
High purity piping design for biotech facilities is not just a routing exercise. It is a contamination-control, cleanability, weldability, drainability, and validation decision. In biotech manufacturing, a piping system does more than move fluid from one vessel to another. It has to protect process integrity, support repeatable cleaning and sterilization, minimize microbial and residue risk, remain inspectable after fabrication, and hold up under the documentation and qualification burden of a regulated environment.
That is why good high purity piping design starts with geometry, not with appearance. A system can look hygienic on a P&ID and still fail in operation if the branch layout creates dead legs, the slope does not drain as installed, the weld strategy is unrealistic, or the local valve and sample assemblies are harder to clean than the main line. In biotech facilities, the weakest hygienic detail usually matters more than the cleanest straight run.
This guide explains how engineers design high purity piping for biotech facilities, where 316L and surface-finish decisions genuinely matter, how dead-leg control and drainability shape the layout, why weld design cannot be separated from piping design, and how maintenance and validation expectations should be built into the system before fabrication starts.
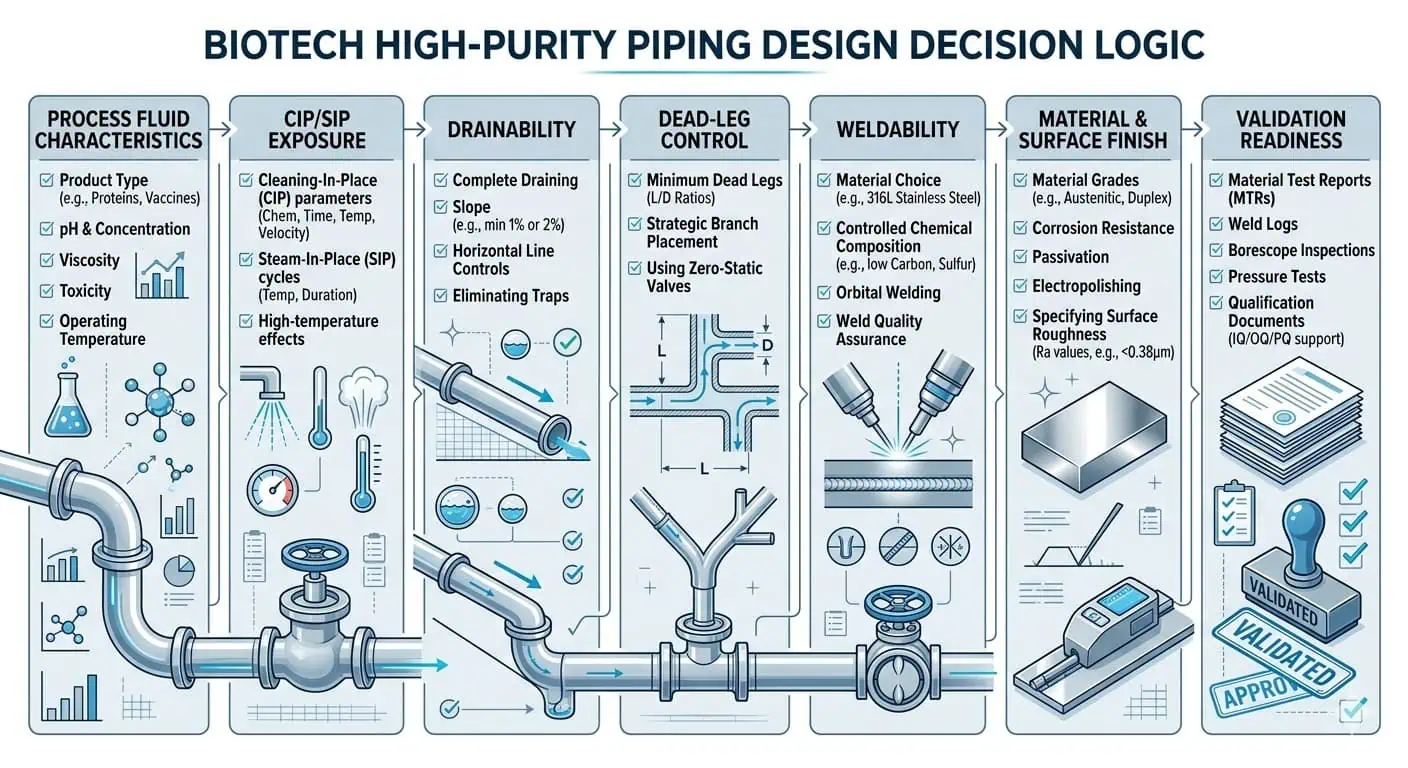
What High Purity Piping Design Really Means in Biotech Facilities
High Purity Design Is a Contamination-Control Decision
In biotech systems, piping design is part of contamination control. ASME BPE is useful here because it does not treat materials, surface requirements, design for cleanability and sterility, fabrication, examination, inspection, testing, and certification as isolated subjects. It treats them as one connected hygienic engineering framework. FDA equipment rules point in the same direction: equipment must be appropriately designed, product-contact surfaces must be non-reactive, non-additive, and non-absorptive, and equipment must be cleanable and maintainable under written control.
Typical engineering reality: the piping loop that creates the most trouble is rarely the main run between vessels. It is more often the sample branch, the instrument tee, the distribution point, or the local manifold that was treated as a minor detail during design. In biotech work, the design question is never just “Will this fluid transfer?” It is “Will this assembly still be cleanable, drainable, inspectable, and defensible after fabrication and validation?”
Consejo experto: if the design review focuses only on throughput, pressure drop, and line routing, it is still incomplete for a high purity biotech system. The design has to be reviewed through the lens of hygienic failure, not only process flow.
Why Biotech Facilities Need More Than General Sanitary Piping Practice
Biotech facilities usually place tighter demands on piping than general sanitary service. The consequence of residue retention, microbial contamination, poor clean steam response, or undocumented field change is higher. In many biotech facilities, the piping system is not judged only by whether it operates safely. It is judged by whether it can support qualification, cleaning verification, maintenance history, change control, and long-term process consistency.
Caso típico de la industria: a layout concept that works acceptably in a general sanitary plant may still be rejected in a biotech project because the local branch arrangement, surface condition, or documentation burden is harder to defend in a validation-focused environment. The issue is not that biotech uses completely different physics. The issue is that the tolerance for ambiguous hygienic behavior is much lower.
A High Purity System Must Perform Hygienically, Not Just Look Hygienic
Sanitary-looking hardware does not automatically create a high purity system. A polished tube, a hygienic ferrule, or a clean-looking weld cap does not prove that the installed system drains properly, that the branch geometry is acceptable, or that the hardest local assembly will clean reliably in place. Biotech piping design succeeds when the local geometry and fabrication practice support the hygienic intent of the specification.
That is the reason this article should be read together with Selección de Conexiones de Grado Alimentario para Sistemas de Fermentación. The wrong local assembly can defeat an otherwise strong system design.
Start with the Real Process: Fluid, Cleaning, and Validation Burden
Process Fluid Characteristics Change the Design
Different biotech fluids create different hygienic design burdens. Buffer solutions, media, product intermediates, harvest streams, WFI-related services, CIP chemicals, SIP steam, and utility-related hygienic fluids do not behave the same way in piping. Some challenge corrosion margin. Some challenge seal compatibility. Some make dead-leg and sample-point design more sensitive because residue behavior or microbial consequence is higher.
Conclusión técnica: good piping design starts with the actual process and cleaning media, not with a default tubing standard. If the design team cannot clearly describe what the line will carry, what temperatures it will see, and how it will be cleaned or sterilized, it is too early to finalize the piping details.
CIP, SIP, and Thermal-Chemical Exposure Are Design Inputs
Cleaning and sterilization are not operating details added later. They are design inputs. FDA 21 CFR 211.67 requires equipment to be cleaned, maintained, and, where appropriate, sanitized and/or sterilized under controlled procedures. In practical piping design, that means the system has to be designed for the real thermal and chemical regime it will see. Clean steam exposure, hot WFI, caustic, acid, sanitizer, cooldown, and repeated thermal cycling all affect what materials, seals, welds, and local fittings will survive reliably.
Caso típico de la industria: a line was sized and routed correctly for process transfer, but repeated SIP exposure later revealed weaknesses at local seal interfaces and hard-to-drain valve orientations. The problem was not that the process flow was wrong. The problem was that the cleaning and sterilization environment had not been treated as part of the original design basis.
Consejo experto: if a local assembly would not survive its real CIP/SIP exposure without special caution, then the problem is design-related even if the main line still looks acceptable.
Validation Burden Changes What “Good Design” Means
In biotech facilities, design is partly judged by how well the system can be validated and maintained under change control. A line that is operable but difficult to inspect, document, or defend during qualification is not a strong high purity design. That is why experienced engineers ask early: where are the highest-risk local assemblies, how will they be inspected, and what evidence will be needed after fabrication and startup?
For the lifecycle side of this logic, connect this article with Consideraciones de Mantenimiento y Validación para Tuberías de Grado Alimentario.
The Core Design Principles That Decide Whether the System Will Succeed
Capacidad de drenaje
True drainability is one of the first design requirements in a high purity piping system. Residual liquid after cleaning or steaming is not just a maintenance nuisance. It changes the hygienic behavior of the next cycle, increases local contamination risk, complicates drying, and makes troubleshooting less reliable. FDA’s equipment Q&A ties equipment design, cleaning, maintenance, and product-contact surface suitability together, which is exactly how drainability should be judged in a biotech loop.
Caso típico de la industria: the design drawing showed a draining line, but after supports were adjusted in the field, one installed span created a subtle low point that repeatedly retained liquid after CIP. The problem did not appear in the main process model. It appeared in the installed geometry.
Consejo experto: never assume drainability from drawing intent alone. Review slope, support spacing, valve body orientation, drain-point location, and final installed behavior together.
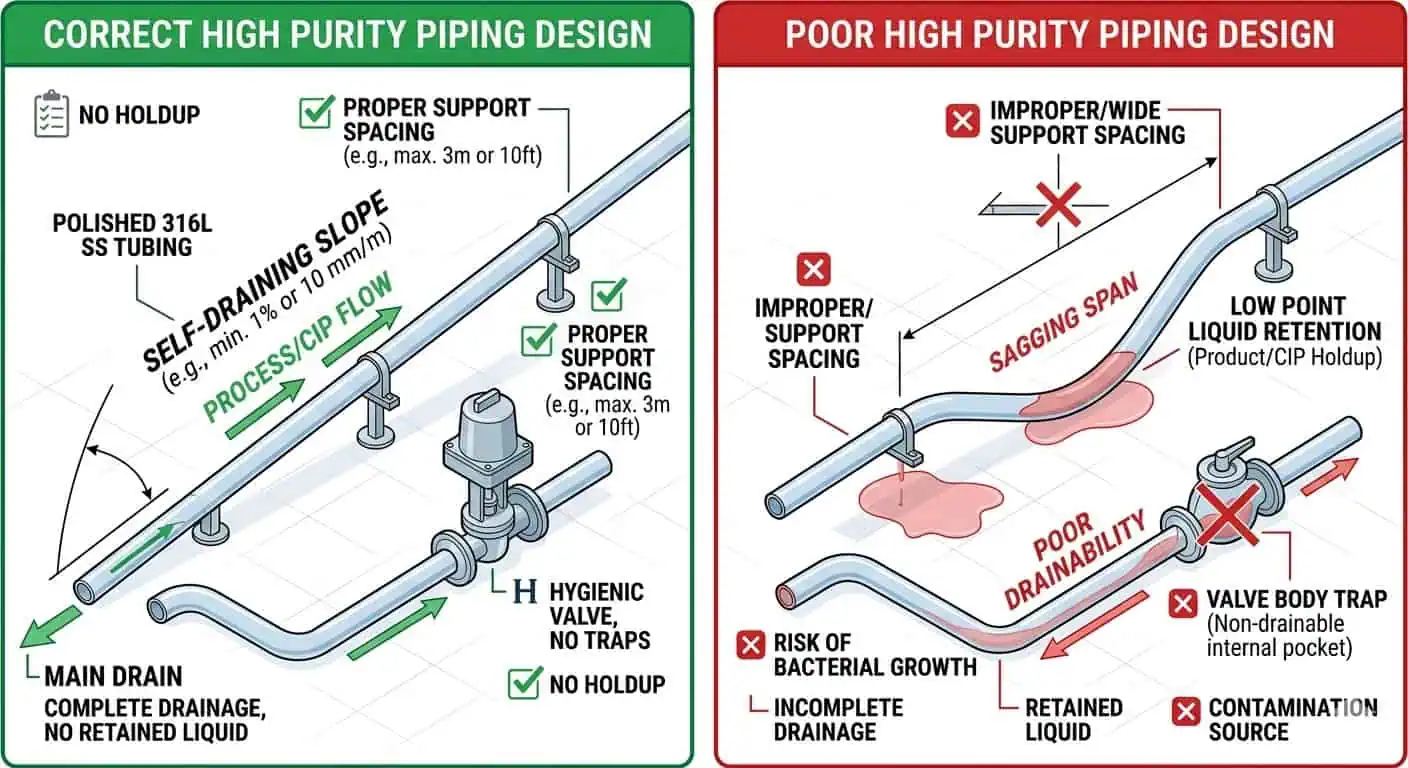
Dead-Leg Control
Dead-leg control remains one of the most important and most underestimated parts of high purity piping design. A line can be made from the correct material, carry the right finish, and still become hard to clean if the branch design creates stagnant zones. Sample take-offs, instrument tees, local manifolds, and added branches are where this problem most often appears.
Caso típico de la industria: the main loop repeatedly met operating and cleaning targets, but one sample branch continued to generate hygienic concern. The root cause was not detergent concentration or cycle time. It was weak local fluid exchange in a branch that had never been critically reviewed as part of the CIP/SIP design.
Consejo experto: evalúe la derivación local más difícil, no la sección recta más fácil. Si un conjunto local no puede intercambiar fluido y drenar de manera confiable, ese es el diseño que importa.
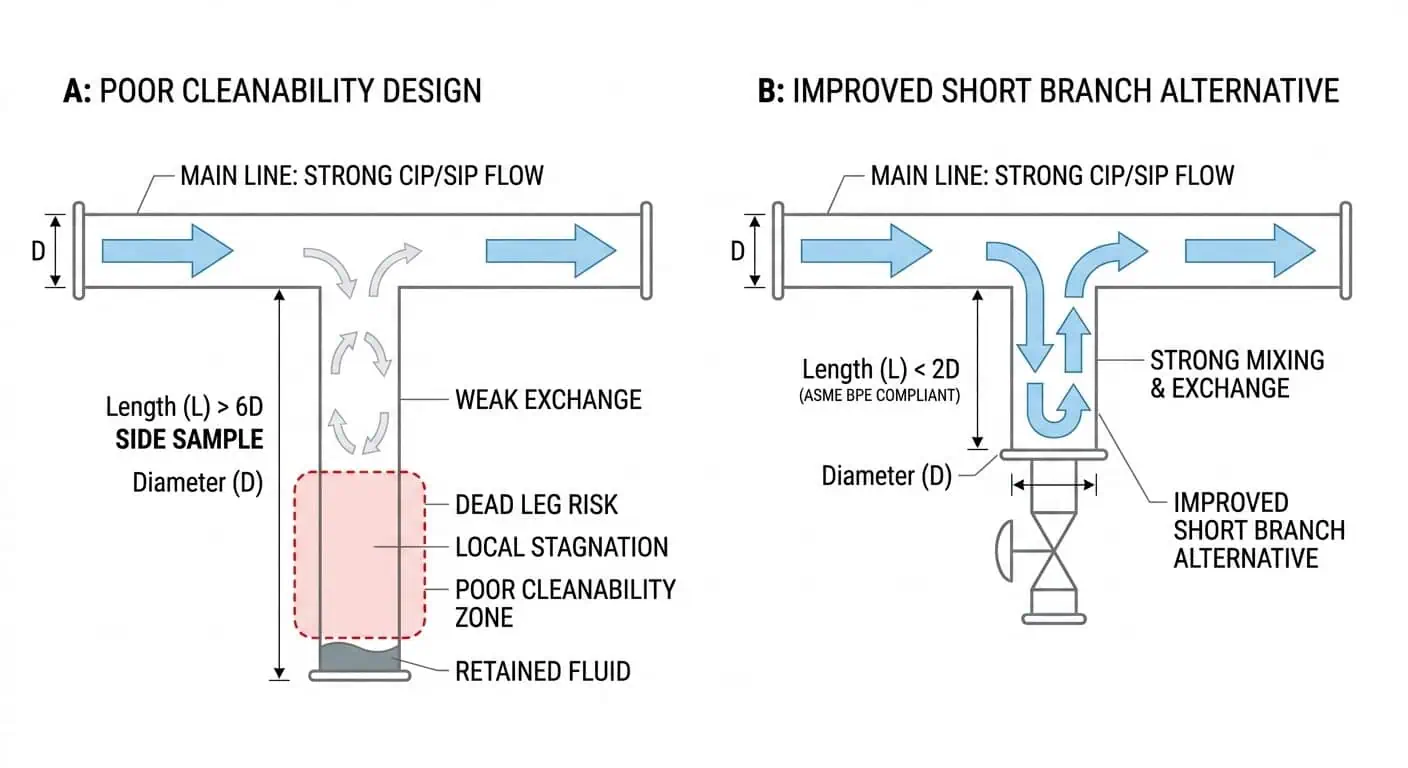
Cleanable Flow Path
Una línea que transfiere bien el producto no se limpia automáticamente bien. Production flow and cleaning flow are related but not identical design problems. In high purity systems, the main process route may be hydraulically acceptable while one local assembly still becomes the weak point because its internal path, orientation, or branch configuration prevents effective cleaning action.
Research-based design logic: ASME BPE’s scope overview directly links surface requirements, design for cleanability and sterility, component manufacture, fabrication including material joining, piping, examinations, inspections, testing, and certifications. That matters because it reinforces a practical engineering truth: cleanability is not a chemical setting alone. It is designed into the wetted path.
Inspection Access and Controlled Intervention
Not every location should be designed for routine opening, and not every location should be permanently closed without good reason. High purity systems need a controlled balance between minimizing interfaces and allowing justified access where seals, diaphragms, or local assemblies will need lifecycle attention. This is one of the most important design tradeoffs in biotech piping, because every added disassembly interface may improve access while also increasing seal-related hygienic risk.
Materials and Surface Requirements in High Purity Biotech Piping
Why 316L Is Commonly the Baseline
316L is commonly used in high purity biotech piping because it provides a practical balance of corrosion margin, weldability, and industry acceptance for critical hygienic service. That does not make it the answer to every process problem, but it does explain why many projects treat it as the baseline metallic material for critical product-contact and high-purity service. ASME BPE covers materials and surface requirements within the broader hygienic design framework, which is exactly how engineers should evaluate 316L in practice.
For the full material-side discussion, connect this article with Acero Inoxidable 316L en Aplicaciones de Bioprocesos.
Surface Finish Is a Hygienic Performance Variable
Surface finish is not a cosmetic choice in biotech piping. It affects local cleanability, inspection confidence, and the practical behavior of product-contact surfaces after fabrication. The key design question is not whether one finish sounds “more premium.” It is whether the chosen finish supports the hygienic target of the process and the inspection expectations of the facility.
Caso típico de la industria: the alloy was acceptable, but the project later revisited the finish requirement because the original surface expectation no longer matched the cleanability and inspection burden of the final process area. The lesson was not that the metal family was wrong. The lesson was that finish had not been defined with enough process-specific discipline.
Fact point: ASTM A270 explicitly states that pharmaceutical quality may be requested as a supplementary requirement, which is a useful reminder that tubing decisions in higher-purity projects often go beyond generic sanitary tubing supply.
Non-Metallic Wetted Parts Matter Too
High purity piping design does not stop at the stainless tube wall. Gaskets, diaphragms, valve seats, hoses, and other non-metallic wetted parts often become the first weak point in cleaning, steam exposure, or lifecycle reliability. In many project deviations, the stainless steel was acceptable, but the local elastomer or polymer strategy was not controlled tightly enough for the real process and sterilization regime.
Consejo experto: if the design team is debating alloy choice but has not defined seal compatibility and replacement logic, the hygienic design review is still incomplete.
Welded Construction, Orbital Welding, and Fabrication Reality
Piping Design Cannot Be Separated from Weld Design
In a high purity biotech system, weldability is part of design, not a fabrication afterthought. The more a system depends on controlled weld quality for hygienic performance, the more unrealistic it becomes to finalize routing and branch logic without considering how the welds will actually be made, purged, inspected, and documented.
Caso típico de ingeniería: the material and route were acceptable on paper, but one local assembly became difficult to defend because the weld access, purge quality, and inspection practicality had not been considered when the branch arrangement was first laid out. The weakness did not begin in fabrication alone. It began in design.
Why Orbital Welding Logic Matters
Orbital weld logic matters in biotech not because it sounds more advanced, but because repeatability, controllability, and documentation matter more in high purity service. In practice, engineers need to think about whether a proposed layout supports reliable weld execution, repeatable purge conditions, and inspection access at the exact locations that will later matter most to QA and maintenance.
Consejo experto: during design review, always ask whether the line is merely drawable or actually weldable under controlled high purity conditions.
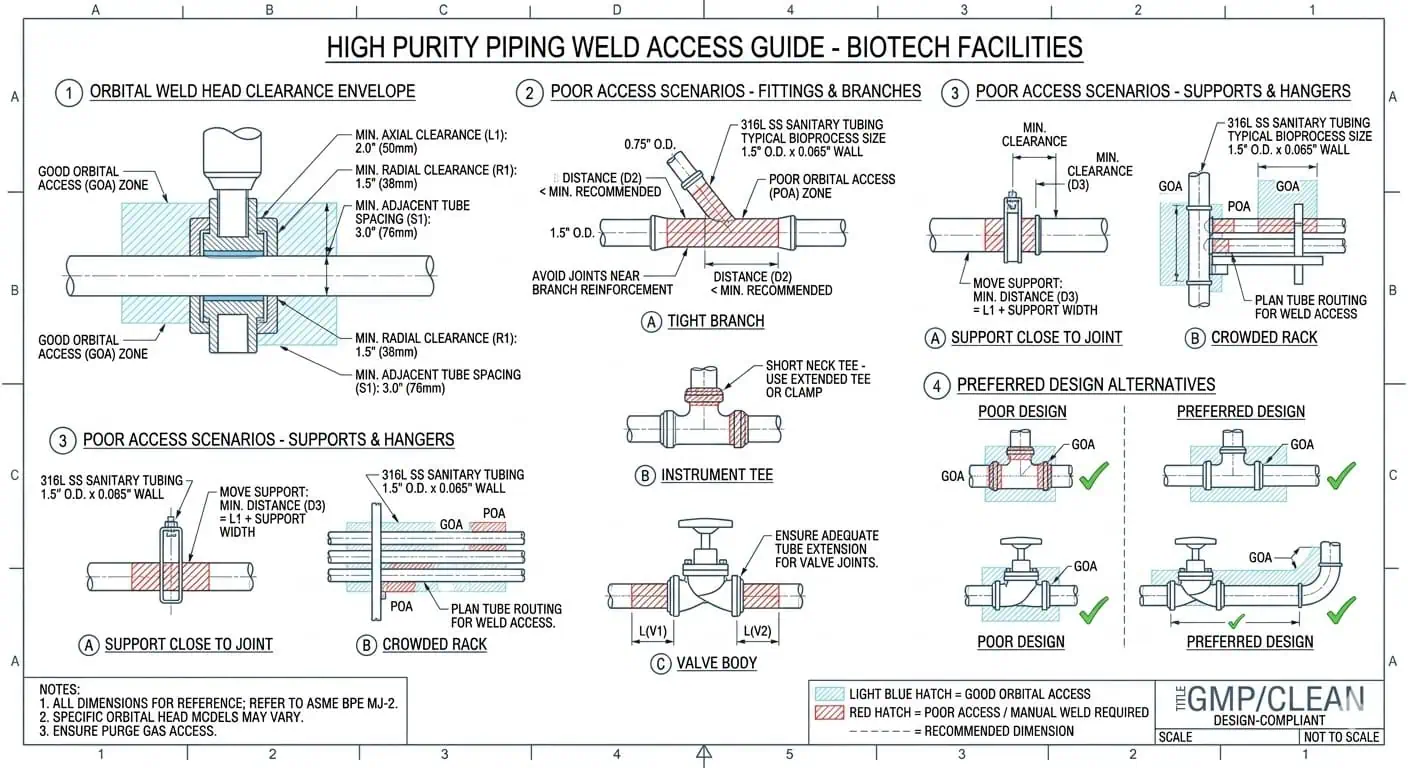
The Base Material Is Often Correct, but the Weld Zone Becomes the Weak Point
One of the most realistic biotech piping failures is a system where the selected material was correct, but the weld-adjacent condition became the hygienic weak point. When that happens, teams sometimes over-focus on material upgrading when the real issue is weld execution, local finish control, or the original assembly geometry. The engineering lesson is straightforward: a correct alloy cannot rescue an inconsistent hygienic weld zone.
Valves, Branches, Sample Points, and Local Assemblies
Sample Point Design
Sample points are small in size but high in consequence. They combine branch geometry, operator interaction, seal count, local fluid-exchange risk, and repeated hygienic scrutiny. This is why sample-point design deserves more engineering attention than its footprint suggests. In many biotech facilities, the most repeated hygienic question is not about the main transfer line. It is about the local sample assembly.
Caso típico de la industria: a main loop performed acceptably, but the same sample location repeatedly triggered additional review because the assembly trapped fluid and never exchanged cleaning media as effectively as the main circuit.
Valve Manifolds and Branch-Dense Areas
Valve clusters and branch-dense manifolds are where good high purity designs often become fragile. The issue is not that valves are bad. The issue is that valves, branches, seals, and instrumentation all multiply local hygienic complexity. As local complexity rises, the design needs stronger control over drainability, weld density, seal interfaces, and inspection logic.
Instrument Tees and Sensor Connections
Instrument tees and sensor fittings are often underestimated because they are small, but they can dominate local cleanability risk. If the design team does not treat them as critical local assemblies, they often become the location where residue retention, local stagnation, or documentation mismatch first becomes visible.
Consejo experto: design the local assembly that is hardest to clean and easiest to overlook. That is usually where the real system risk lives.
Connection Strategy: Welded Versus Clamp-Type Versus Controlled Access
Where More Permanent Welded Connections Are Preferred
More permanent welded connections are often the safer choice where hygienic consequence is high and routine access is not needed. In stable product-contact areas, reducing interface count can reduce seal-related variability and remove one layer of local assembly risk. This does not make welding automatically better everywhere. It means permanent joining often becomes preferable where the location does not benefit enough from repeated opening to justify the extra interface.
Where Clamp-Type Hygienic Access Still Makes Sense
Clamp-fit connections still make sense where controlled access, gasket replacement, or modular change is a real lifecycle requirement. The mistake is not using clamp connections at all. The mistake is placing them in high-consequence locations without acknowledging the added seal-control burden they create.
For the local fitting-side logic, connect this article with Selección de Conexiones de Grado Alimentario para Sistemas de Fermentación.
The Better Question Is Not “Which Connection Is Better?”
The better question is whether the location benefits more from access or from reduced interface count. That is the real engineering tradeoff. A connection style that is excellent in one area can become the wrong answer somewhere else if it introduces unnecessary seal control, extra cleanability burden, or excessive maintenance intervention in a high-purity zone.
Designing for Maintenance and Validation from Day One
A Good Design Is Easier to Maintain
The easiest high purity piping system to maintain is the one whose local hygienic risks were reduced during design instead of delegated to maintenance later. Weak drainability, awkward seal access, badly oriented valves, and difficult-to-inspect local assemblies all become maintenance burdens after startup. They are easier to correct on a drawing than in a live biotech facility.
For the lifecycle side, connect this article with Consideraciones de Mantenimiento y Validación para Tuberías de Grado Alimentario.
Validation Starts in Design
Validation does not begin after installation. It begins when the design team decides what the system must be able to prove. If the line will need strong evidence of cleanability, inspectability, or controlled local geometry, then those requirements must already be visible in the design. Systems that are operable but difficult to validate usually reveal that the design team treated qualification logic too late in the project.
Caso típico de la industria: a piping loop was mechanically successful, but qualification took longer than expected because the hardest-to-clean local assemblies had not been clearly identified during design. The technical fix was minor. The schedule impact was not.
Documentation and Traceability Should Be Anticipated in Design
A high purity design also needs a realistic evidence chain. Material identity, weld records, finish expectations, and field changes become much harder to close if the design never defined how the final system would be documented and released. That is why high purity piping design should also connect to the turnover and traceability logic of the project.
Common Design Mistakes in High Purity Biotech Piping
Designing for Product Flow but Not for Cleaning Flow
A loop may transfer process fluid well and still underperform during cleaning if the local assemblies never exchange cleaning media effectively enough.
Accepting Sanitary-Looking Layouts Without Checking Dead Legs
Appearance does not replace local branch review. Dead-leg behavior often hides in small assemblies, not in the main route.
Over-Focusing on 316L While Ignoring Welds and Geometry
A stronger alloy does not correct poor weld execution, poor drainability, or difficult branch geometry.
Using One Design Logic Across All Process Areas
High-consequence product-contact lines, sample branches, utility-related hygienic services, and lower-risk areas should not all be designed to one undifferentiated standard.
Leaving Validation and Documentation Questions Until Late Project Stages
If the system only begins to think about evidence, traceability, and qualification after fabrication, the project is already operating with less control than it should.
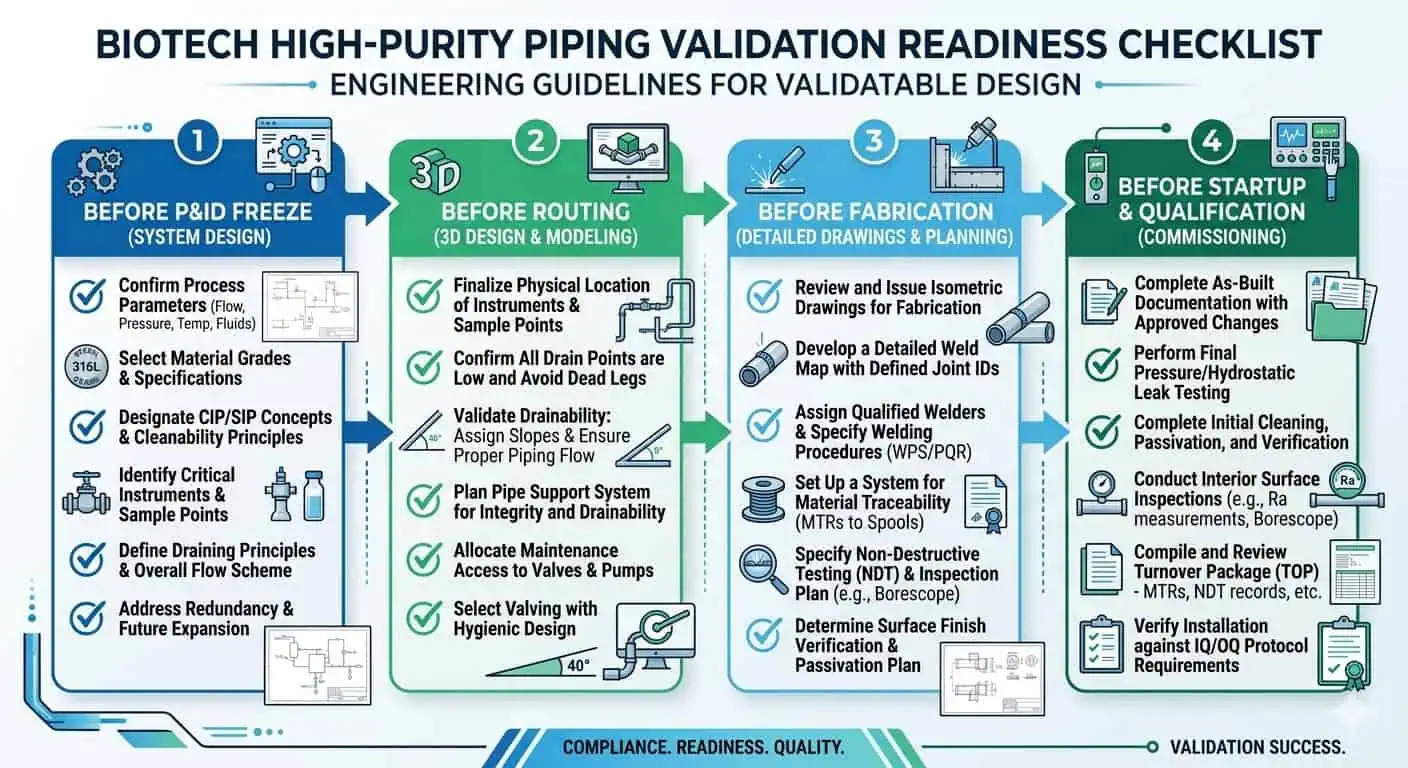
Practical Design Checklist for High Purity Piping in Biotech Facilities
Before P&ID Freeze
- Define the real process and cleaning media.
- Identify the highest-risk local assemblies.
- Decide where the system needs access and where it needs fewer interfaces.
- Define the hygienic and validation burden of each major process area.
Before Detailed Routing
- Review drainability, slope logic, and low-point risk.
- Check branch length, take-off direction, and local dead-leg potential.
- Review valve and sample-point orientations as installed assemblies, not symbols only.
- Confirm weld access and purge practicality at critical locations.
Before Fabrication
- Define material, finish, and seal requirements together.
- Align routing with realistic weld strategy and inspection logic.
- Identify the assemblies that will need tighter hygienic review after fabrication.
- Make sure the documentation chain is compatible with the final design intent.
Before Startup and Qualification
- Confirm the installed loop still drains as intended.
- Review the hardest-to-clean local assemblies, not just the main run.
- Confirm that field changes did not introduce new hygienic weak points.
- Verify that the final system still supports the intended validation and maintenance approach.
| Design Topic | What Good Design Looks Like | Lo que Suele Salir Mal |
|---|---|---|
| Capacidad de drenaje | The installed line drains predictably at local low-risk and high-risk points | The drawing drains, but the installed support geometry does not |
| Control de tramo muerto | Branches are reviewed as local hygienic assemblies | Sample points and instrument tees are treated as minor details |
| Weld strategy | Routing supports realistic purge, access, and inspection | Design assumes weld quality without checking fabrication practicality |
| Connection logic | Access and interface count are balanced by process need | Clamp or welded choices are copied without location-specific review |
| Validation readiness | The hardest assemblies are identifiable and defensible | Qualification logic starts after fabrication instead of during design |
Conclusion: High Purity Piping Design Succeeds When Geometry, Materials, Weldability, and Validation Logic Are Treated as One System
High purity piping design for biotech facilities succeeds when geometry, materials, weldability, cleanability, drainability, and validation logic are designed as one system. The strongest biotech piping projects do not rely on alloy name, surface appearance, or catalog component style alone. They reduce local hygienic risk at the branch, sample point, valve assembly, and weld zone where the process is least tolerant of failure.
If you want a stronger biotech topic cluster, connect this article with Acero Inoxidable 316L en Aplicaciones de Bioprocesos, Selección de Conexiones de Grado Alimentario para Sistemas de Fermentacióny Consideraciones de Mantenimiento y Validación para Tuberías de Grado Alimentario.
PREGUNTAS FRECUENTES
¿Cuál es el factor de diseño más importante en tuberías de biotecnología de alta pureza?
The most important factor is usually cleanable, drainable geometry. If the system does not drain well or creates dead-leg behavior at local assemblies, stronger materials and better-looking finishes will not solve the core hygienic problem.
¿Se requiere siempre 316L en tuberías higiénicas de grado alimentario para biotecnología?
No automáticamente. 316L is a common baseline for critical high purity service, but the final decision still depends on process chemistry, cleaning and sterilization exposure, finish requirement, and the risk profile of the local assembly.
¿Por qué las ramas muertas son un problema tan grande en los sistemas de alta pureza?
Because dead legs weaken local fluid exchange, cleaning effectiveness, and drainability. They are one of the most common reasons a local branch or sample point keeps creating hygienic concern even when the main loop appears acceptable.
¿Cuándo se deben usar conexiones de ajuste por abrazadera en tuberías de biotecnología?
Use them where controlled access, seal replacement, or modular intervention is genuinely needed. They can be the right choice, but every added interface also increases seal-related hygienic risk, so they should not be used by default in every high-purity location.
¿Resuelve el electropulido problemas de soldadura deficiente o de geometría deficiente?
No. A tighter finish can support a higher hygienic target, but it does not fix weak drainability, bad branch geometry, or a poor weld zone.
¿Cómo afecta la validación a las decisiones de diseño de tuberías?
Validation changes what the system must be able to prove, not just how it must run. A piping design that is operable but difficult to inspect, document, or defend may still be a weak biotech design.




