يستخدم الفولاذ المقاوم للصدأ 316L على نطاق واسع في تطبيقات العمليات الحيوية لأنه يوفر للمهندسين توازنًا عمليًا بين مقاومة التآكل، وقابلية اللحام، وقبول النظافة، والتوافق مع متطلبات التصنيع والتوثيق عالي النقاء. في أنظمة التكنولوجيا الحيوية، هذا الأمر مهم لأن المادة لا تقوم فقط بتحمل الضغط. يجب أن تدعم قابلية التنظيف، اللحام المتكرر، أسطح تلامس المنتج المستقرة، والتحكم طويل الأجل تحت دورات التنظيف والتطهير والتشغيل المتكررة.
هذا لا يعني أن 316L هو الإجابة الصحيحة تلقائياً في كل مكان. في مشاريع العمليات الحيوية الحقيقية، يعتمد اختيار المادة الصحيحة على سائل العملية الفعلي، نظام التنظيف في المكان/التعقيم في المكان، متطلبات تشطيب السطح، كثافة اللحام في النظام، حساسية العملية للتآكل الموضعي أو احتباس الرواسب، والعبء التوثيقي المطلوب للإطلاق. قرار 316L الجيد ليس قرار “معدن متميز”. إنه قرار هندسي صحي قائم على المخاطر.
يشرح هذا الدليل سبب شيوع الفولاذ 316L في خدمات العمليات الحيوية، حيث يضيف قيمة حقيقية، وأين قد يكون الفولاذ 304/304L مقبولاً، والمشاكل التي لا يحلها الفولاذ 316L لا حل عندما تكون المشكلة الحقيقية هي الهندسة، تنفيذ اللحام، التحكم في الإحكام، أو انضباط التوثيق.
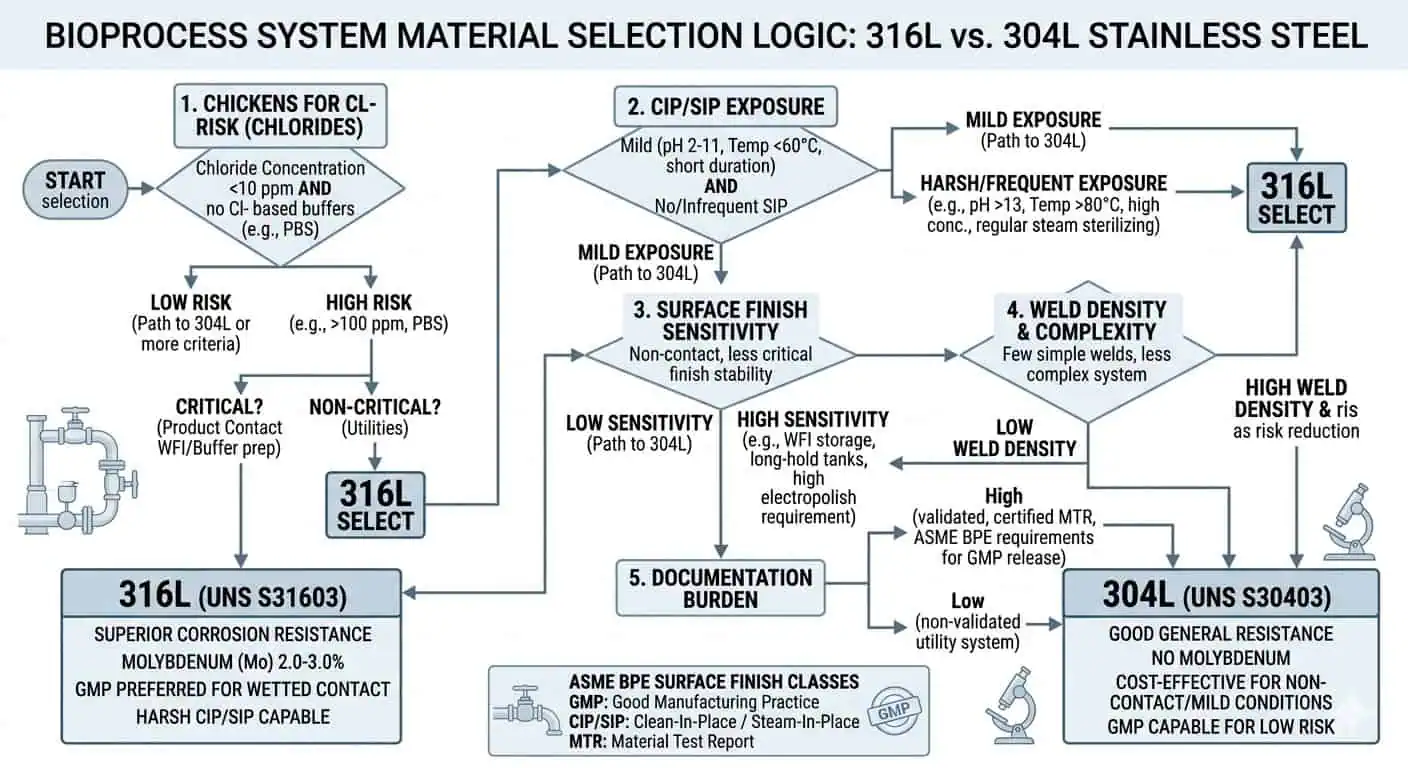
ما الذي يجعل 316L مختلفًا في خدمة العمليات الحيوية
لماذا يهم الموليبدينوم
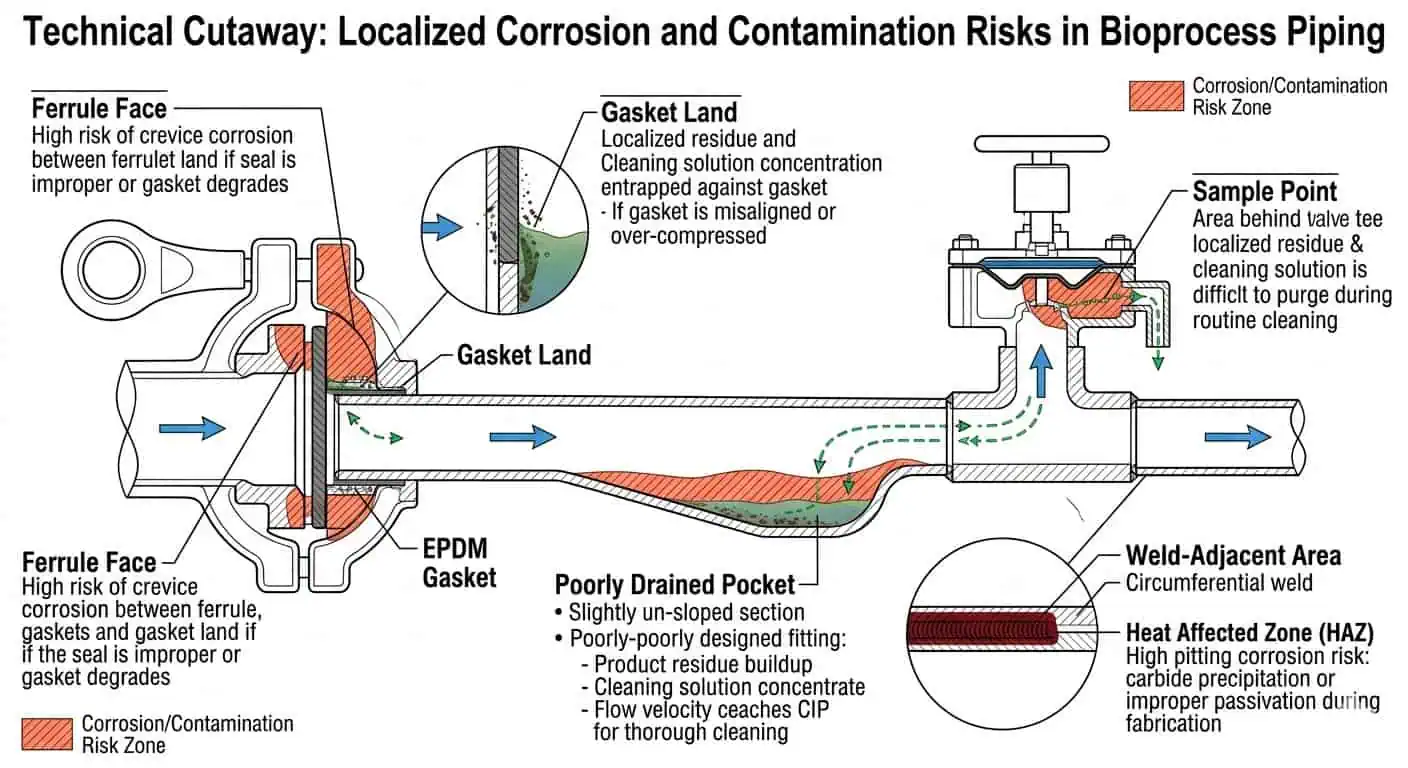
الفرق العملي الأهم بين 316L و304/304L هو إضافة الموليبدينوم. تشير إرشادات معهد النيكل إلى أن النوع 316 يحتوي على حوالي 2–3% موليبدينوم ويوفر مقاومة أفضل للتآكل من النوع 304، خاصة في بيئات الكلوريد التي تميل إلى التسبب في التنقيب. في أنظمة العمليات الحيوية، يهم هذا أقل في مركز الأنبوب المستقيم وأكثر في الميزات المحلية حيث يصبح التعرض مركزًا: وجوه الحلقات، وأراضي الحشيات، ونقاط العينة، ومقاعد الصمامات، والمناطق المجاورة للحام، والجيوب سيئة الصرف حيث يمكن أن يبقى بقايا المنتج أو محلول التنظيف لفترة أطول من المقصود. تشير إرشادات تصميم معهد النيكل وإرشادات معهد النيكل لأنابيب الفولاذ المقاوم للصدأ في نفس الاتجاه: تؤدي الدرجات الحاملة للموليبدينوم أداءً أفضل حيث يصبح التآكل الموضعي هو الخطر الحقيقي.
رؤية بحثية: غالبًا ما يتحدث المهندسون عن “مقاومة التآكل” كما لو كانت خاصية موحدة عبر الخط بأكمله. في الممارسة العملية، عادةً ما تكشف أنظمة العمليات الحيوية عن هامش المواد الخاص بها أولاً عند التجميع المحلي حيث يكون خطر الشقوق، أو احتباس الرطوبة، أو تركيز التنظيف هو الأعلى.
لماذا تهم درجة “L” منخفضة الكربون في الأنظمة الصحية الملحومة
تعد الدرجة “L” منخفضة الكربون مهمة لأن أنابيب العمليات الحيوية تُصنع وتُلحم بشكل كبير. تخلق مجمعات التوزيع، وصلات الفروع، نقاط أخذ العينات، مسارات اللحام المداري، تجميعات الهياكل، ومجموعات الصمامات جميعها مناطق متأثرة بالحرارة يجب أن تظل مقاومة للتآكل وقابلة للتنظيف بمرور الوقت. تشير بيانات معهد النيكل على الفولاذ المقاوم للصدأ الأوستنيتي المشكل إلى أن 316L يوفر مقاومة أكبر لترسيب الكربيدات أثناء اللحام مقارنة بـ 316 القياسي، وهو أحد الأسباب التي تجعل الدرجات منخفضة الكربون مستخدمة على نطاق واسع في تصنيع العمليات الصحية.
نصيحة الخبراء: في عمل العمليات الحيوية، لا تسأل فقط عما إذا كان النظام “316L”. اسأل عما إذا كانت منطقة اللحام 316L لا تزال تحافظ على النية الصحية للتصميم بعد التصنيع، والإصلاح، والتخميل، والخدمة المتكررة.
ما يتحسن به 316L — وما لا يفعله
316L تحسن هامش التآكل. لا تخلق تلقائيًا نظامًا صحيًا. يمكنه تحسين مقاومة التآكل الموضعي، وتوفير ملاءمة أقوى للخدمة الملحومة الصحية، ودعم توقعات العمليات الحيوية الأكثر تطلبًا. لكنه لا يصلح الأرجل الميتة، أو سوء قابلية التصريف، أو هندسة الفروع الضعيفة، أو اللحامات الخشنة، أو اختيار الحشيات غير الصحيح، أو إمكانية التتبع غير المكتملة.
حالة صناعية نموذجية: قام فريق بترقية تجميع محلي حرج إلى 316L بعد تكرار مخاوف التفتيش، لكن المشكلة الحقيقية اتضح أنها سوء تصريف العودة وترتيب فرع يصعب تنظيفه. حسّن ترقية السبيكة جزءًا واحدًا من الخطر، لكن الضعف الصحي المهيمن بقي لأن الهندسة لم تُصحح أبدًا.

لهذا السبب يجب قراءة هذه المقالة معاً مع تصميم الأنابيب عالية النقاء لمرافق التكنولوجيا الحيوية و اعتبارات الصيانة والتحقق لأنظمة الأنابيب الصحية. يجب ألا يتم فصل قرارات المواد وقرارات التصميم الصحي لفترة طويلة.
لماذا يُعد الفولاذ المقاوم للصدأ 316L شائعًا جدًا في تطبيقات العمليات الحيوية
لماذا تعامل أنظمة العمليات الحيوية غالبًا الفولاذ المقاوم للصدأ 316L كخط أساس عملي
في العديد من أنظمة العمليات الحيوية الحرجة، يُعامل الفولاذ المقاوم للصدأ 316L كخط أساس عملي لأن المخاطر أوسع من مجرد التآكل السائب وحده. يجب أن تظل الأسطح الملامسة للمنتج غير متفاعلة، وغير مضافة، وغير ممتصة تحت ظروف التشغيل والتنظيف الفعلية. هذا هو المنطق المدمج في FDA 21 CFR 211.65. في المنشآت الحقيقية، يتم دعم هذا المطلب ليس فقط باختيار السبيكة، ولكن بمدى جودة عمل تلك السبيكة مع التصنيع عالي النقاء، والتحكم في اللحام، والقابلية للتنظيف، والتفتيش، والتوثيق طويل الأجل.
واقع الصناعة: لا تلجأ الفرق إلى الفولاذ المقاوم للصدأ 316L بشكل افتراضي لأنه يبدو أكثر تقدمًا. بل تلجأ إليه لأنه أسهل في الدفاع عنه في بيئة عالية العواقب، وعالية التوثيق، وعالية القابلية للتنظيف حيث يكون التآكل المحلي أو عدم استقرار السطح مكلفًا لتفسيره بعد بدء التشغيل.
كيف يدعم ASME BPE و ASTM A270 هذا المنطق
يدعم ASME BPE رؤية النظام بأكمله بدلاً من قرار بسيط يعتمد على السبيكة فقط. يذكر ASME أن BPE ينطبق على المعدات المستخدمة في الصناعات الحيوية، والصيدلانية، والصناعات الصحية ذات الصلة ويغطي المواد، والتصميم، والتصنيع، والتفتيش، والاختبار، والشهادات. هذا هو بالضبط السبب في أنه لا ينبغي مناقشة الفولاذ المقاوم للصدأ 316L في تطبيقات العمليات الحيوية كموضوع يعتمد على المعدن وحده.
يُعزز معيار ASTM A270 جانب الأنابيب من نفس المنطق. ينص نطاقه على أنه يمكن طلب الجودة الصيدلانية كمطلب تكميلي. وهذا مهم لأنه يؤكد نقطة هندسية مهمة: في خدمة العمليات الحيوية، غالبًا ما يتجاوز قرار الأنابيب توريد الأنابيب الصحية الأساسية. قد يحتاج المشروع إلى حزمة صحية أكثر تحكمًا من تلك التي تتطلبها الخدمة الغذائية العامة أو الصحية.
لماذا لا يزال 304/304L يظهر في بعض المشاريع
يمكن أن يظهر 304 أو 304L في المشاريع المتعلقة بالعمليات الحيوية، ولكن عادةً في أدوار أكثر محدودية. قد تستخدم الخدمات المساعدة منخفضة المخاطر، أو الأنظمة القديمة، أو التجميعات الداعمة غير الحرجة، أو التصميمات التي تعتمد تاريخيًا على الميزانية مواد من عائلة 304. الخطأ الهندسي ليس في عدم الاعتراف بوجود 304. الخطأ هو افتراض أن نفس المنطق ينطبق تلقائيًا على خدمة العملية المبللة ذات العواقب الأعلى دون التحقق من نمط الفشل الذي يحاول المشروع منعه بالفعل.
حيث يضيف 316L قيمة حقيقية في أنظمة العمليات الحيوية
أنابيب ملامسة المنتج وحلقات النقل عالية النقاء
تعد أنابيب ملامسة المنتج وحلقات النقل عالية النقاء الأماكن الأوضح حيث يكسب 316L عادةً موقعه. هذه هي الأنظمة حيث تتداخل قابلية التنظيف، وتحمل التآكل، واللحام المتكرر، وثقة التوثيق. في تلك البيئات، لا يبقى التآكل المحلي أو تدهور السطح مشكلة ميكانيكية بحتة. يصبح بسرعة مشكلة في جودة المنتج، أو التفتيش، أو التأهيل.
الأقسام المعرضة للتنظيف في المكان (CIP) / التعقيم في المكان (SIP)
التعرض المتكرر للتنظيف في المكان (CIP) والتعقيم في المكان (SIP) غالباً ما يبرر استخدام الفولاذ المقاوم للصدأ 316L حتى عندما لا يبدو سائل العملية نفسه عدوانياً بشكل خاص. في تطبيقات العمليات الحيوية، تتعرض المواد لبيئة مشتركة من المنتج، وماء الشطف، وعوامل التنظيف الكيميائية، والمطهرات، والماء الساخن، والبخار النظيف. يمكن أن يكون التعرض الكلي أكثر تطلباً من سائل العملية نفسه.
حالة صناعية نموذجية: لم يظهر قسم من خط الأنابيب قلقاً أولاً أثناء نقل العملية. بل ظهر القلق بعد أن جعل التعرض المتكرر للتنظيف والبخار استقرار السطح المحلي وجودة اللحام أكثر أهمية مما كان مفترضاً في الأصل. هذا أحد أكثر الأسباب شيوعاً لتقليل الفرق للحمل الحقيقي على المادة.
هذا أيضاً هو السبب في أن FDA 21 CFR 211.67 ذو صلة بمناقشة المواد. التنظيف والصيانة ليسا تفاصيل تشغيلية ثانوية. إنهما جزء مما يجب أن يتحمله المعدات في حالة مضبوطة.
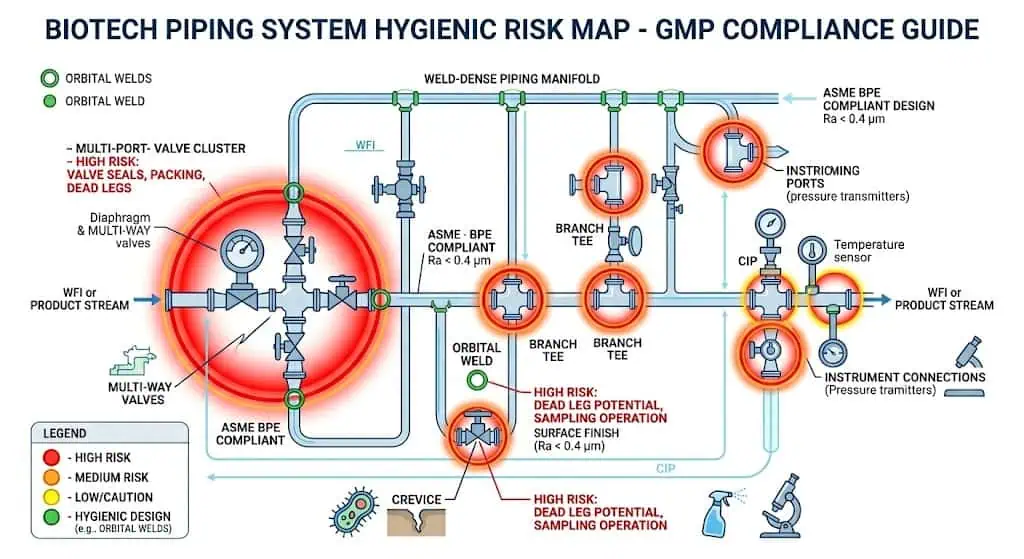
مجمعات توزيع ذات كثافة لحام، وفروع أخذ العينات، والتجميعات المحلية
كلما أصبح النظام أكثر تعقيداً محلياً وأكثر لحاماً، كلما أصبحت القاعدة المادية الأقوى أكثر قيمة. تجمع مجمعات توزيع العينات، ومجموعات الصمامات، والمنصات ذات الفروع الكثيرة، والتوصيلات الثلاثية للأجهزة، وتجميعات التوزيع بين كثافة اللحام، وواجهات الإغلاق، وهندسة الفروع، وحساسية الفحص. هذه هي المواقع حيث يصبح هامش المادة مرئياً أولاً لأن الحمل الصحي يتركز هناك.
نصيحة الخبراء: إذا لم يستطع مشروع حساس للتكلفة ترقية النظام بأكمله، ابدأ بالتجميعات المحلية ذات العواقب الأعلى. في العملية الحيوية، غالباً ما يكون المجمع المحلي أكثر أهمية من المسار المستقيم.
حيث لا يحل الفولاذ المقاوم للصدأ 316L المشكلة الحقيقية
هندسة رديئة
لا يحل الفولاذ المقاوم للصدأ 316L مشكلة الأرجل الميتة، أو الميلان غير الكافي، أو الصرف السيء، أو هندسة الفروع صعبة التنظيف. يبقى الرجل الميت رجلاً ميتاً في سبيكة تحتوي على الموليبدينوم. إذا كان نمط الفشل الصحي الحقيقي هو تبادل السوائل غير الكافي أو الاحتفاظ المحلي، فقد يحسن ترقية المادة تحمل التآكل مع ترك مشكلة التنظيف الرئيسية دون تغيير.
تنفيذ اللحام الضعيف وحالة السطح
لا ينقذ السبيكة الصحيحة منطقة اللحام الرديئة. يمكن أن يقلل التلوين الحراري المحلي، أو جودة التنقية الرديئة، أو الكفاف غير المنتظم، أو تاريخ الإصلاح، أو التشطيب التالف من القيمة الصحية لقرار المادة الجيد. هذا هو السبب في أنه يجب مراجعة التشطيب وتنفيذ اللحام معًا مع اختيار السبيكة، وليس بعد ذلك كمشكلة ورشة فقط.
لهذا السبب، يرتبط هذا الموضوع مباشرة بـ الفولاذ المقاوم للصدأ 316L المصقول كهربائياً مقابل الفولاذ المقاوم للصدأ 316L القياسي. في الخدمة عالية النقاء، تهم حالة السطح المبللة المثبتة أكثر من علامة السبيكة وحدها.
استراتيجية الختم الخاطئة
العديد من “مشاكل المواد” الظاهرة هي في الواقع مشاكل واجهة الختم. إذا كانت المادة الخاطئة للجوان، الحجاب الحاجز، أو المقعد هي التي تتحكم في نمط الفشل المحلي، فإن درجة فولاذ مقاوم للصدأ أفضل لن تصحح السبب الحقيقي. في أنظمة العمليات الحيوية، يجب مراجعة استراتيجية المعدن والمطاط معًا.
توثيق ضعيف وإمكانية التتبع
في العمل المنظم للعمليات الحيوية، فإن مادة صحيحة تقنيًا بدون سلسلة أدلة نظيفة لا تزال تشكل خطرًا على المشروع. إذا لم يتمكن المشروع من إثبات أي مادة تم تركيبها وأين، وكيف تم لحامها، وما هي التشطيب الذي تلقيته، وكيف تم التحكم في التغييرات الميدانية، فإن الخطر لم يُغلق بالكامل. لهذا السبب يجب أن يرتبط هذا المقال أيضًا بـ التوثيق وإمكانية التتبع في مشاريع أنابيب الأدوية.
316L مقابل 304L في تطبيقات العمليات الحيوية
هامش التآكل
الفرق الأكثر عملية بين 316L و304L في خدمة العمليات الحيوية ليس القوة. إنه هامش التآكل. يحتوي النوع 316 على الموليبدينوم الذي يحسن المقاومة في الظروف المعرضة للكلوريد وفي أنواع البيئات المحلية حيث يصبح الهجوم النقري أو الشقوقي أكثر واقعية. هذا الهامش يهم أكثر حيث تكون العواقب الصحية للهجوم المحلي عالية، وليس حيث يكون النظام بالفعل منخفض المخاطر وسهل التفتيش.
التصنيع وإمكانية اللحام
يجعل التركيب الكيميائي منخفض الكربون في 316L مناسبًا بشكل أفضل للعديد من الأنظمة الصحية الملحومة مقارنة بـ316 القياسي. لا يلغي ذلك الحاجة إلى انضباط قوي في اللحام، لكنه يساعد في جعل السبيكة أسهل للدفاع عنها في الأنظمة ذات الفروع الكثيفة واللحام المداري حيث يكون العبء الصحي عند اللحام كبيرًا.
التكلفة مقابل مخاطر دورة الحياة
المقارنة الحقيقية ليست سعر السبيكة وحدها. إنها مخاطر دورة الحياة. إذا قللت المادة ذات الدرجة الأعلى من إعادة العمل، أو تصعيد التفتيش، أو الاستبدال المرتبط بالتآكل، أو تأخير التأهيل، أو عدم الاستقرار الصحي المحلي، فقد يكون التكلفة الإجمالية أسهل للتبرير مما يشير إليه فرق المواد الخام.
نصيحة الخبراء: لا تقارن بين 304L و316L فقط بسعر الشراء لكل متر. قارن بينهما بتكلفة الفشل في التجميع المحدد حيث يكون الفرق مهمًا.
قاعدة الاختيار السريع
- ابدأ بـ 316L للخدمة الحيوية المبللة في العمليات الحيوية، حلقات النقل عالية النقاء، التجميعات المعرضة لـ CIP/SIP، والتجميعات الصحية المحلية الكثيفة اللحام.
- احتفظ بـ 304/304L قيد المراجعة فقط حيث تكون الخدمة منخفضة المخاطر حقًا والعواقب الصحية أقل ماديًا.
- لا تصعد إلى ما بعد 316L ما لم يبرره التركيب الكيميائي الفعلي، أو تاريخ الفشل المتكرر، أو آلية تآكل محددة.
تشطيب السطح، التلميع الكهربائي، ولماذا لا يكفي الفولاذ المقاوم للصدأ 316L وحده
السبيكة الأساسية ليست النتيجة الصحية الكاملة
تعتمد النتيجة الصحية النهائية على السطح المثبت، وليس فقط على عائلة السبيكة الأساسية. في تطبيقات العمليات الحيوية، يمكن لنفس الفولاذ المقاوم للصدأ 316L أن ينتج نتائج مختلفة جدًا اعتمادًا على تشطيب السطح، وجودة اللحام، وتاريخ إعادة العمل، وانضباط التخميل، والهندسة المحلية. لهذا السبب يجب تحديد المادة والتشطيب معًا أينما كان الهدف الصحي حساسًا لحالة السطح.
عندما يصبح الفولاذ المقاوم للصدأ 316L الملمع كهربائيًا أسهل في الدفاع عنه
عادة ما يتم تبرير الفولاذ المقاوم للصدأ 316L الملمع كهربائيًا عندما تكون اتساق التشطيب، وقابلية الفحص، وثقة قابلية التنظيف المتعلقة بالسطح أكثر أهمية مما يمكن أن يدعمه تشطيب قياسي بشكل مريح. النقطة ليست أن التلميع الكهربائي دائمًا متفوق في المجرد. النقطة هي ما إذا كانت عملية وعبء ضمان الجودة يجعلان تباين التشطيب أكثر صعوبة في الدفاع عنه.
حالة هندسية نموذجية
حالة عملية حيوية واقعية ليست “فشل الفولاذ المقاوم للصدأ 316L.” كانت المواصفة القياسية 316L مقبولة كمادة أساسية، لكن التشطيب المحلي وحالة اللحام لم يدعما بشكل كامل ثقة الفحص الصحي التي تطلبها المشروع في النهاية. في تلك الحالة، المشكلة ليست في عائلة السبيكة الخاطئة، بل أن الهدف الصحي كان حساسًا للتشطيب.
كيف يجب على المهندسين تحديد 316L لتطبيقات العمليات الحيوية
حدد الدرجة بوضوح
لا تكتب “فولاذ مقاوم للصدأ” على رسم العمليات الحيوية وتفترض أن سلسلة التوريد ستفسره بشكل صحيح. إذا كان 316L مطلوبًا، حدده بوضوح للأجزاء المبللة الفعلية التي تهم: الأنابيب، الحلقات، أجسام الصمامات، مجمعات التوزيع، تجميعات العينات، وصلات الفروع، والواجهات المحلية الملامسة للمنتج.
حدد السبيكة، التشطيب، اللحام، والوثائق معًا
مواصفة 316L القوية ليست مجرد إشارة للمادة. يجب أن تحدد أيضًا التشطيب المطلوب، توقعات التصنيع، منطق قبول اللحام، توقعات التخميل أو المعالجة اللاحقة حيثما كان ذلك مناسبًا، وحزمة الوثائق المطلوبة للإصدار. ملاحظة السبيكة الغامضة عادةً ما تخلق ضوضاء مشروع أكثر لاحقًا، وليس أقل.
راجع المادة مع نظام التنظيف
سائل العملية هو جزء فقط من بيئة المادة. يجب على المهندسين مراجعة كيمياء التنظيف الفعلية، والتعرض للتطهير، واستخدام البخار النظيف، والرطوبة أثناء الإيقاف، والدورات الحرارية مع سائل العملية. قد يبدو خط العملية معتدلاً في الخدمة، لكنه قد يصبح متطلبًا في التعرض الكامل لدورة الحياة.
تحديد طلب العطاء وحمل التسليم مبكرًا
أفضل وقت لتحديد ما يجب أن يتضمنه حزمة 316L هو قبل طلب العطاء. إذا كان المشروع يتوقع أنابيب بجودة صيدلانية، أو أدلة على التشطيب، أو سجلات اللحام، أو تتبع قوي، فيجب كتابة هذه المتطلبات قبل بدء التصنيع. إعادة بناء سلسلة الأدلة لاحقًا عادةً ما يكون أبطأ وأقل موثوقية.
الأخطاء الشائعة عند استخدام 316L في تطبيقات العمليات الحيوية
معاملة 316L كترقية عالمية
تحسن 316L هامش المادة، لكنها لا تحل تلقائيًا المخاطر الصحية السائدة في كل تجميع.
مقارنة تكلفة المواد الخام فقط
في التجميعات الصحية عالية العواقب، يهم خطر دورة الحياة أكثر من تكلفة المعدن الخام وحدها.
تجاهل تشطيب السطح
غالبًا ما تؤثر الحالة النهائية المبللة على النتيجة الصحية بشكل أكثر مباشرة من مجرد تسمية السبيكة وحدها.
تجاهل تنفيذ اللحام
السبيكة الأساسية الصحيحة مع تنفيذ لحام ضعيف لا تزال نظامًا حيويًا ضعيفًا.
استخدام 316L لتجنب إصلاح مشاكل التصميم
إذا كانت المشكلة الحقيقية هي هندسة الفرع، أو سلوك الأرجل الميتة، أو قابلية التصريف، يمكن لـ 316L تحسين التسامح دون إصلاح السبب الصحي المهيمن.
نسيان التوثيق وإمكانية التتبع
اختيار المواد الصحيح تقنيًا لا يزال يخلق مخاطر المشروع إذا كانت سلسلة الأدلة المثبتة ضعيفة.
قائمة مراجعة عملية للاختيار
استخدم 316L إذا
- التطبيق هو خدمة عملية حيوية حرجة مبللة.
- يتعرض التجميع بشكل متكرر لـ CIP أو SIP.
- التجميع المحلي يكون كثيف اللحام، كثيف الفروع، أو حساس للفحص.
- النتيجة الصحية لتآكل موضعي أو عدم استقرار السطح عالية.
304/304L قد يظل كافياً إذا
- الخدمة تكون منخفضة المخاطر حقاً.
- التنظيف والتعرض الحراري يكونان أكثر اعتدالاً.
- النتيجة الصحية المحلية تكون أقل مادياً.
- المشروع له مبرر واضح وموثق لاستخدام خيار سبيكة أقل.
تصعيد المراجعة إلى ما بعد 316L إذا
- الكيمياء تكون غير عادية أو عدوانية بشكل غير عادي.
- هناك تاريخ فشل محلي متكرر لا يفسره 316L والتصنيع الجيد.
- العملية حساسة للغاية للإنهاء وحالة السطح بما يتجاوز حزمة 316L القياسية.
- يحتوي المشروع على آلية تآكل محددة بوضوح تتطلب أكثر من خط أساس 316L عام.
أسئلة يجب الإجابة عليها قبل طلب عرض السعر
- ما هو سائل العملية أو المنتج الذي يتلامس مع السطح؟
- ما هو التعرض الفعلي للتنظيف في المكان (CIP)، والتعقيم في المكان (SIP)، والتعقيم، والإغلاق الذي سيتعرض له التجميع؟
- أي التجميعات المحلية لديها أعلى كثافة لحام أو مخاطر شقوق؟
- ما هو إنهاء السطح الذي يتطلبه الهدف الصحي فعليًا؟
- ما هي حزمة التوثيق وإمكانية التتبع التي ستحتاجها ضمان الجودة أو المشروع لاحقًا؟
- هل يحل 316L الخطر السائد، أم يحسن فقط جزءًا واحدًا من مشكلة أكبر؟

الخلاصة: 316L هو الإجابة الصحيحة فقط عندما يغير الخطر الصحي الحقيقي للتجميع.
يستخدم الفولاذ 316L على نطاق واسع في تطبيقات العمليات الحيوية لأسباب هندسية جيدة، لكنه يصبح الخيار الصحيح فقط عندما يغير المخاطر الصحية الحقيقية للتجميع الدقيق قيد المراجعة. إنه ذو قيمة لأنه يدعم هامش تآكل أقوى، ومحاذاة أفضل مع التصنيع الصحي الملحوم، وخط أساس أكثر قابلية للدفاع في أنظمة العمليات الحيوية ذات العواقب الأعلى. لكنه ليس بديلاً عن الهندسة الجيدة، وتنفيذ اللحام القوي، واستراتيجية الختم الصحيحة، أو سلسلة التوثيق النظيفة.
لمجموعة موضوع التكنولوجيا الحيوية الكاملة، قم بربط هذه المقالة مع تصميم الأنابيب عالية النقاء لمرافق التكنولوجيا الحيوية, اعتبارات الصيانة والتحقق لأنظمة الأنابيب الصحية, الفولاذ المقاوم للصدأ 316L المصقول كهربائياً مقابل الفولاذ المقاوم للصدأ 316L القياسي, التوثيق وإمكانية التتبع في مشاريع أنابيب الأدويةو اختيار التركيبات الغذائية لمرافق ممارسات التصنيع الجيدة.
الأسئلة الشائعة
هل مادة 316L مطلوبة دائمًا في التطبيقات الحيوية؟
لا. يستخدم على نطاق واسع كخط أساس عملي للخدمة الحرجة المبتلة في العمليات الحيوية، لكن الاختيار النهائي لا يزال يعتمد على الكيمياء الفعلية، والتعرض للتنظيف، ومتطلبات التشطيب، وكثافة اللحام، والعواقب الصحية.
لماذا يُفضل استخدام الفولاذ المقاوم للصدأ 316L على 304L في أنابيب التكنولوجيا الحيوية؟
السبب الأكثر عملية هو هامش تآكل أقوى. يحسن الموليبدينوم في 316/316L المقاومة في الظروف المعرضة للكلوريدات والتآكل الموضعي، وهو ما يهم في الحلقات، وأراضي الحشيات، والمناطق المجاورة للحام، والتجميعات المحلية ذات التصريف الضعيف.
هل تلغي الفولاذ المقاوم للصدأ 316L الحاجة إلى التلميع الكهربائي؟
لا. التلميع الكهربائي و316L ليسا نفس القرار. يعالج 316L خط أساس السبيكة، بينما يعالج التلميع الكهربائي حالة السطح النهائية حيث تكون هناك حاجة إلى قابلية تنظيف أضيق وثقة في التفتيش.
هل يمكن استخدام الفولاذ المقاوم للصدأ 304L في بعض أنظمة العمليات الحيوية؟
نعم، في بعض المناطق المبررة ذات المخاطر المنخفضة. المفتاح هو ما إذا كانت الخدمة والعواقب الصحية أقل حقاً، وليس ما إذا كان المشروع يريد ببساطة تقليل تكلفة المواد.
ما هي المشاكل التي لا يحلها الفولاذ المقاوم للصدأ 316L؟
لا يحل المشاكل مثل الأرجل الميتة، وضعف قابلية التصريف، وتنفيذ اللحام الضعيف، واستراتيجية الختم غير الصحيحة، أو ضعف إمكانية التتبع. تتطلب هذه المشاكل تصميمًا، وتصنيعًا، ومراقبة دورة الحياة، وليس فقط سبيكة أفضل.
أين يجب أن أُولِي أولوية لـ 316L في مشروع حساس للتكلفة؟
ابدأ بالتجميعات المبللة ذات العواقب الأعلى. في أنظمة العمليات الحيوية، يعني ذلك عادةً حلقات النقل الحرجة، مجمعات توزيع ذات كثافة لحام عالية، نقاط أخذ العينات، واجهات الصمامات، والتجميعات المحلية المعرضة للتنظيف/التعقيم في الموقع.




