
Le nettoyage et la maintenance des systèmes de tuyauterie agroalimentaire ne se limitent pas à l'exécution d'un cycle CIP selon un calendrier. Dans les usines alimentaires et de boissons, la performance hygiénique fiable dépend du fait que le système soit véritablement vidangeable, nettoyable, inspectable et entretenu avec les bonnes pièces de rechange et une routine de vérification. La plupart des échecs de nettoyage ne commencent pas au milieu d'un tube droit. Ils commencent au niveau des détails : joints avec joints, zones mortes, raccordements de branchement, corps de vannes, points d'échantillonnage, poches à faible vidange et zones de soudure rugueuses ou teintées par la chaleur.
Si vous entretenez la tuyauterie agroalimentaire uniquement par intervalle de temps, vous manquerez les points de risque réels. Si vous l'entretenez en fonction de la géométrie, de la méthode de nettoyage, de l'état des joints, de la qualité des soudures et des modèles de déviation récurrents, vous pouvez réduire le risque de contamination, réduire les temps d'arrêt imprévus et améliorer la préparation aux audits en même temps. Ce guide explique comment les ingénieurs abordent la maintenance de la tuyauterie agroalimentaire dans les systèmes réels de transformation des aliments, avec des routines pratiques, une logique de conception basée sur des normes, des cas sur le terrain et des conseils d'inspection au niveau des composants.
Ce que signifie vraiment la maintenance de la tuyauterie agroalimentaire
La maintenance ne se limite pas au nettoyage
La maintenance de la tuyauterie agroalimentaire comprend le nettoyage, l'inspection, le remplacement, la vérification et la documentation. Une ligne peut sembler visuellement propre et échouer hygiéniquement si un joint a gonflé, une branche retient de l'eau de rinçage ou un siège de vanne piège des résidus de produit. Dans un service de qualité alimentaire, la maintenance doit répondre à cinq questions à chaque fois : La surface en contact avec le produit a-t-elle été nettoyée ? La zone a-t-elle été entièrement vidangée ? Les assemblages à risque le plus élevé ont-ils été inspectés ? Les pièces endommagées ou incompatibles ont-elles été remplacées ? L'usine peut-elle prouver ce qui a été fait ?
C'est pourquoi un programme de maintenance agroalimentaire ne doit pas être rédigé comme une fiche de maintenance mécanique générale. Il doit être rédigé comme un système de contrôle de l'hygiène pour les équipements en contact avec le produit. La règle de la FDA sur les équipements suit la même logique : les surfaces en contact avec les aliments doivent être suffisamment nettoyables, résistantes à la corrosion et capables de résister à leur environnement alimentaire et de nettoyage prévu. En pratique, cela signifie que la maintenance doit contrôler à la fois la propreté et l'état des composants, et pas seulement l'apparence visible.
Qu'est-ce qui différencie la tuyauterie agroalimentaire de la tuyauterie industrielle standard
La tuyauterie agroalimentaire est entretenue différemment car les critères de défaillance sont différents. Dans la tuyauterie industrielle générale, la principale préoccupation d'entretien peut être l'intégrité mécanique, la tolérance à la corrosion, le confinement de la pression ou les vibrations. Dans la tuyauterie agroalimentaire, vous devez également protéger la nettoyabilité, la vidange, l'intégrité du contact alimentaire et le contrôle des changements. Cela rend les ferrules, les soudures internes, la géométrie des dérivations, les joints, les vannes et les points d'échantillonnage beaucoup plus importants que dans une conduite utilitaire ou de procédé typique.
Si vous avez besoin d'une comparaison côté conception avant de construire la routine d'entretien, lisez raccords agroalimentaires vs raccords industriels. Cela aide à expliquer pourquoi les connexions hygiéniques nécessitent une logique d'inspection et de nettoyage différente de celle des composants industriels généraux.
Pourquoi la défaillance de maintenance commence généralement par les détails, et non par le tube principal
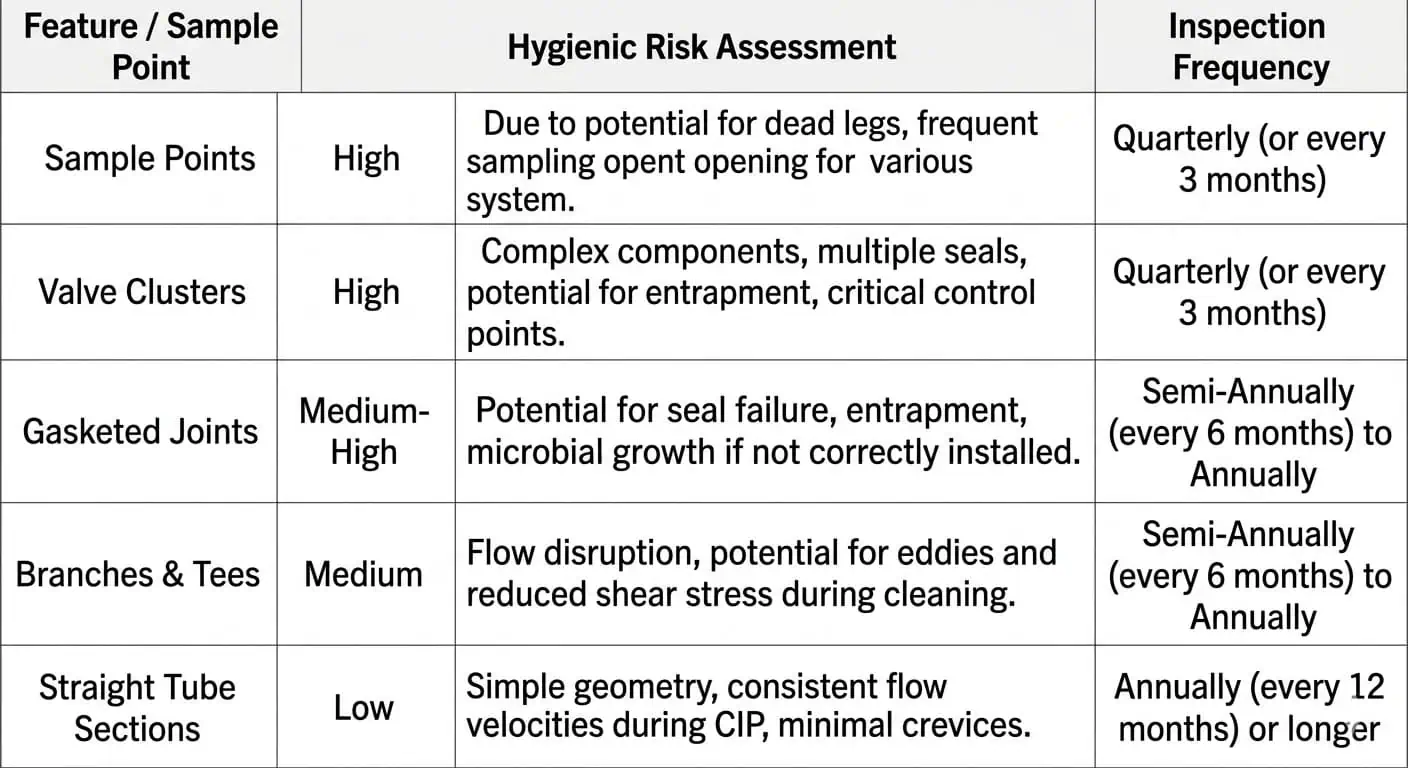
Les points de maintenance à risque le plus élevé dans une ligne hygiénique ne sont généralement pas les tubes droits. Ce sont les endroits où le produit ralentit, où la couverture de nettoyage en place (CIP) diminue, où les joints vieillissent ou où la qualité de fabrication affecte directement la nettoyabilité. En pratique, les points chauds de maintenance les plus courants sont :
- Les joints à bride et les faces de ferrule
- Les joints et les sièges de vanne
- Branches en impasse et tés d'instrumentation
- Soupapes d'échantillonnage et clapets anti-retour
- Prélèvements horizontaux et points bas
- Racines de soudure, zones de décoloration de soudure et transitions internes rugueuses
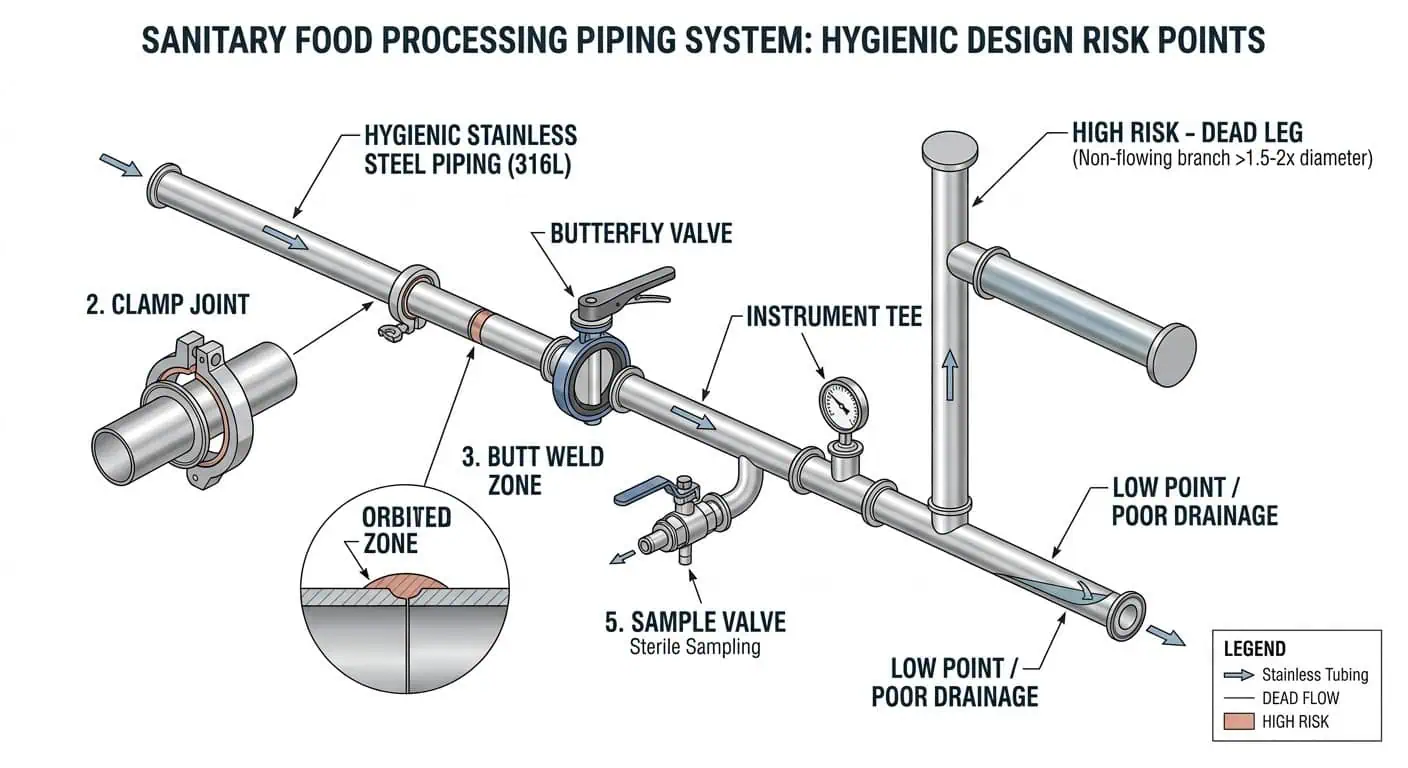
C'est pourquoi un système de tuyauterie agroalimentaire ne doit jamais être inspecté avec un état d'esprit de “ toute la ligne est correcte ”. La ligne n'est aussi hygiénique que son détail de contact avec le produit le plus problématique.
Commencez par la méthode de nettoyage : CIP, COP ou démontage manuel
Quand le CIP est le bon choix
Le nettoyage en place est le bon choix lorsque le système est fermé, reproductible et conçu pour assurer une couverture complète, un drainage et un contact chimique. Le CIP est le plus efficace dans les grands systèmes de liquides alimentaires, les campagnes de production répétées et les lignes où l'ouverture manuelle ajouterait un risque de contamination ou des temps d'arrêt excessifs. Il est particulièrement utile pour les systèmes laitiers, de boissons et de transfert d'ingrédients avec une conception hygiénique stable et des paramètres de nettoyage validés.
Le nettoyage en place (CIP) ne fonctionne que dans la mesure où la géométrie du système le permet. Un programme chimique puissant ne peut pas compenser entièrement les espaces morts, les pentes insuffisantes, les joints piégés ou les dérivations à faible débit qui ne bénéficient jamais d'un mouillage et d'un cisaillement appropriés. C'est là que de nombreuses usines surestiment ce que signifie automatiquement “ agroalimentaire ”. Les directives de nettoyabilité 3-A soulignent clairement : l'équipement présenté comme compatible CIP n'est pas toujours véritablement adapté à un CIP complet, à moins que la nettoyabilité n'ait été intégrée dès la conception. Sur le terrain, cela signifie que le tableau de retour peut sembler acceptable alors qu'une dérivation mal placée continue d'échouer à la vérification.
Quand le nettoyage hors place (COP) ou le nettoyage manuel reste nécessaire
Le nettoyage hors place ou le démontage manuel sont encore nécessaires lorsque l'équipement comprend des pièces qui ne peuvent pas être nettoyées de manière fiable en place. Les petits composants amovibles, les internes complexes des vannes, les points d'échantillonnage spéciaux et les assemblages fréquemment ouverts nécessitent souvent une inspection et un nettoyage manuels pour maîtriser le risque hygiénique. Dans certains systèmes, le CIP peut bien nettoyer la ligne principale, mais pas les pièces amovibles ou à faible débit qui y sont attachées.
Un programme de maintenance pratique ne doit pas considérer le nettoyage manuel comme un échec de conception. Il doit le considérer comme la méthode de contrôle appropriée lorsque la géométrie ou les conditions de service l'exigent. Une erreur de maintenance répétée consiste à supposer qu'un dispositif de pulvérisation ou une chimie en circulation rend automatiquement un assemblage entièrement nettoyable. Sur les lignes réelles, la meilleure question est de savoir si la surface en contact avec le produit la plus difficile à nettoyer peut réellement être atteinte, drainée et vérifiée sans suppositions.
La méthode de nettoyage doit correspondre à la géométrie de l'équipement
La bonne méthode de nettoyage dépend autant de la géométrie que de la chimie. Si une dérivation est trop longue, un corps de vanne retient du liquide après le rinçage, ou un point d'échantillonnage présente une mauvaise circulation, le plan de maintenance doit en tenir compte. Les systèmes hygiéniques ne sont pas “ adaptés au CIP ” simplement parce qu'ils sont en acier inoxydable ou parce qu'ils utilisent une connexion de type agroalimentaire. Les ingénieurs de maintenance doivent toujours se demander :
- L'assemblage se vide-t-il complètement après le cycle ?
- Le fluide de nettoyage atteint-il réellement la zone à risque le plus élevé ?
- Le modèle d'écoulement est-il suffisamment puissant pour éliminer les résidus ?
- Le composant peut-il être vérifié sans démontage inutile ?
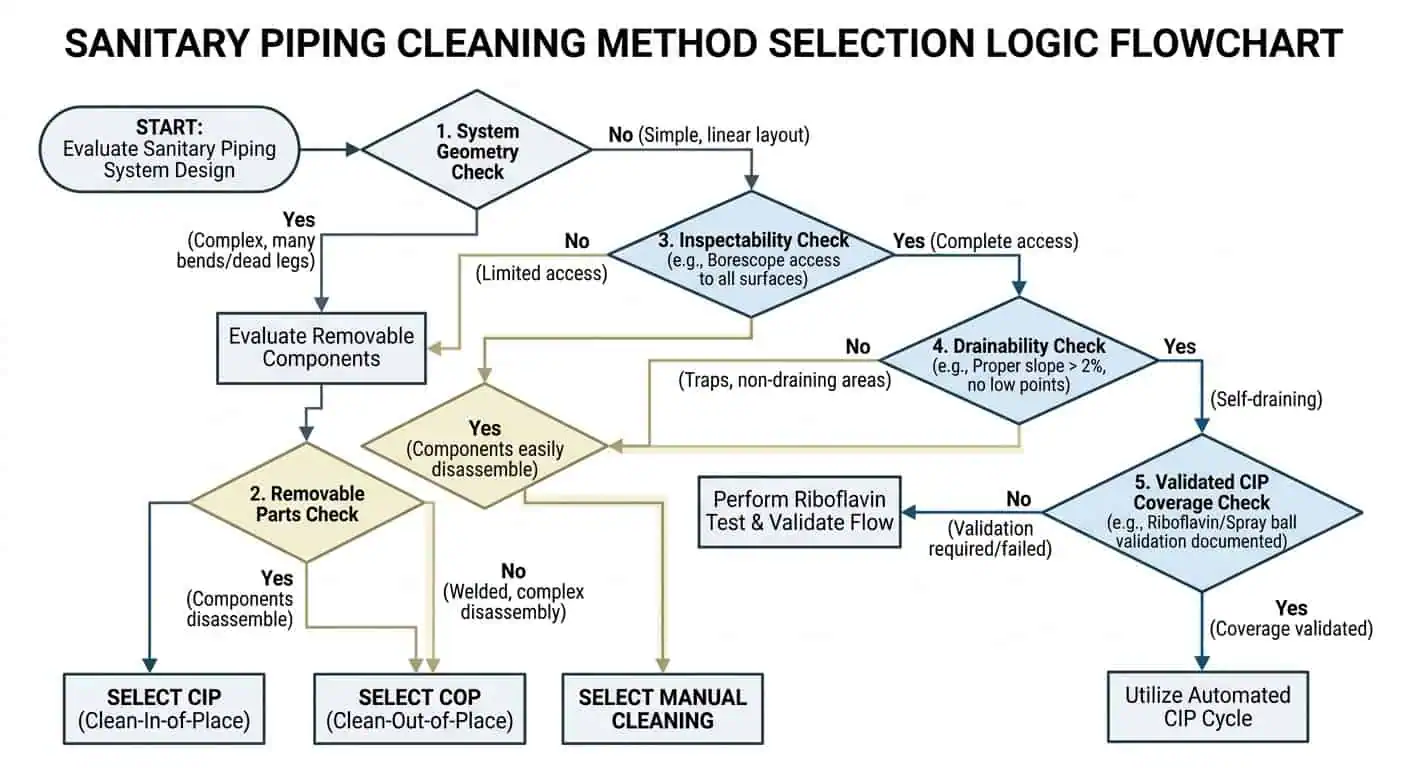
Cette série de questions empêche l'une des erreurs de maintenance les plus courantes dans les usines agroalimentaires : supposer que l'ensemble du système est nettoyé parce que les principaux paramètres de retour semblaient acceptables.
Les Véritables Priorités de Maintenance dans les Systèmes de Tuyauterie Agroalimentaire
Vidange et Contrôle des Points Bas
Un mauvais drainage est l'un des moyens les plus rapides de transformer une ligne nettoyable en un risque hygiénique récurrent. L'eau de rinçage résiduelle, le talon de produit ou la solution de nettoyage diluée laissés dans un point bas peuvent favoriser la croissance microbienne, créer des transferts et fausser les performances de nettoyage ultérieures. Dans les travaux de maintenance, les points bas méritent une attention particulière après toute modification, réparation ou ajustement de support, car la pente et la vidange peuvent changer plus facilement que de nombreuses équipes ne le pensent.
Emplacements typiques à haut risque :
- Points bas entre les supports
- Corps de vannes qui ne se vident pas complètement
- Branches horizontales et prises d'instrumentation
- Connexions de capteurs en impasse
- Points d'échantillonnage mal orientés
Lorsqu'une ligne échoue de manière répétée aux contrôles d'hygiène même si le temps et la chimie de nettoyage en place semblent normaux, un mauvais drainage est l'une des premières choses à vérifier sur le terrain. Les directives EHEDG sont utiles ici car elles traitent la géométrie difficile à nettoyer comme un problème de conception hygiénique, et pas seulement comme un problème d'assainissement. Pour les ingénieurs de maintenance, cela signifie qu'un mauvais drainage doit généralement être corrigé physiquement plutôt que d'être géré uniquement en augmentant la concentration chimique ou en prolongeant le temps de cycle.
Impasses, branches et zones stagnantes
Les impasses augmentent la charge de maintenance car elles réduisent l'efficacité du nettoyage et rendent la vérification plus difficile. Un problème d'impasse n'est pas limité à la conception initiale de l'usine. Il peut être introduit ultérieurement par l'ajout d'instruments, de prises d'échantillons, de raccordements temporaires ou de tés mal placés lors de modifications. Les équipes de maintenance doivent examiner les nouvelles branches avec la même discipline hygiénique utilisée lors de la fabrication initiale, car même une petite branche peut créer un problème de nettoyage répété si elle piège le produit ou l'eau de rinçage.
Dans les opérations quotidiennes, les défaillances liées aux impasses apparaissent souvent comme :
- Résidu persistant dans une branche après un CIP par ailleurs acceptable
- Positifs ATP ou microbiologiques provenant d'un port ou d'un assemblage spécifique
- Rétention d'odeur à un emplacement répété
- Transfert lors du changement de produit à partir de vannes d'échantillonnage ou de branches à faible débit
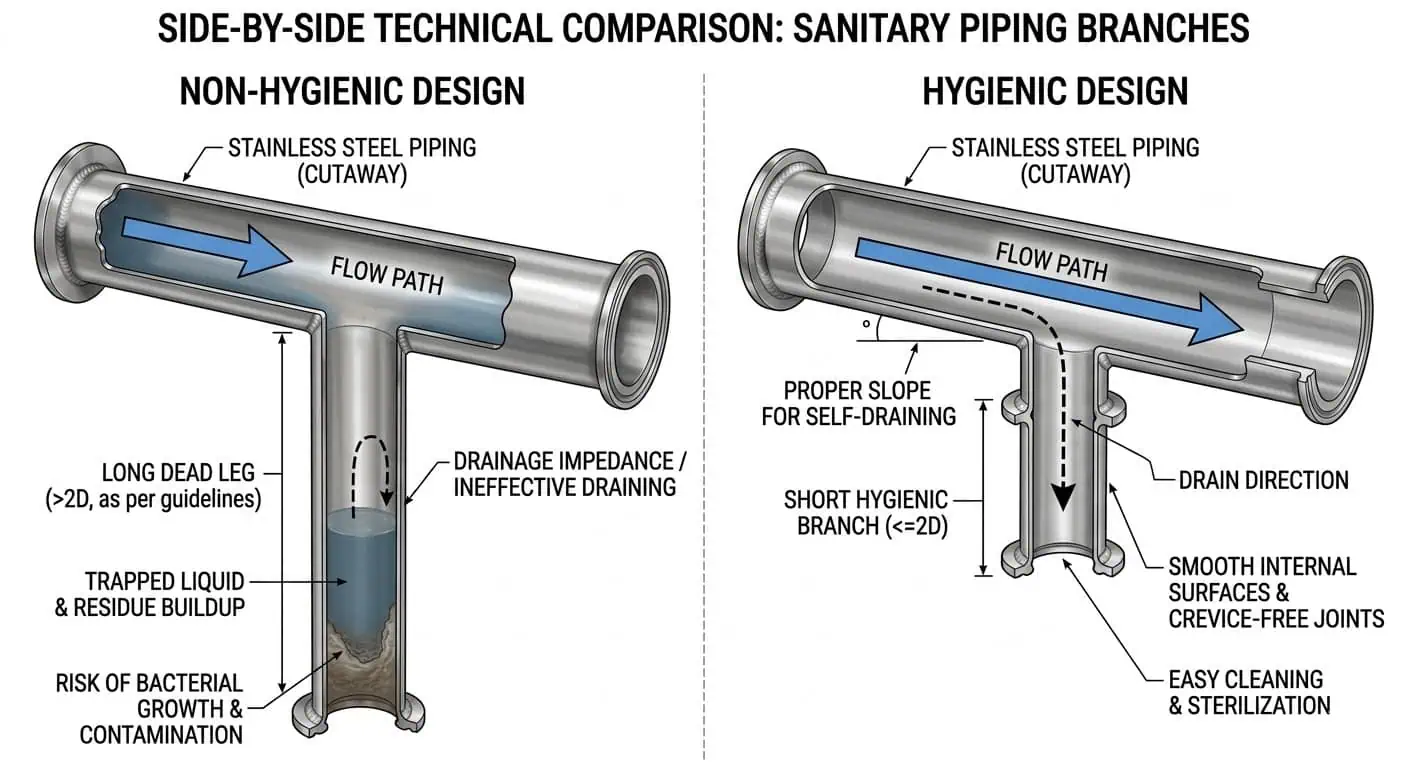
Cela devient particulièrement évident au niveau des branches d'échantillonnage. Sur plus d'une ligne laitière, la boucle CIP principale semblait acceptable du côté retour, mais des résidus continuaient d'apparaître dans le même assemblage d'échantillonnage. Le problème n'était pas la force du détergent. C'était la géométrie de la branche et le faible débit local. Une fois la branche raccourcie, réorientée ou retirée de la logique supposée CIP uniquement, le résultat du nettoyage est devenu beaucoup plus stable.
État de la soudure et finition de surface
La qualité de soudure affecte directement la nettoyabilité, pas seulement l'apparence. La rugosité interne de la soudure, un mauvais contour, l'oxydation de la soudure et les transitions locales de type fissure peuvent tous augmenter la rétention de résidus et rendre l'assainissement moins fiable. Dans un service hygiénique, une mauvaise soudure interne est un problème de maintenance dès le premier jour d'exploitation, car elle augmente la fréquence à laquelle la ligne nécessite une attention et la difficulté à vérifier l'état de propreté.
La finition de surface est importante pour la même raison. Une surface de contact avec le produit plus lisse et correctement finie est plus facile à nettoyer et moins susceptible de retenir un film de produit. Mais la finition seule ne suffit pas. Si la soudure interne est irrégulière ou si la transition du manchon est en escalier, la ligne peut toujours se comporter comme un système difficile à nettoyer, même lorsque la finition du métal de base semble acceptable sur le papier. En pratique, c'est pourquoi une mise à niveau de matériau seule déçoit parfois : une ligne peut passer à l'acier 316L et continuer à présenter des écarts ATP si le vrai problème est le profil de soudure, la coloration thermique ou un mauvais drainage autour de la zone de soudure.
Jointures, Joints et Assemblages à Colliers
De nombreuses défaillances de systèmes agroalimentaires commencent au niveau du joint bien avant d'apparaître comme un problème majeur de ligne. Un joint peut gonfler, s'aplatir, se fissurer, retenir des odeurs ou perdre sa résilience en raison d'une exposition chimique, d'un historique de température ou d'une surcompression répétée. Lorsque cela se produit, la ligne peut présenter des fuites précoces, un resserrage répété, un film de produit au bord du manchon ou une instabilité de nettoyage après les changements de production.
Lors d'une inspection de routine, recherchez :
- Gonflement ou ramollissement après exposition aux produits chimiques de nettoyage en place (CIP)
- Coupures, éraflures ou déchirures au bord du joint
- Aplatissement ou perte de récupération élastique
- Produit piégé ou rétention d'odeur
- Signes que le joint a été forcé en alignement par la pression de la bride
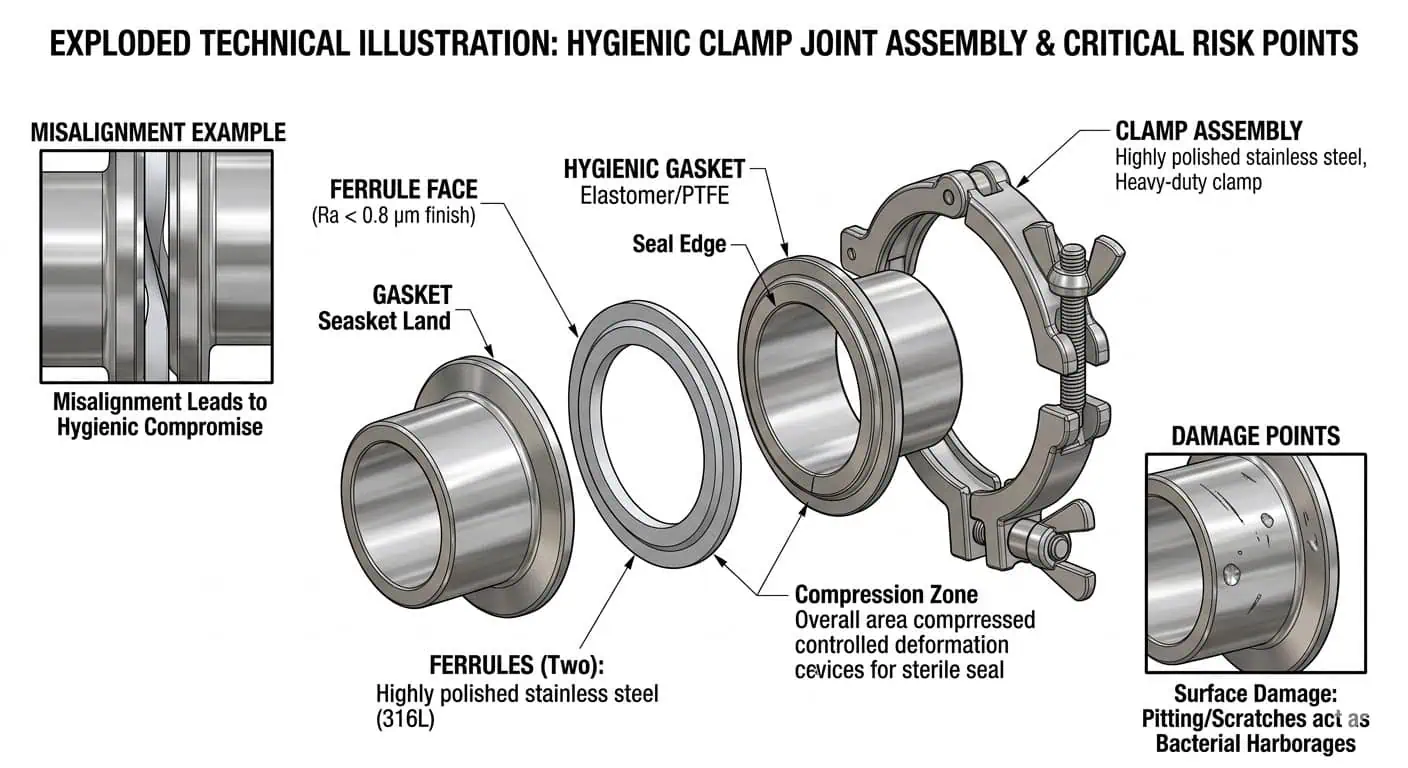
En pratique, un joint à bride qui continue de fuir n'est souvent pas un problème de couple. C'est plus souvent un problème d'alignement, un problème de composé de joint ou un problème de contrôle de compression répété. Sur les lignes de boissons et de produits laitiers, une constatation courante sur le terrain est que les opérateurs continuent de serrer le même joint jusqu'à ce que la fuite visible s'arrête, mais les viroles étaient légèrement désalignées dès le départ. Le résultat est un joint chargé de manière inégale, une durée de vie du joint d'étanchéité réduite et un joint qui devient plus difficile à nettoyer en interne même lorsqu'il ne goutte plus en externe. Un joint sec n'est pas automatiquement un joint correct sur le plan hygiénique. Si des fuites répétées de la bride sont liées à la géométrie des viroles, à la compression du joint ou à des détails de connexion difficiles à nettoyer, consultez notre guide pour le choix de la connexion par rapport à comment choisir des raccords hygiéniques pour l'agroalimentaire.
Vannes, Points de Prélèvement et Autres Ensembles à Haut Risque
Les vannes et les points de prélèvement doivent être traités comme des ensembles nécessitant plus d'entretien que les tuyaux droits. Les vannes papillon, les vannes à membrane, les vannes d'échantillonnage, les zones à double étanchéité et les clapets anti-retour contiennent tous plus de détails internes, plus de zones d'ombre ou plus de surfaces d'étanchéité qu'une section droite de tuyauterie. Ces caractéristiques les rendent essentielles pour le contrôle des processus, mais elles les rendent également plus sensibles à la méthode de nettoyage, au comportement du produit et à la discipline d'inspection.
Dans la planification de la maintenance basée sur les risques, ces ensembles méritent un examen plus fréquent que les simples tronçons de tuyauterie car ils sont plus susceptibles de devenir le premier point faible hygiénique. Pour les ingénieurs de maintenance, la question la plus utile n'est pas “La ligne entière a-t-elle été nettoyée ?” mais “Quel ensemble sur cette ligne est le plus difficile à nettoyer et le plus facile à négliger ?”
Routines de Maintenance Quotidiennes, Hebdomadaires et Périodiques
Contrôles Quotidiens
Les contrôles agroalimentaires quotidiens doivent se concentrer sur les indicateurs de risque visibles et les points de problème récurrents. Une bonne pratique quotidienne inclut une vérification rapide de :
- Fuites ou suintements visibles aux joints de serrage
- Résidus autour des viroles, des sorties de vannes ou des points d'échantillonnage
- Mauvais drainage après le nettoyage ou le changement de produit
- Odeur inhabituelle ou entraînement dans les assemblages en contact avec le produit
- Signes externes d'entrée d'eau de lavage ou d'éclaboussures dans des zones qui doivent rester contrôlées
D'un point de vue d'inspection, les contrôles quotidiens fonctionnent mieux lorsqu'ils ne sont pas répartis uniformément sur chaque composant. L'objectif est de vérifier les assemblages qui créent historiquement le plus d'instabilité hygiénique.
Contrôles hebdomadaires
Les contrôles hebdomadaires doivent confirmer l'état des composants contrôlés et des assemblages qui vieillissent plus vite que les tuyaux droits. Les éléments typiques de l'examen hebdomadaire incluent l'état des joints, les emplacements de fuites récurrents, le fonctionnement des vannes, la propreté des points d'échantillonnage et les premiers signes de corrosion ou d'accumulation minérale autour des raccords agroalimentaires.
Les usines qui obtiennent de meilleurs résultats à long terme traitent généralement les contrôles hebdomadaires comme une occasion de confirmer les tendances, et pas seulement de rechercher des défaillances évidentes. Si une branche nécessite constamment la même attention chaque semaine, cela indique généralement un indice de géométrie ou de contrôle des composants qui mérite d'être escaladé.
Inspection approfondie périodique
L'inspection périodique doit ouvrir des assemblages représentatifs à haut risque plutôt que de se fier uniquement à l'apparence externe. Dans les usines agroalimentaires, cela signifie souvent ouvrir des joints à collier sélectionnés, examiner les internes des vannes, vérifier les zones adjacentes aux soudures, confirmer l'utilisation des pièces de rechange correctes et utiliser l'inspection par endoscope si justifiée. Cette revue plus approfondie est particulièrement précieuse après des changements de procédé, une nouvelle chimie, des changements de produit ou des écarts répétés de nettoyage.
Qu'est-ce qui devrait déclencher une maintenance immédiate
Certaines conditions devraient déclencher une enquête immédiate au lieu d'attendre l'intervalle planifié suivant. Celles-ci incluent :
- Fuite répétée au même joint à collier
- Écarts ATP ou microbiologiques provenant d'un emplacement répété
- Eau de rinçage retenue persistante après nettoyage
- Décoloration visible autour d'une soudure ou d'une face de virole
- Préoccupation récurrente de transfert d'allergènes après changement de produit
- Rétention d'odeur inattendue dans une vanne ou un ensemble d'échantillonnage
| Fréquence | Priorité Maintenance | Cibles typiques |
|---|---|---|
| Quotidien | État hygiénique visible et anomalies de fonctionnement | Fuites, résidus, reflux, odeurs, joints à problèmes répétés |
| Hebdomadaire | État des joints et revue récurrente des assemblages | Garnitures, vannes, points d'échantillonnage, assemblages de brides, accumulation précoce |
| Périodique | Inspection et vérification interne représentative | Joints ouverts, internes de vannes, zones de soudure, géométrie de branchement, pièces de rechange |
| Événement déclencheur | Inspection corrective immédiate | Échecs ATP, fuites répétées, entraînement, eau retenue, signes de corrosion |
Comment les programmes de maintenance se dégradent en pratique
Lorsque le même joint de bride continue de fuir
Une fuite répétée au même joint de bride signifie généralement que l'usine traite le symptôme, pas la cause. Sur plus d'une ligne de boissons, l'appel pour une fuite revenait toujours au même assemblage, même après plusieurs tentatives de resserrage. La cause réelle s'est avérée être une combinaison d'un léger désalignement de la bague de serrage et d'un joint déjà endommagé par une surcompression répétée. Une fois l'assemblage corrigé et le joint approuvé installé, le problème de fuite récurrent a disparu. La leçon est simple : arrêtez de traiter chaque fuite récurrente comme un problème de couple.
Quand la boucle principale de CIP semble bonne mais qu'une branche échoue toujours
Certains des problèmes de maintenance agroalimentaire les plus difficiles proviennent de géométries locales qui sont cachées derrière des données de nettoyage acceptables à l'échelle du système. Cela est particulièrement courant au niveau des branches d'échantillonnage, des tés d'instrument à cul-de-sac court et des grappes de vannes à faible débit. Sur les lignes laitières, il n'est pas rare de voir des résultats acceptables du côté retour du CIP tandis que la même branche continue de montrer des résidus retenus ou des échecs ATP répétés. Dans ces situations, la meilleure correction est généralement géométrique ou procédurale plutôt que chimique.
Quand une mise à niveau de matériau ne résout pas le problème
Les mises à niveau de matériau peuvent réduire le risque lié à la corrosion, mais elles ne corrigent pas une mauvaise géométrie hygiénique. Une erreur répétée sur le terrain est de passer à un alliage plus résistant à la corrosion et de s'attendre à ce que les écarts d'assainissement disparaissent. En réalité, les lignes avec des soudures rugueuses, des poches mortes, un mauvais drainage ou un contrôle faible des joints peuvent rester difficiles à entretenir, quel que soit l'alliage. Si vous devez revoir le côté matériau du problème, consultez l'acier inoxydable 316L dans les applications agroalimentaires, mais gardez cette revue liée à la drainabilité et à l'accès à l'inspection.
Pourquoi l'inspection basée sur les risques fonctionne généralement mieux qu'un démontage complet de la ligne
De nombreuses usines perdent trop de temps à ouvrir les mauvaises parties du système. Un démontage complet de la ligne semble minutieux, mais il augmente souvent la main-d'œuvre, le temps de redémarrage et l'exposition sans améliorer le contrôle des véritables points faibles hygiéniques. Un modèle plus efficace est l'inspection basée sur les risques, où les points d'échantillonnage, les grappes de vannes, les joints avec joints, les branches à faible drainage et les zones à problèmes récurrents reçoivent plus d'attention que les tronçons de tube droits.
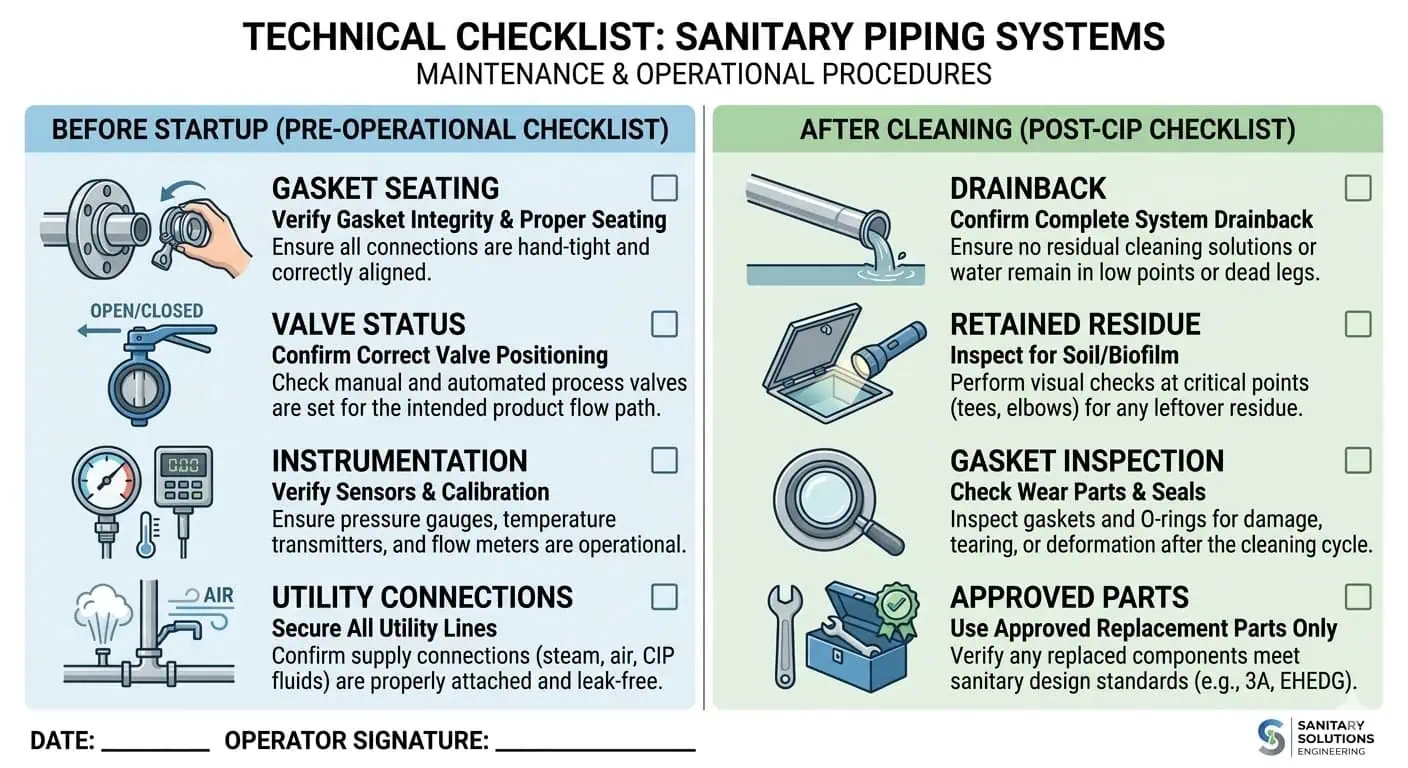
Liste de contrôle pratique pour la maintenance des systèmes de tuyauterie agroalimentaire
Avant le démarrage
- Vérifier l'assemblage correct et la mise en place des joints
- Confirmer qu'aucune eau piégée évidente ne reste dans les points bas critiques
- Vérifier que les joints ouverts ont été réassemblés avec des pièces approuvées
- Vérifier les positions des vannes et l'état de l'assemblage d'échantillonnage
- Confirmer que la ligne est libérée du nettoyage et prête pour le contact avec le produit
Après changement ou nettoyage
- Inspecter les joints critiques et les emplacements à problèmes récurrents
- Confirmer l'absence de transfert visible ou d'odeur inhabituelle
- Vérifier le reflux aux points bas à plus haut risque
- Contrôler les points d'échantillonnage et les assemblages de branche pour résidus retenus
Pendant l'arrêt de routine
- Ouvrir des joints représentatifs à haut risque
- Inspecter les internes de vannes sélectionnées et les assemblages d'échantillonnage
- Examiner les zones adjacentes aux soudures pour rugosité, décoloration ou accumulation
- Confirmer que l'historique de remplacement correspond aux matériaux et tailles approuvés
Avant l'audit ou la revue de validation
- Confirmer la traçabilité des pièces de rechange critiques
- Vérifier la méthode de nettoyage documentée et les déclencheurs de maintenance
- Examiner les registres d'inspection pour les assemblages à problèmes répétés
- Confirmer que les matériaux approuvés et les pièces de rechange hygiéniques sont toujours contrôlés correctement
Comment réduire les problèmes de maintenance grâce à une meilleure conception et sélection des composants
Utiliser des raccords agroalimentaires qui correspondent au régime de nettoyage réel
Le système agroalimentaire le plus facile à entretenir est celui dont les raccords ont été sélectionnés pour la méthode de nettoyage réelle, pas seulement pour la taille nominale et le coût. Si vous choisissez de nouveaux composants ou examinez un point de problème répété, utilisez notre guide sur comment choisir des raccords hygiéniques pour l'agroalimentaire pour aligner le choix du raccord avec le CIP, l'inspection manuelle, la vidange et le risque hygiénique.
Séparez la sélection des matériaux de la revue de géométrie—mais jamais de la réalité de la maintenance
Le bon alliage aide, mais il ne remplace pas le besoin d'une géométrie maintenable et de joints contrôlés. Si votre système fonctionne dans le lait, la saumure, les boissons ou d'autres services agroalimentaires agressifs, examinez le côté matériel avec l'acier inoxydable 316L dans les applications agroalimentaires, mais gardez cette revue liée à la vidange, la qualité de soudure et l'accès à l'inspection.
Ne mélangez pas la commodité industrielle avec les exigences de service agroalimentaire
Un composant qui est mécaniquement pratique n'est pas toujours le composant agroalimentaire le plus facile à maintenir. Si une ligne continue de créer des problèmes d'assainissement ou de remplacement, comparez le style de connexion et de composant aux exigences agroalimentaires plutôt que de supposer que toute pièce en acier inoxydable est suffisamment proche. Cette distinction est expliquée dans raccords agroalimentaires vs raccords industriels.
Conclusion : La maintenance des tuyauteries agroalimentaires est un système de conception, de nettoyage et d'inspection—pas une tâche unique
La maintenance des tuyauteries agroalimentaires fonctionne mieux lorsque la méthode de nettoyage, la routine d'inspection, le contrôle des composants et la conception hygiénique sont traités comme un seul système. L'objectif principal n'est pas seulement de maintenir l'acier inoxydable propre en apparence. Il s'agit de garantir que les surfaces en contact avec le produit soient véritablement nettoyables, drainables, vérifiables et fiables dans toutes les conditions de production, de changement de production et d'audit.
Si vous souhaitez moins de déviations en matière d'hygiène, moins de fuites répétées et un meilleur contrôle des temps d'arrêt, concentrez-vous d'abord sur les assemblages qui présentent le plus de risque hygiénique : joints, raccords à collier, soudures, dérivations, vannes et points d'échantillonnage. Puis, soutenez ce travail par une meilleure sélection des raccords, une meilleure revue des matériaux et de meilleurs registres d'inspection. Cette combinaison transforme un système de tuyauterie agroalimentaire d'une charge de maintenance en un actif hygiénique stable. Pour un chemin de spécification hygiénique complet, consultez comment choisir des raccords hygiéniques pour l'agroalimentaire, raccords agroalimentaires vs raccords industrielset l'acier inoxydable 316L dans les applications agroalimentaires. Pour les projets destinés aux marchés de l'UE, les décisions concernant les matériaux et le contact alimentaire doivent également être conformes à Règlement (CE) n° 1935/2004.
FAQ
À quelle fréquence les systèmes de tuyauterie agroalimentaire doivent-ils être inspectés ?
La fréquence d'inspection doit être basée sur le risque hygiénique, l'intensité du nettoyage et l'historique des déviations. Les contrôles quotidiens doivent se concentrer sur les fuites visibles, les résidus et le mauvais drainage. Les contrôles hebdomadaires doivent se concentrer sur les joints, l'état des vannes et les assemblages à problèmes répétés. Les inspections périodiques plus approfondies doivent ouvrir des points représentatifs à haut risque et vérifier l'état interne.
Le nettoyage en place (CIP) est-il suffisant pour tous les systèmes de tuyauterie agroalimentaire ?
Non. Le CIP (nettoyage en place) n'est efficace que lorsque la géométrie de l'équipement, la couverture du flux et le drainage le permettent. Certains assemblages nécessitent encore une inspection manuelle, un nettoyage hors place ou un démontage ciblé car leur géométrie est plus difficile à nettoyer de manière fiable en place.
Quelles parties d'un système de tuyauterie agroalimentaire tombent en panne en premier ?
Les premiers points faibles en matière d'hygiène ne sont généralement pas les tubes droits. Ce sont plus souvent les joints, les raccords à collier, les internes de vannes, les points d'échantillonnage, les zones adjacentes aux soudures et les dérivations à faible drainage où les résidus ou l'exposition chimique sont plus difficiles à contrôler.
Comment les impasses affectent-elles la maintenance des tuyauteries agroalimentaires ?
Les bras morts augmentent la difficulté de nettoyage et la charge d'inspection. Ils peuvent piéger le produit ou l'eau de rinçage, réduire l'efficacité du CIP et devenir des sources répétées de résidus, d'échecs ATP ou de positifs microbiologiques s'ils ne sont pas examinés et entretenus correctement.
Les joints doivent-ils être remplacés selon un calendrier fixe ou en fonction de leur état ?
La plupart des installations nécessitent les deux approches. L'examen basé sur l'état est essentiel car les joints d'étanchéité tombent en panne en raison de la chimie, de la chaleur, de l'historique de compression et de la manipulation. Mais les systèmes agroalimentaires critiques bénéficient souvent aussi d'un intervalle de remplacement contrôlé afin que les joints ne soient pas poussés jusqu'à la défaillance en service de production.
Est-ce que l'acier 316L réduit les problèmes de maintenance des tuyauteries agroalimentaires ?
Il peut réduire les problèmes liés à la corrosion dans des services plus exigeants, mais il ne remplace pas la conception agroalimentaire ou la discipline d'inspection. Un système en 316L avec une mauvaise capacité de drainage, une qualité de soudure faible ou un contrôle incorrect des joints d'étanchéité peut encore devenir difficile à maintenir de manière hygiénique.




