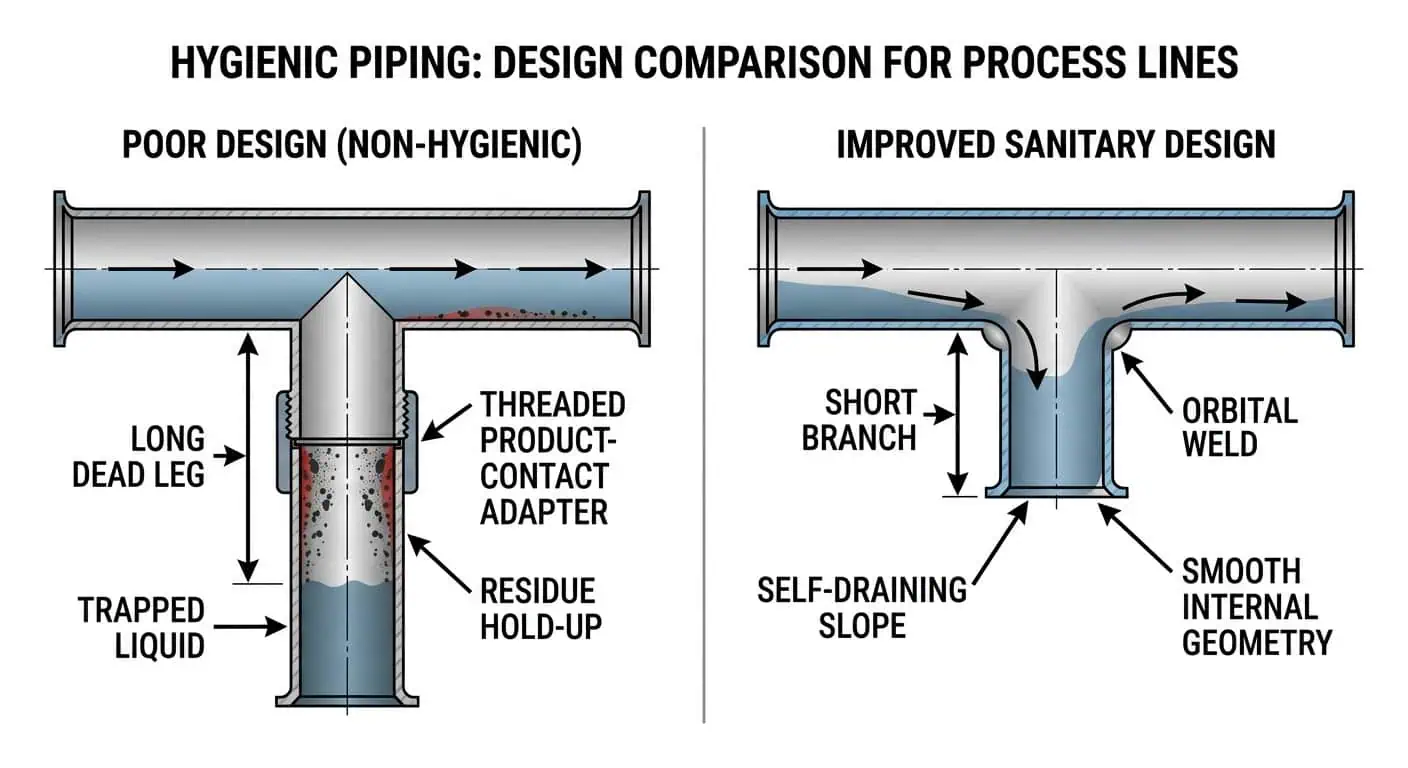
Bei vielen hygienischen Ausfällen liegt die Ursache nicht darin, dass der gewählte Stahlgrad “falsch” war, sondern dass das System lokale Bedingungen schuf, die das Material dann tolerieren musste. Lange Totstrecken, Niedrigstrom-Abzweigungen, ungeeignete Gefälleneigungen, unfertige Schweißbereiche, raue manuelle Reparaturen, Gewindeadapter mit Produktkontakt und schwer zu reinigende Verbindungsdetails können alle eine solide Spezifikation in ein Wartungsproblem verwandeln.
ASME BPE behandelt eine Totstrecke als einen Bereich der Einschließung und bewertet sie anhand der L/D-Geometrie, weshalb Abzweigungslänge und Abzweigungsdetails auf hygienischen Leitungen nicht als Zeichnungsdetails abgetan werden sollten. Diese geometrische Logik ist in der Körperpflege genauso wichtig wie in der Bioprozessrohrleitung, wenn Rückstände, langsames Spülen oder Validierungsempfindlichkeit hoch sind.
Deshalb sollten Ingenieure die Materialauswahl zusammen mit überprüfen. Hygienische vs. industrielle Rohrarmaturen. Eine Leitung kann die richtige Legierungsqualität aufweisen und dennoch unterdurchschnittlich abschneiden, wenn die Fittings und die Verbindungsgeometrie nicht für hygienische Reinigung und Entwässerung geeignet sind.
Praktische Industriefälle
Lotion-Transfer-Schleife: Oberflächenprobleme traten früher als erwartet auf
Eine Lotion-Transfer-Schleife in einer Mehrprodukt-Körperpflegeanlage wurde auf Basis einer 316L-Produktkontaktspezifikation gebaut. Dennoch entwickelte die Leitung früher als erwartet hygienebezogene Wartungsprobleme während des Hochfrequenz-Umstellbetriebs. Die Untersuchung wies nicht allein auf die Legierungsqualität hin. Stattdessen waren die Hauptursachen unvollständige Rückentwässerung an einem Abzweigungsabschnitt, schwer zu reinigende lokale Geometrie und inkonsistente Oberflächenqualität nach der Fertigung. Nachdem der Abzweig neu gestaltet wurde, das Totstreckenverhältnis reduziert und die Anforderung an die Oberflächengüte für das neu geschweißte Rohrstück verschärft wurde, verbesserte sich die Leitungsregeneration und die Reinigungskonsistenz wurde leichter aufrechtzuerhalten.
Ein zweites Praxisbeispiel betraf eine Shampoo-Umwälzanlage, bei der die Bediener nach alkalischer Reinigung und Spülpausen wiederholt Verfärbungen in der Nähe eines Ventilblocks beobachteten. Die Ursache war kein Austausch des Grundmaterials; es handelte sich um eine raue manuelle Nachschweißung und eine lokale Spalte an der Dichtungsauflage. Der Austausch des betroffenen Rohrstücks durch eine fachgerecht bearbeitete 316L-Hygiene-Baugruppe und die Korrektur der Dichtungskompression beendeten das wiederkehrende Problem.
Ein drittes Beispiel stammte von einer Cremefüllanlage, bei der im späteren Projektverlauf eine nicht standardisierte Probenahmeverbindung auf der Produktseite mit einem Gewinde-Adapter hinzugefügt wurde. Die Anlage bestand zunächst die Produktionsinbetriebnahme, aber der Adapterbereich wurde zum ersten wiederkehrenden Hygieneproblem, weil Rückstandsablagerungen und schlechte Entleerbarkeit die nominelle Materialqualität überwogen. Die Korrekturmaßnahme bestand darin, das gewindeführende Produktkontaktteil zu entfernen und durch eine hygienische Membran-Abzweigung zu ersetzen, die dem umgebenden Hygienekonstruktionsstandard entsprach.
Expertenmeinung:
Diese Art von Fall ist in der Hygienetechnik üblich: Das Material mag geeignet sein, aber das System funktioniert dennoch nicht optimal, weil Geometrie, Schweißausführung oder Anschlussdetails nicht auf dem gleichen Standard wie die Materialspezifikation kontrolliert wurden.
Wie Oberflächengüte, Schweißen und Passivierung die Leistung von 316L beeinflussen
Oberflächengüte ist ein Thema der Reinigungsfähigkeit, nicht nur ein visuelles Detail
Für hygienische Systeme beeinflusst die Oberflächengüte die Reinigungsfähigkeit, Rückstandsfreiheit, Umrüstzeiten und das langfristige Wartungsverhalten. Käufer behandeln die Oberflächengüte manchmal als kosmetisches Merkmal, aber Ingenieure sehen sie als Teil der prozessbezogenen Leistungsspezifikation. In der 3-A-Hygiene-Richtlinie werden produktberührende Oberflächen im Allgemeinen mit einer Rauheit von mindestens 32 µin. Ra / 0,8 µm Ra oder glatter erwartet, aber in der praktischen Anlagenarbeit muss das Rauheitsziel weiterhin die Produktrheologie, Reinigungsmethode und Inspektionserwartungen widerspiegeln.
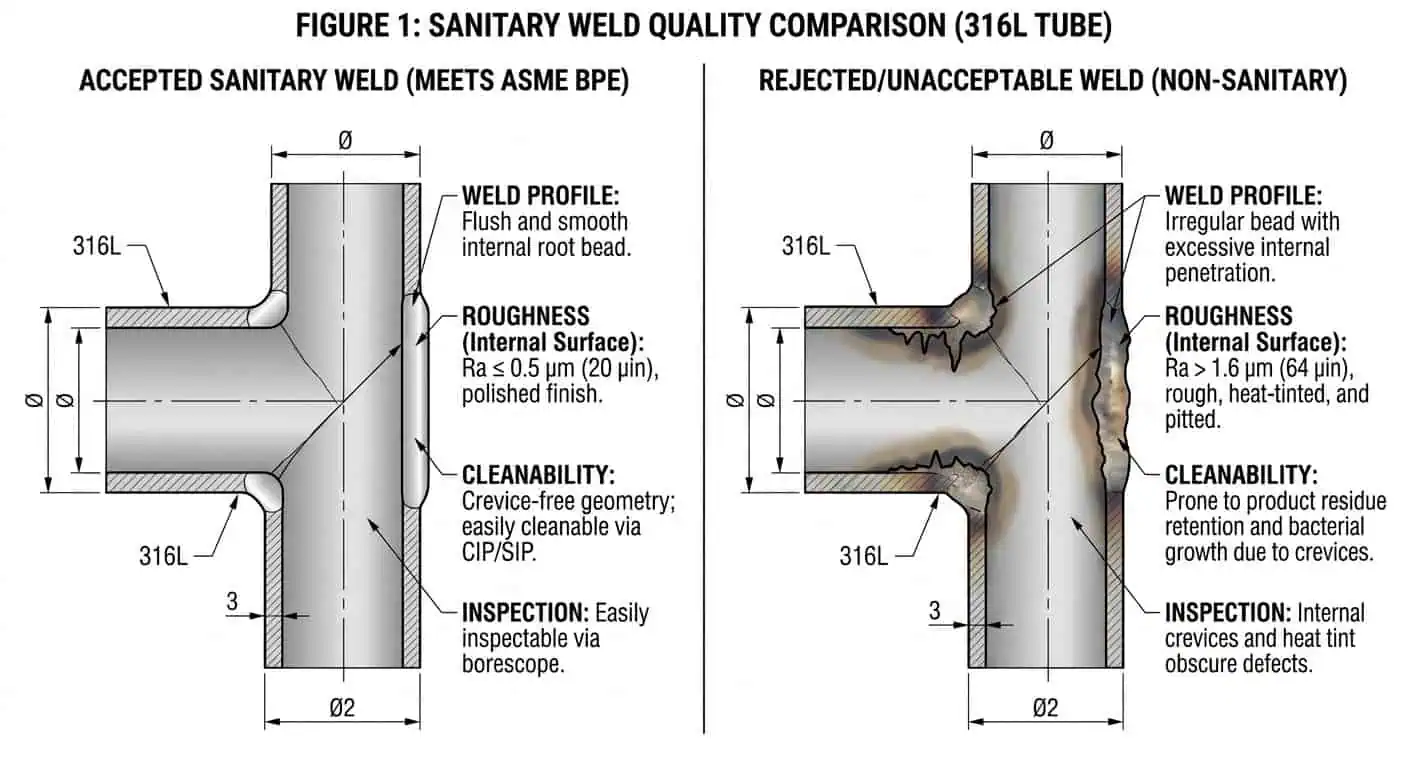
Warum die Schweißausführung in produktberührenden Baugruppen wichtig ist
Die Schweißqualität ist oft einer der wichtigsten praktischen Faktoren in einem 316L-Hygienesystem. Schlecht kontrollierte Schweißnähte können Verfärbungen, lokale Rauheit, Unterbrand, ungleichmäßige innere Geometrie oder Bereiche verursachen, die schwerer zu reinigen und zu inspizieren sind. Im Produktkontaktbereich wird dies zu einem echten Betriebsproblem anstatt zu einem kosmetischen.
- Das innere Schweißprofil sollte einen glatten Produktfluss und Reinigungsfähigkeit unterstützen.
- Wärmefärbung und Zustand nach dem Schweißen sollten auf hygienischen Produktkontaktflächen nicht ignoriert werden.
- Fertigungssteuerungen sollten den Hygieneerwartungen der Leitung entsprechen, nicht nur der Materialangabe.
- Nachschweißungen vor Ort sollten mit derselben Disziplin überprüft werden wie Werkstattschweißungen, wenn sie auf der Produktseite verbleiben.
Passivierung sollte als kontrollierter Fertigungsschritt behandelt werden
Passivierung ist am nützlichsten, wenn sie als Teil eines definierten Fertigungs- und Veredelungsprozesses behandelt wird. Sie sollte nicht auf einen Marketingbegriff reduziert werden. ASTM A967 behandelt mehrere chemische Passivierungsbehandlungen für Edelstahlteile, während ASTM A380 die Reinigung, Entzunderung und Passivierung neuer Edelstahlteile, Baugruppen, Ausrüstungen und installierter Systeme adressiert. In Kosmetik- und Pflegeproduktleitungen liefert Passivierung nur dann zuverlässigen Wert, wenn die vorangehenden Fertigungs-, Vorreinigungs- und Oberflächenvorbereitungsschritte bereits kontrolliert sind.
ASTM A380 macht auch einen wichtigen Konstruktionspunkt, der oft bei der Beschaffung übersehen wird: Edelstahlsysteme sollten so gestaltet sein, dass Bereiche minimiert werden, in denen Schmutz oder Reinigungslösungen eingeschlossen werden können, und eine effektive Zirkulation und Entfernung von Reinigungslösungen ermöglichen. Dieses Prinzip unterstützt direkt hygienische Entleerbarkeitsprüfungen in Kosmetik- und Pflegeproduktrohrleitungen.
Für Reinigungszyklusplanung und reinigungsbezogene Risiken sollte dieses Thema auch zusammen mit Reinigungsaspekte in hygienischen Prozesssystemen, denn selbst eine gut ausgewählte 316L-Leitung kann unterdurchschnittlich abschneiden, wenn die Reinigungsabdeckung, die Chemikalienauswahl, die Spülverifizierung oder das Zyklusdesign nicht angemessen sind.
| Fertigung / Oberflächenfaktor | Warum es im hygienischen Einsatz wichtig ist | Praktische Käuferbedenken |
|---|---|---|
| Innere Oberflächenbeschaffenheit | Beeinflusst die Rückstandsabgabe und Reinigungseffizienz | Sollte definiert, nicht angenommen werden |
| Schweißausführungsqualität | Beeinflusst die Reinigungsfähigkeit und lokale Korrosionsempfindlichkeit | Kritisch in produktberührenden Abschnitten |
| Nachschweißbehandlung | Hilft, den Zustand der produktberührenden Oberfläche zu stabilisieren | Benötigt klaren Fertigungsbereich |
| Passivierungskontrolle | Unterstützt den endgültigen Oberflächenzustand bei korrekter Prozessführung | Sollte als Teil der Endbearbeitung spezifiziert werden, nicht als vage Verkaufssprache |
Tipp:
Wenn ein Lieferant nur bestätigt, dass “Material = 316L” ist, aber die produktberührende Oberflächenbeschaffenheit, Schweißqualitätserwartungen, Handhabung von Feldreparaturen und Nachfertigungsbehandlung nicht erklären kann, ist die Überprüfung noch nicht abgeschlossen.
Wann 316L sinnvoll ist und wann 304 möglicherweise noch ausreicht
Geeignete Anwendungen für 316L in Personal Care- und Kosmetikleitungen
316L ist in der Regel die sinnvollste Wahl, wenn hohe Anforderungen an Hygiene, häufige Reinigung und lange Lebensdauer bestehen. Dies trifft oft auf hochwertige Körperpflegeprodukte, Produktionslinien mit häufigen Produktwechseln, produktberührende Kreisläufe, die lange feucht bleiben, und Systeme zu, bei denen ungeplante Wartung oder Kontaminationsrisiken betrieblich kostspielig sind. Es ist auch eine vernünftige Wahl, wenn die Anlage nach Inbetriebnahme nur begrenzte Toleranz für Nacharbeit hat, da lokale Korrosionsbefunde an hygienischen produktberührenden Rohrleitungen nach der Qualifizierung und Inbetriebnahme des Systems stets störender zu beheben sind.
Situationen, in denen 304 noch in Betracht gezogen werden kann
304 kann unter ausgewählten Bedingungen mit geringerem Risiko noch geeignet sein, jedoch nur, wenn das Expositions- und Reinigungsprofil sorgfältig geprüft wurde. Es ist keine gute Ingenieurpraxis, nur aufgrund der Anfangskosten auf eine niedrigere Qualität zu setzen, wenn die Leitung wiederholter Reinigung, Feuchtigkeitsretention oder anspruchsvoller Produktchemie ausgesetzt ist. In tatsächlichen Projektprüfungen bleibt 304 tendenziell besser vertretbar bei milderen Expositionsbedingungen, in Abschnitten mit geringeren Konsequenzen oder dort, wo die tatsächlichen Risikotreiber durch die Anlagenplanung eliminiert wurden, anstatt sie dem Material zu überlassen.
| Zustand | Unterstützt es die Rechtfertigung für 316L? | Technischer Grund |
|---|---|---|
| Häufige Produktwechsel mit aggressivem Reinigungsplan | Ja, oft stark | Höhere Reinigungslast erhöht den Wert des zusätzlichen Materialspiels |
| Produktberührende Abschnitte mit schwer zu reinigender Geometrie | Ja, aber das Design muss noch korrigiert werden | 316L hilft, kann aber allein ein schlechtes hygienisches Design nicht ausgleichen |
| Geringe Exposition, geringere Reinigungslast, einfachere Produktfamilie | Möglicherweise nicht immer | 304 kann geprüft werden, wenn das Risikoprofil deutlich niedriger ist |
| Nicht produktberührender oder geringerfolgenbereich | Oft schwächerer Treiber | Andere Materialien können je nach Anwendung akzeptabel sein |
Expertenmeinung:
Überspezifizierung von Material kann ein wichtigeres Problem verdecken. Wenn die Leitung schlechte Entwässerung, unnötige Toträume, unkontrollierte Nachschweißungen oder schwache Dichtungsdisziplin aufweist, wird ein Upgrade auf 316L allein kein hygienisches System schaffen.
Was Käufer und Ingenieure bei der Bestellung von 316L-Hygienikkomponenten spezifizieren sollten
Materialgüte allein ist keine vollständige Spezifikation
Die Angabe “316L Hygienisch-Fittings” in einer Anfrage reicht nicht aus, um die hygienische Leistung zu steuern. Einkäufer sollten definieren, welche Teile produktberührend sind, welcher Oberflächengrad erwartet wird, wie geschweißte Baugruppen geliefert werden sollen, welche Dokumentation für die Verifizierung erforderlich ist und welche Dichtungs- oder Dichtmaterialien für das tatsächliche Produkt und das Reinigungsregime zugelassen sind. Einige der am leichtesten vermeidbaren Probleme im Feld entstehen durch unvollständige Einkaufsbedingungen und nicht durch offensichtlich falsche Materialauswahl.
Wichtige Anfrage- und technische Überprüfungspunkte
| Anfrage-/Überprüfungspunkt | Warum es definiert werden sollte |
|---|---|
| Produktberührender Materialumfang | Klärt, welche benetzten Teile die 316L-Anforderung erfüllen müssen |
| Oberflächengüteerwartung | Unterstützt hygienische Reinigbarkeit und Wechselkonsistenz |
| Schweißmontageanforderungen | Reduziert Unsicherheiten bei der Produktkontakt-Fertigungsqualität |
| Nachbearbeitung nach der Fertigung | Definiert, ob Passivierung oder andere Endbearbeitungsschritte erwartet werden |
| Rückverfolgbarkeit und Zertifikate | Hilft Käufern zu überprüfen, dass das gelieferte Material der genehmigten Spezifikation entspricht |
| Dichtungs- / Dichtungskompatibilität | Materialwahl allein gewährleistet nicht die hygienische Verbindungsleistung |
| Entwässerungsfähigkeit und Geometrieüberprüfung | Verhindert, dass Materialverbesserungen durch schlechte hygienische Anordnung verschwendet werden |
Fragen, die Beschaffungsteams vor der Genehmigung stellen sollten
- Welches Reinigungsregime wird dieser Abschnitt im täglichen Betrieb tatsächlich erfahren, einschließlich Haltezeiten und Abschaltbedingungen?
- Handelt es sich um eine echte Produktkontaktzone oder nur um eine angrenzende Zone zum Prozess?
- Sind die Anforderungen an Oberfläche und Schweißnähte klar genug für einen hygienischen Lieferanten definiert?
- Könnte eine schlechte Geometrie mehr Risiko schaffen als die Legierungsgüte selbst?
- Welche Nachweise zeigen, dass die gelieferten Komponenten dem genehmigten Material- und Oberflächenumfang entsprechen?
- Sind Dichtungsverbindungen, Schmiermittel und alle elastischen Backup-Elemente sowohl mit dem Produkt als auch mit den Reinigungschemikalien kompatibel?
Ein häufiger Beschaffungsfehler besteht darin, 316L-Rohre und -Fittings korrekt zu spezifizieren, während Dichtungsmaterial, Oberflächenbeschaffenheit der Klemmenhardware, Handhabung von Reparaturschweißungen oder Inspektionsannahme zu vage bleiben. Bei der Inbetriebnahme vor Ort können diese Auslassungen mehr Korrekturarbeit verursachen als die Metallgüte selbst.
Praktischer Hinweis für Käufer:
Eine starke hygienische Ausschreibung spezifiziert nicht nur die Stahlgüte. Sie definiert den gelieferten Zustand, den das Werk für Reinigung, Betrieb, Inspektion und langfristige Wartung benötigt.
Ein praktisches Bewertungsrahmenwerk für 316L in Anlagen der Körperpflegeindustrie
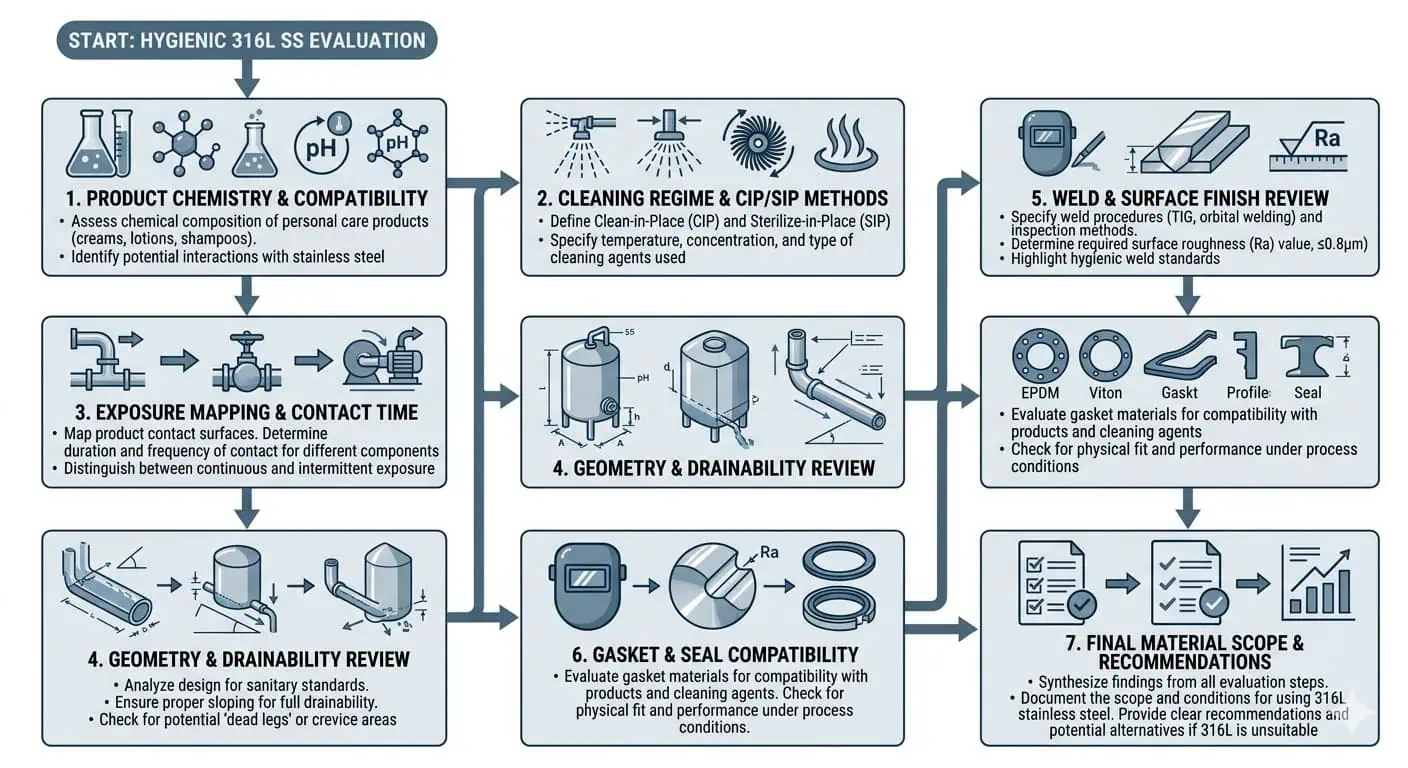
Eine schrittweise Überprüfungssequenz, die Ingenieure tatsächlich nutzen können
- Produktfamilie und tatsächliche Exposition definieren: Viskosität, Rückstände, Feuchtigkeit, Reinigungschemie und Abschaltverhalten überprüfen.
- Hochrisiko-Produktkontaktzonen von niedrigeren Risikobereichen trennen: Nicht alle Leitungsabschnitte benötigen die gleiche Materiallogik.
- Geometrie und Entleerbarkeit überprüfen: Prüfen, ob das System lokale Stagnation, Schattenzonen oder langsam spülende Punkte erzeugt.
- Fertigungsqualitätserwartungen prüfen: insbesondere Oberflächenbeschaffenheit, Schweißausführung und gelieferter Produktkontaktzustand.
- Entscheiden Sie, wo 316L echten Lebenszykluswert hinzufügt: verwenden Sie ihn dort, wo er die Wartungsempfindlichkeit, Hygienerisiko oder Betriebsunsicherheit reduziert.
| Bewertungsschritt | Hauptfrage | Typische Ausgabe |
|---|---|---|
| Expositionsprüfung | Welche chemischen, Feuchtigkeits- und Reinigungsbedingungen wird die Leitung tatsächlich erfahren? | Risikokarte für Materialauswahl |
| Systemzonierung | Welche Teile sind wirklich hochkritische Produktkontaktbereiche? | Priorisierter Materialumfang |
| Geometrieüberprüfung | Kann die Leitung ordnungsgemäß ablaufen, spülen und reinigen? | Designkorrekturen vor Überdimensionierung der Legierung |
| Fertigungsüberprüfung | Entsprechen Oberflächengüte und Schweißqualität den hygienischen Erwartungen? | Lieferanten-/QA-Anforderungsliste |
| Endgültige Materialentscheidung | Wo bringt 316L einen echten Lebenszyklusnutzen? | Ausgewogene Spezifikation anstelle von pauschaler Aufrüstung |
Diese Abfolge funktioniert auch gut bei Retrofit-Audits. Bei den meisten Retrofit-Überprüfungen stellen Ingenieure fest, dass ein großer Teil des zukünftigen Hygienerisikos in der Geometrie, Feldmodifikationen, der Dichtungswahl oder undokumentierter Oberflächenbearbeitung liegt und nicht allein in der Basislegerungsangabe. Genau deshalb sollte die Materialprüfung Teil einer umfassenderen hygienischen Designprüfung sein und nicht eine eigenständige Einkaufsübung.
Häufig gestellte Fragen zu 316L in der Kosmetikproduktion
Ist 316L immer für Kosmetik- oder Körperpflegeproduktionslinien erforderlich?
Nein. 316L wird oft für höherriskante hygienische Produktkontaktdienste bevorzugt, sollte aber nicht als universelle Anforderung für jeden Abschnitt jeder Leitung behandelt werden. Die endgültige Auswahl hängt von Produktchemie, Reinigungsaufgabe, Geometrie, Oberflächenqualität und Lebenszykluserwartungen ab.
Wann gilt 304 noch als akzeptabel?
304 kann weiterhin für niedrigriskante Dienstbedingungen geprüft werden wo Produktexposition milder ist, Reinigungsaufwand geringer ist und die Leitungsgestaltung gut kontrolliert ist. Die Entscheidung sollte auf einer technischen Prüfung basieren, nicht auf einer einfachen Kostensenkung oder einer Gewohnheit, die von einer anderen Prozessleitung übernommen wurde.
Löst 316L alle Korrosions- und Hygienerisiken von selbst?
Nein. Schlechte Entwässerung, schlechte Schweißnahtqualität, spaltanfällige Geometrie, Dichtungsinkompatibilität und schwache Reinigungsleistung können auch dann Betriebsprobleme verursachen, wenn 316L korrekt spezifiziert ist.
Ist die Oberflächenbeschaffenheit wirklich genauso wichtig wie die Materialgüte?
In vielen hygienischen Systemen, ja. Oberflächenbeschaffenheit beeinflusst direkt die Reinigungsfähigkeit, Rückstandsabgabe, Prüfqualität und wie konsistent die Leitung nach Produktwechsel und Reinigung in einen sauberen Betriebszustand zurückkehrt.
Was sollten Käufer neben 316L auf der Bestellung anfordern?
Käufer sollten eine vollständige Definition des gelieferten Zustands anfordern, einschließlich des produktberührenden Bereichs, der Oberflächenerwartung, der Fertigungsqualität, der Nachbehandlungsanforderungen, der Dichtungskompatibilität und der Dokumentation, die zur Überprüfung des tatsächlich gelieferten Materials erforderlich ist.
Was ist der tatsächliche Unterschied zwischen 316L und 304 in Produktionslinien für die Körperpflege?
Der wirkliche Unterschied liegt in der Risikomarge im hygienischen produktberührenden Betrieb. 316L wird häufig dort gewählt, wo wiederholte Reinigung, feuchtere Betriebsbedingungen, stärkere chemische Belastung oder höhere Hygienekonsequenzen die lokale Korrosion und die langfristige Instandhaltung empfindlicher machen. 304 kann in Abschnitten mit geringerem Risiko noch funktionieren, aber 316L bietet normalerweise mehr Betriebsmarge, wo der Prozess härter für das System ist.
Abschließende technische Erkenntnis
In Personal Care- und Kosmetikproduktionsleitungen ist rostfreier Stahl 316L am besten als hygienische Risikokontrollentscheidung zu verstehen – nicht als automatisches Upgrade für jede Komponente. Es schafft in der Regel den größten Wert dort, wo der produktberührende Einsatz, die Reinigungsintensität und die langfristige Wartungsempfindlichkeit hoch sind. Aber das Endergebnis hängt immer noch davon ab, ob das System nach demselben hygienischen Standard entworfen, gefertigt, gereinigt und repariert wird.
Expertenmeinung:
Ein gut gestaltetes hygienisches System mit klarer Entwässerbarkeit, kontrollierter Schweißqualität, realistischer Reinigungsvalidierung und gut spezifizierten Dichtungen funktioniert in der Regel besser als eine schlecht gestaltete Linie, die einfach die Legierungsangabe aufwertet. Die Materialauswahl sollte die hygienische Technik unterstützen – nicht ersetzen.
Wenn Ihr Team prüft, ob 316L Edelstahl für Produktionslinien der Körperpflege ist in einer Lotion-, Shampoo-, Creme-, Gel-, Serum- oder Mehrprodukt-Kosmetiklinie gerechtfertigt, ist der effektivste nächste Schritt, die tatsächlichen Produktkontaktzonen, das Reinigungsregime, die Schweißnahtqualitätserwartungen, die Dichtungskompatibilität und die Systemgeometrie gemeinsam zu bewerten.
Verwandte Lektüre
- Hygienische Rohrleitungsgestaltung für die Kosmetikherstellung
- Hygienefittings vs. Industrielle Rohrverbindungen
- Reinigungsaspekte in Hygienisch-Prozesssystemen
- Edelstahl-Materialleitfaden
- ASME BPE Bioprozessausrüstung
- 3-A Primer für Hygienische Standards und Praktiken
- ASTM A380 Reinigungs-, Entzunderungs-, Beiz- und Passivierungsleitfaden
- ASTM A967 Chemische Passivierungsbehandlungen für Edelstahl




