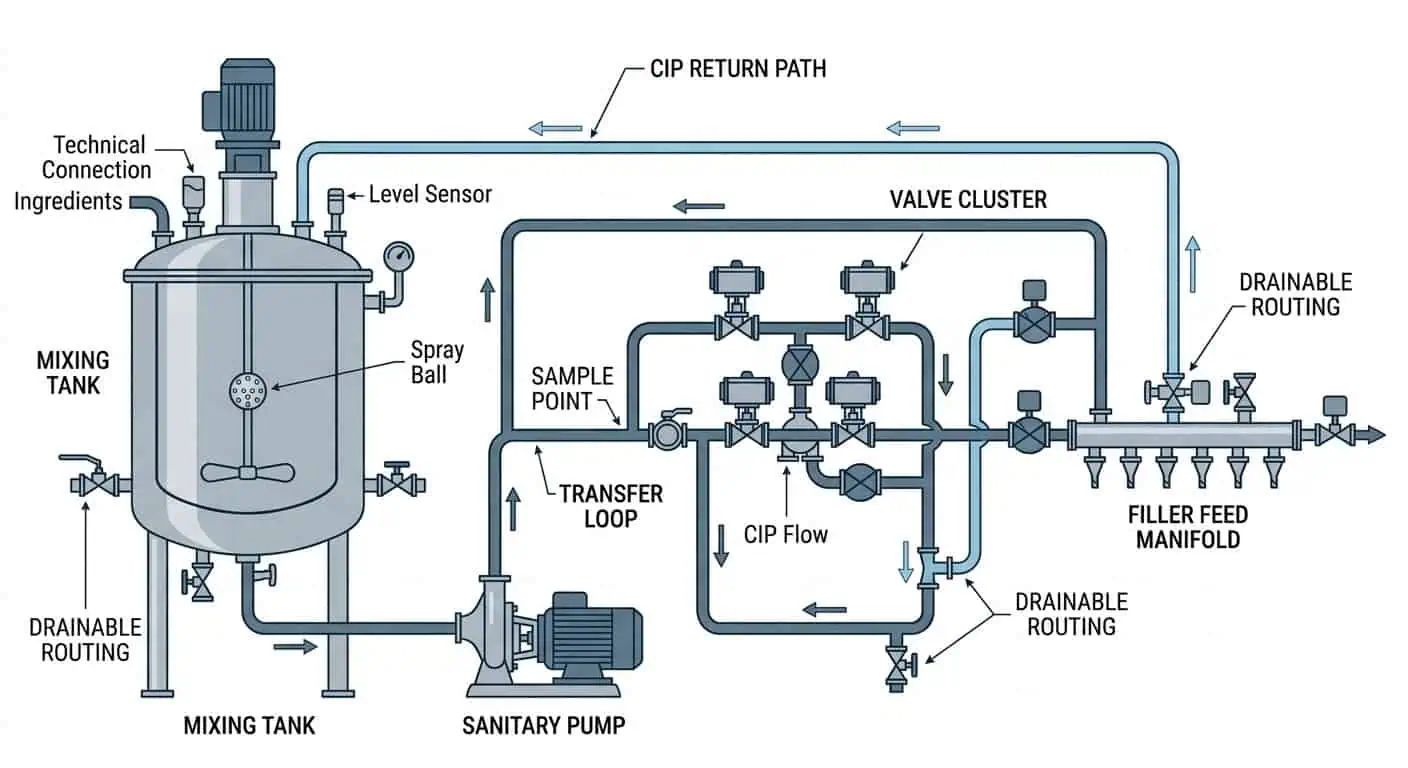
La progettazione di tubazioni alimentari per la produzione cosmetica dovrebbe fare più che spostare il prodotto da un recipiente all'altro. Nella produzione reale, il sistema di tubazioni deve drenare in modo prevedibile, pulire in modo uniforme, evitare l'intrappolamento di residui e ridurre il numero di punti in cui prodotto, acqua di risciacquo o prodotti chimici per la pulizia possono rimanere dopo la produzione o il CIP. Ecco perché gli ingegneri giudicano una linea di processo cosmetica non solo in base al grado del materiale o al tipo di raccordo, ma in base al layout, alla geometria delle ramificazioni, al controllo dei punti bassi, ai dettagli di connessione, alla qualità della saldatura e a come la linea installata si comporta effettivamente durante i cambi di produzione.
Expert Insight:
Molti problemi igienici negli impianti cosmetici non sono causati solo dal grado dell'acciaio inossidabile. Di solito sono creati dalla geometria locale—tronchi morti, scarso drenaggio, sacche a basso flusso, punti di campionamento difficili da pulire o modifiche in campo che cambiano il modo in cui la linea drena e si pulisce.
Se si desidera progettare tubazioni alimentari per la produzione di lozioni, shampoo, creme, sieri, gel o saponi liquidi, l'approccio più utile è iniziare con la pulibilità e la drenabilità, quindi rivedere layout, dettagli delle ramificazioni, selezione delle connessioni, controllo della fabbricazione e comportamento durante il ciclo di pulizia come un unico sistema. La guida GMP della FDA per i cosmetici inquadra le GMP come un modo per minimizzare il rischio di adulterazione e falsa etichettatura, mentre la bozza di guida GMP della FDA afferma che l'agenzia ha considerato la ISO 22716 nell'aggiornamento delle pratiche attuali. Per un ingegnere, ciò si traduce in tubazioni più facili da pulire, ispezionare e controllare nella produzione di routine piuttosto che solo più facili da fabbricare.
Cosa significa la progettazione di tubazioni alimentari nella produzione cosmetica
Perché le linee di processo cosmetico hanno bisogno di più del semplice tubo in acciaio di base
Le linee di produzione cosmetica spesso gestiscono prodotti che sono più impegnativi di quanto appaiano inizialmente. Creme, lozioni, sistemi tensioattivi, gel e formulazioni contenenti fragranze possono lasciare residui, aderire alle superfici, intrappolare aria in punti alti locali o comportarsi diversamente durante lo spegnimento e il riavvio rispetto a un fluido a bassa viscosità. Una linea che sembra accettabile sulla carta può comunque diventare difficile da risciacquare, lenta da verificare dopo la pulizia o vulnerabile alla diluizione del primo lotto se il routing e la geometria delle ramificazioni non supportano il servizio igienico.
- Protezione del prodotto: la linea dovrebbe ridurre la ritenzione di residui e le opportunità di contaminazione.
- Drenaggio completo: il prodotto e la soluzione di pulizia non dovrebbero rimanere in punti bassi nascosti.
- Pulizia prevedibile: il sistema dovrebbe rispondere in modo coerente alla routine effettiva di pulizia CIP o manuale dell'impianto.
- Cambi di produzione ripetibili: il cambio di prodotti non dovrebbe dipendere da congetture o tempi di recupero prolungati.
Obiettivi di progettazione che gli ingegneri dovrebbero stabilire prima del tracciamento della linea
Prima che il primo spezzone venga tracciato, gli ingegneri dovrebbero definire cosa deve ottenere la tubazione in esercizio. Nel servizio cosmetico, i principali obiettivi di progettazione sono solitamente pulibilità, drenabilità, basso volume di ritenzione, ridotta esposizione a tratti morti, accesso per ispezione e manutenzione pratica. Se questi obiettivi non vengono inseriti nella revisione del progetto in anticipo, il progetto spesso compensa in seguito utilizzando pulizie più aggressive, cambi di produzione più lunghi o ripetute modifiche in campo.
ISO 22716 è utile qui perché copre produzione, controllo, stoccaggio e spedizione per prodotti cosmetici, non solo l'aspetto materiale del processo. Questa logica GMP più ampia si adatta bene alle revisioni delle tubazioni alimentari: la linea fa parte del controllo del processo, non solo di un'utilità di trasporto.
Questo è anche il motivo per cui le tubazioni alimentari dovrebbero essere riviste insieme a Acciaio Inossidabile 316L per Linee di Produzione di Cura Personale. La scelta del materiale può supportare le prestazioni igieniche, ma non può compensare un percorso debole o una progettazione di diramazione scadente.
Come la Disposizione del Processo Influisce sulla Pulibilità e sulle Prestazioni di Cambio
Progettare in Base al Flusso del Prodotto, Non Solo allo Spazio Disponibile
Uno degli errori di disposizione più comuni è il percorso delle linee di prodotto attorno allo spazio aperto del pavimento invece che attorno alla logica del flusso igienico. Negli impianti cosmetici, ciò di solito crea cambi di elevazione non necessari, lunghezze extra dei tratti di tubazione e zone locali di ritenzione che aumentano la perdita di prodotto e il carico di pulizia. Una buona disposizione alimentare inizia dal percorso reale del processo—miscelazione, trasferimento, ritenzione, riempimento e ritorno—non da qualsiasi percorso sembri più facile durante la disposizione delle tubazioni.
Perché i Percorsi del Prodotto Più Brevi e Semplici Di Solito Funzionano Meglio
I percorsi di contatto con il prodotto più brevi di solito riducono sia il rischio igienico che la perdita operativa. Un circuito più lungo aumenta la superficie, il volume residuo, la domanda di pulizia e il numero di giunti o transizioni che devono funzionare correttamente. Questo è ancora più importante nella produzione cosmetica multi-SKU, dove i cambi ripetuti rendono ogni tasca extra o sezione a risciacquo lento più visibile nel tempo.
In un progetto di trasferimento di lozioni, un percorso di ricircolo era stato instradato aggirando un'interferenza strutturale invece di essere semplificato vicino al gruppo di serbatoi. Il risultato non è stato un guasto immediato, ma un drenaggio più lungo, un maggior ritenimento di prodotto e un recupero post-pulizia più lento. Dopo che il percorso è stato accorciato e una variazione di quota non necessaria è stata rimossa, sia il recupero della linea che la coerenza del cambio sono migliorati.
Suggerimento:
Se una linea a contatto con il prodotto segue un percorso più lungo solo perché “il tubo può essere posizionato lì”, consideratelo come un avviso di progettazione. In servizio alimentare, l'instradamento per convenienza spesso si ripresenta successivamente come residuo, ritardo nel risciacquo o perdita di prodotto non necessaria.
Le decisioni di layout dovrebbero anche essere riviste insieme a Considerazioni sulla Pulizia nei Sistemi di Processo Igienici, perché le prestazioni di pulizia sono fortemente influenzate dalla lunghezza della linea, dalla logica di ritorno e dal numero di punti in cui il flusso diventa debole o stagnante.
Punti Morti, Rami e Tasche a Basso Flusso: Dove le Linee Alimentari Solitamente Falliscono Prima
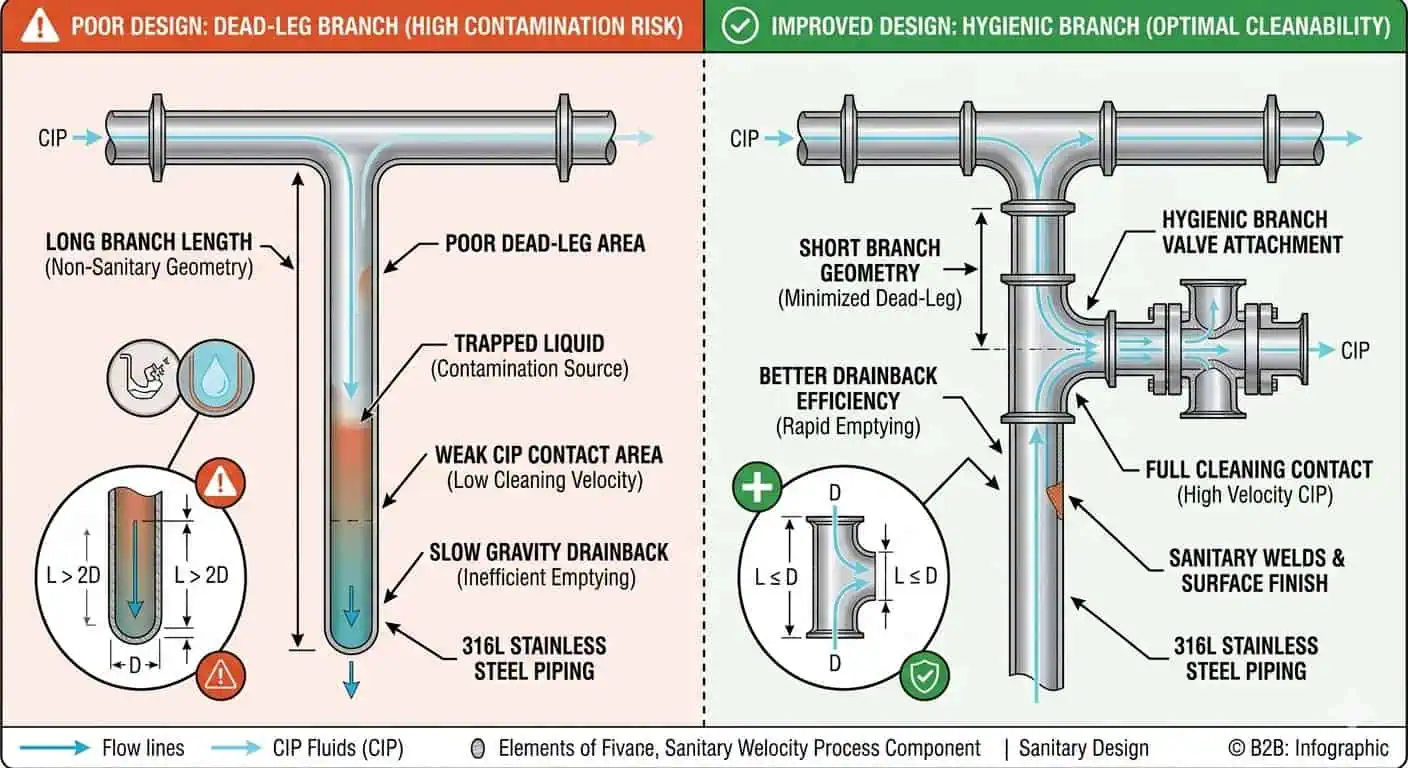
Perché i Punti Morti Contano Più nel Servizio Cosmetico di Quanto Molti Acquirenti Si Aspettano
I punti morti sono uno dei rischi igienici più persistenti nelle tubazioni alimentari perché creano zone di intrappolamento e una debole risposta alla pulizia. Lo Studio ASME sui Rami per Attrezzature di Bioprocessing definisce un punto morto come un'area di intrappolamento in un recipiente o in una tubazione che potrebbe portare alla contaminazione del prodotto, e nota che più studi supportano la progettazione con un rapporto L/D inferiore a due perché la pulizia dipende dal contatto effettivo tra la soluzione CIP e il ramo. La stessa logica ingegneristica è altamente rilevante nel servizio cosmetico ogni volta che i prodotti sono viscosi, soggetti a residui o soggetti a frequenti cambi.
Orientamento del Rame, Lunghezza e Drenaggio Devono Essere Rivisti Insieme
Gli ingegneri non dovrebbero valutare una diramazione solo come “presente” o “non presente”.” Le vere domande sono quanto è lunga, dove punta, se drena naturalmente e se crea liquido intrappolato o aria intrappolata quando il sistema si ferma. Una diramazione può rimanere accettabile dal punto di vista igienico solo se la sua geometria corrisponde al reale funzionamento e comportamento di pulizia della linea.
- Punti di campionamento: spesso diventano ad alto rischio se aggiunti in ritardo senza una revisione del drenaggio.
- Tee per strumenti: possono diventare tasche a lento risciacquo in servizio a contatto con il prodotto.
- Prelievi superiori: possono intrappolare liquido o aria se il percorso e il comportamento in arresto vengono ignorati.
- Gruppi di valvole e collettori: spesso creano i primi problemi igienici locali nelle linee multi-prodotto.
Posizioni ad Alto Rischio Comuni nelle Tubazioni Cosmetiche
La maggior parte dei riscontri igienici ripetuti appare nei dettagli locali, non nel tubo rettilineo. Le posizioni ad alto rischio di solito includono collettori di alimentazione del riempitivo, derivazioni di campionamento, rami del sensore, tasche delle valvole, connessioni superiori dei rami e modifiche temporanee che non sono mai state completamente riviste come parte del progetto igienico permanente.
Una linea di riempimento crema ha fornito un esempio utile. Il materiale e la finitura della linea principale erano accettabili, ma una connessione di campionamento lato prodotto aggiunta in ritardo utilizzava una geometria che tratteneva residui e rallentava il drenaggio. Il problema non è stato risolto cambiando la classe del materiale. È stato risolto sostituendo la connessione con una derivazione alimentare che corrispondeva al layout igienico circostante e riducendo la zona di ristagno locale.
Questo è anche il motivo per cui il progetto del ramo dovrebbe essere rivisto insieme a Raccordi Tubo Alimentari vs Industriali. Una linea può utilizzare la lega di acciaio inossidabile corretta e comunque sottoperformare se la geometria del raccordo non è veramente adatta per la pulizia igienica e il drenaggio.
Expert Insight:
Uno dei più comuni errori di giudizio ingegneristici è presumere che la corsa principale controlli le prestazioni igieniche. Negli impianti reali, i primi problemi di sanificazione ripetuti hanno maggiori probabilità di provenire da rami, transizioni locali e dettagli di connessione difficili da pulire.
Pendenza, Drenabilità e Progetto del Punto Basso
La Drenabilità Inizia con l'Instradamento, Non con la Valvola di Scarico Finale
Una linea igienica non diventa drenabile solo perché ha uno scarico alla fine. La vera drenabilità inizia con il percorso, la pianificazione dell'elevazione, il posizionamento dei supporti, l'orientamento delle bocche dell'attrezzatura e come il tubo installato si comporta effettivamente dopo il montaggio in campo. Un punto basso che esiste solo sul disegno non è una vera caratteristica igienica se la linea installata si affloscia altrove o crea una falsa tasca vicino a un supporto.
Perché lo scarso drenaggio rallenta la pulizia e aumenta il rischio di riavvio
Lo scarso drenaggio fa più che lasciare liquido dietro. Può aumentare il tempo del ciclo di pulizia, diluire il lotto successivo del prodotto, intrappolare la soluzione di pulizia e rendere le condizioni di riavvio meno prevedibili. Negli impianti per la cura personale, ciò si manifesta spesso come verifica di risciacquo prolungata, qualità instabile del primo lotto o attenzione ripetuta alla stessa area a basso punto dopo le fermate.
Cosa dovrebbero controllare gli ingegneri nei punti bassi e nei cambi di elevazione
| Controllo di progettazione | Cosa rivedere | Perché è importante |
|---|---|---|
| Continuità della pendenza | Confermare che la caduta prevista sia mantenuta in tutte le sezioni dei tratti | Le interruzioni nella pendenza possono creare falsi punti bassi e liquido trattenuto |
| Posizionamento del supporto | Verificare se i supporti installati possono causare cedimenti o ristagni locali | La capacità di drenaggio può essere persa dopo l'installazione anche se il disegno sembrava corretto |
| Transizione apparecchiatura-tubo | Rivedere l'elevazione, l'orientamento e la geometria di transizione del bocchello | Molte zone di prodotto trattenuto iniziano alle interfacce del recipiente o dello skid |
| Bilanciamento del collettore | Verificare se le diramazioni drenano in modo uniforme | I punti bassi irregolari possono rallentare la pulizia e la verifica di riavvio |
ASTM A380 è particolarmente rilevante per questa revisione perché afferma che la progettazione dovrebbe minimizzare le aree dove sporco o soluzioni detergenti potrebbero rimanere intrappolati e dovrebbe garantire una circolazione e rimozione efficace delle soluzioni detergenti. Questo principio supporta direttamente i controlli dei punti bassi, della pendenza e del drenaggio.
In un impianto di ricircolo shampoo, la ripetuta decolorazione e il ritardo nel risciacquo sono stati infine ricondotti a un punto basso locale vicino a un gruppo di valvole che non era evidente nel modello originale. Il lavoro correttivo si è concentrato sulla modifica dell'elevazione del tratto di tubazione e sul miglioramento del drenaggio piuttosto che sulla modifica dell'intera specifica del materiale della linea.
Selezione Connessioni: Morsetti, Valvole, Strumenti e Punti di Campionamento
Non Ogni Connessione Alimentare Si Comporta Allo Stesso Modo nei Cicli di Pulizia Reali
“Alimentare” non è una garanzia di prestazione universale per ogni dettaglio di connessione. Giunti a morsetto, valvole, strumenti e punti di campionamento possono essere tutti igienicamente accettabili nella posizione corretta, ma non si comportano in modo identico sotto ripetuti cambi di prodotto, arresti a umido ed esposizione alla pulizia. La scelta migliore della connessione dipende dalla pulibilità, dalle esigenze di smontaggio, dalla sensibilità del prodotto e se la posizione è un vero punto di rischio di contatto con il prodotto.
Dove le Connessioni per Strumenti e Campionamento Comuni Creano Rischio Igienico
I punti di strumentazione e campionamento sono spesso sottovalutati perché sembrano piccoli rispetto alla tubazione principale. In realtà, possono diventare le prime posizioni in cui compaiono residui, flusso debole o superfici difficili da ispezionare. Tee per manometri, tasche per sonde di temperatura, valvole di campionamento, transizioni per vetri di ispezione e rami di riempimento dovrebbero tutti essere trattati come dettagli critici di progettazione a contatto con il prodotto quando sono posizionati su linee igieniche.
Perché i Dettagli Filettati a Contatto con il Prodotto Dovrebbero Essere Minimizzati
I dettagli filettati a contatto con il prodotto dovrebbero essere minimizzati perché introducono geometrie simili a fessure, gradini locali e difficoltà di ispezione. Nel servizio cosmetico igienico, la comodità durante l'installazione raramente giustifica un rischio aggiuntivo di prodotto trattenuto sul lato bagnato. Se un dettaglio filettato appare in ritardo nel progetto, dovrebbe essere rivisto come una modifica igienica, non solo come un piccolo aggiustamento meccanico.
Una lezione utile sul campo è arrivata da un banco di riempimento creme e sieri dove una connessione per campioni in ritardo è stata aggiunta utilizzando un dettaglio filettato sul lato del prodotto per risparmiare tempo di fabbricazione. Il risultato è stata un'attenzione ripetuta alla sanificazione nella stessa posizione fino a quando la connessione è stata sostituita con un'alternativa igienica più adatta alla geometria della linea circostante.
Queste scelte di connessione dovrebbero anche essere riviste rispetto a Acciaio Inossidabile 316L per Linee di Produzione di Cura Personale, perché una scelta di materiale più resistente non può comunque rimuovere il rischio igienico creato dal dettaglio di connessione a contatto con il prodotto errato.
Saldatura, Finitura Superficiale e Controllo della Fabbricazione nelle Tubazioni Igieniche
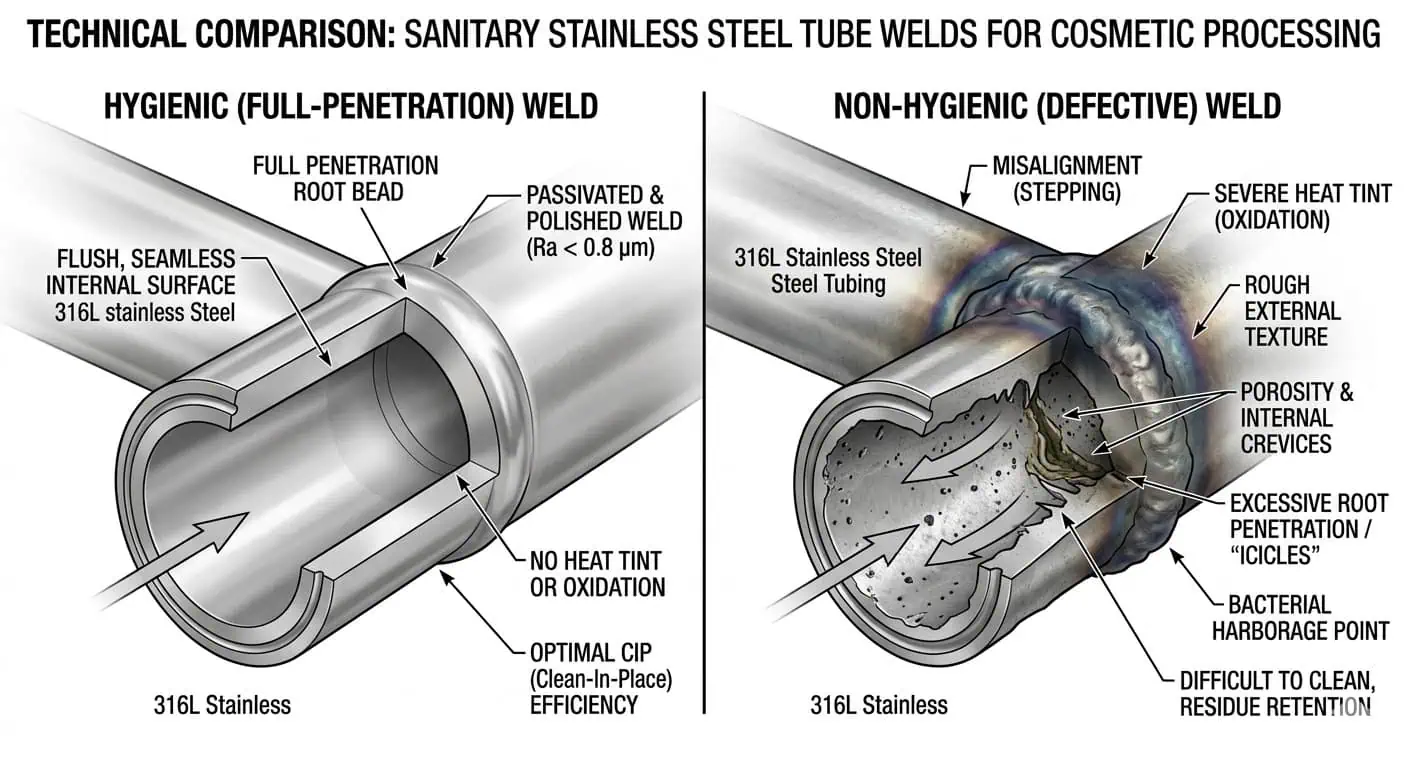
Un Buon Design Igienico Può Essere Perso Durante la Fabbricazione
Un layout corretto sul disegno non garantisce una linea igienica dopo la fabbricazione. Saldature grezze, disallineamento interno, colorazione da calore, modifiche sul campo e un montaggio scadente possono cambiare la pulibilità, il rilascio di residui e il comportamento di drenaggio locale. Ecco perché la qualità della fabbricazione dovrebbe essere trattata come parte del risultato del design sanitario, non come un problema separato dell'officina.
Perché la Finitura Superficiale Dovrebbe Essere Specificata come un Requisito di Prestazione
La finitura superficiale è importante perché influisce sulla facilità di pulizia e ispezione del lato a contatto con il prodotto. In servizi igienici, la condizione superficiale non è solo uno standard visivo. Influenza il rilascio di residui, il recupero del risciacquo, il rischio di contaminazione locale e la ripetibilità con cui la linea ritorna a uno stato pulito dopo la produzione. Le linee guida di progettazione igienica 3-A indicano comunemente superfici a contatto con il prodotto almeno lisce come 32 µin Ra / 0,8 µm, comprese le giunzioni saldate, e prive di cavità, pieghe, crepe, fessure e disallineamenti. Se il requisito di finitura è lasciato vago, i fornitori potrebbero comunque fornire materiale tecnicamente inossidabile ma operativamente meno adatto all'igiene a contatto con il prodotto.
La passivazione e la pulizia post-produzione non devono essere trattate come clausole opzionali
La pulizia post-produzione e la passivazione devono essere definite come parte della condizione igienica consegnata. La norma ASTM A967 copre diversi trattamenti chimici di passivazione per parti in acciaio inossidabile e include test alternativi con criteri di accettazione per confermare l'efficacia del trattamento. Una linea di processo cosmetica può lasciare l'officina apparentemente completa pur presentando ancora scolorimenti, contaminazioni o incoerenze di finitura che riducono le prestazioni igieniche a lungo termine. Gli ingegneri devono definire come deve apparire il lato a contatto con il prodotto alla consegna, non solo quale grado di metallo è stato acquistato.
- Esecuzione della saldatura: il profilo interno e la continuità influenzano la pulibilità.
- Coerenza della finitura: la rugosità locale può creare punti di attenzione ripetuti per la pulizia.
- Controllo della risaldatura in campo: Le riparazioni devono essere esaminate con la stessa disciplina della fabbricazione originale.
- Trattamento post-fabbricazione: La portata della pulizia e della passivazione deve essere chiaramente definita.
Expert Insight:
La parte pericolosa di una saldatura scadente non è che sembra disordinata. È che cambia la geometria locale, le condizioni della superficie e il modo in cui la linea di contatto con il prodotto può effettivamente essere pulita.
Progettazione di tubazioni per CIP e ripetuti cambi di prodotto
Una linea alimentare dovrebbe essere facile da pulire nel modo in cui l'impianto opera effettivamente
La progettazione favorevole alla pulizia dovrebbe basarsi sul comportamento reale dell'impianto, non su ipotesi idealizzate. Alcune linee cosmetiche eseguono lunghe campagne di un prodotto. Altre cambiano formulazioni più volte alla settimana, si fermano bagnate nei fine settimana o eseguono lotti brevi dove il rapido recupero è più importante della portata assoluta. La progettazione delle tubazioni dovrebbe corrispondere a quel modello operativo reale.
Perché la frequenza di cambio dovrebbe influenzare la progettazione delle tubazioni in anticipo
L'alta frequenza di cambio rende ogni debolezza locale più visibile. Una piccola zona di ristagno può essere tollerabile in una lunga campagna di prodotto singolo, ma diventa un costo persistente e un problema igienico in una linea che cambia frequentemente tra formulazioni di lozione, shampoo, crema e gel. Ecco perché l'aspettativa di cambio dovrebbe influenzare il percorso, la lunghezza del ramo, il design del collettore e la scelta della connessione prima che inizi l'approvvigionamento.
Copertura CIP, Logica di Ritorno e Semplicità del Percorso di Flusso
Un dispositivo a spruzzo o un circuito CIP da solo non garantisce la pulibilità. Il percorso di ritorno, la semplicità del flusso, l'esposizione del ramo e il comportamento della velocità locale sono tutti importanti. Lo studio sul ramo di ASME lo dimostra chiaramente: senza il contatto delle soluzioni CIP, non c'è pulizia. In un sistema multi-SKU per lozioni, la linea non ha avuto difficoltà a causa della selezione del materiale di base. Ha avuto difficoltà perché il percorso di ritorno era più complesso di quanto il team di produzione si rendesse conto, lasciando una sezione costantemente più lenta a riprendersi dopo la pulizia. Semplificare il circuito e riclassificare un ramo come ad alto rischio ha prodotto un risultato migliore rispetto all'aumento del solo tempo di pulizia.
Per maggiori dettagli su questo aspetto operativo delle prestazioni igieniche, la revisione del design dovrebbe collegarsi direttamente a Considerazioni sulla Pulizia nei Sistemi di Processo Igienici.
Una Lista di Controllo Pratica per la Revisione del Design delle Tubazioni Alimentari Cosmetiche
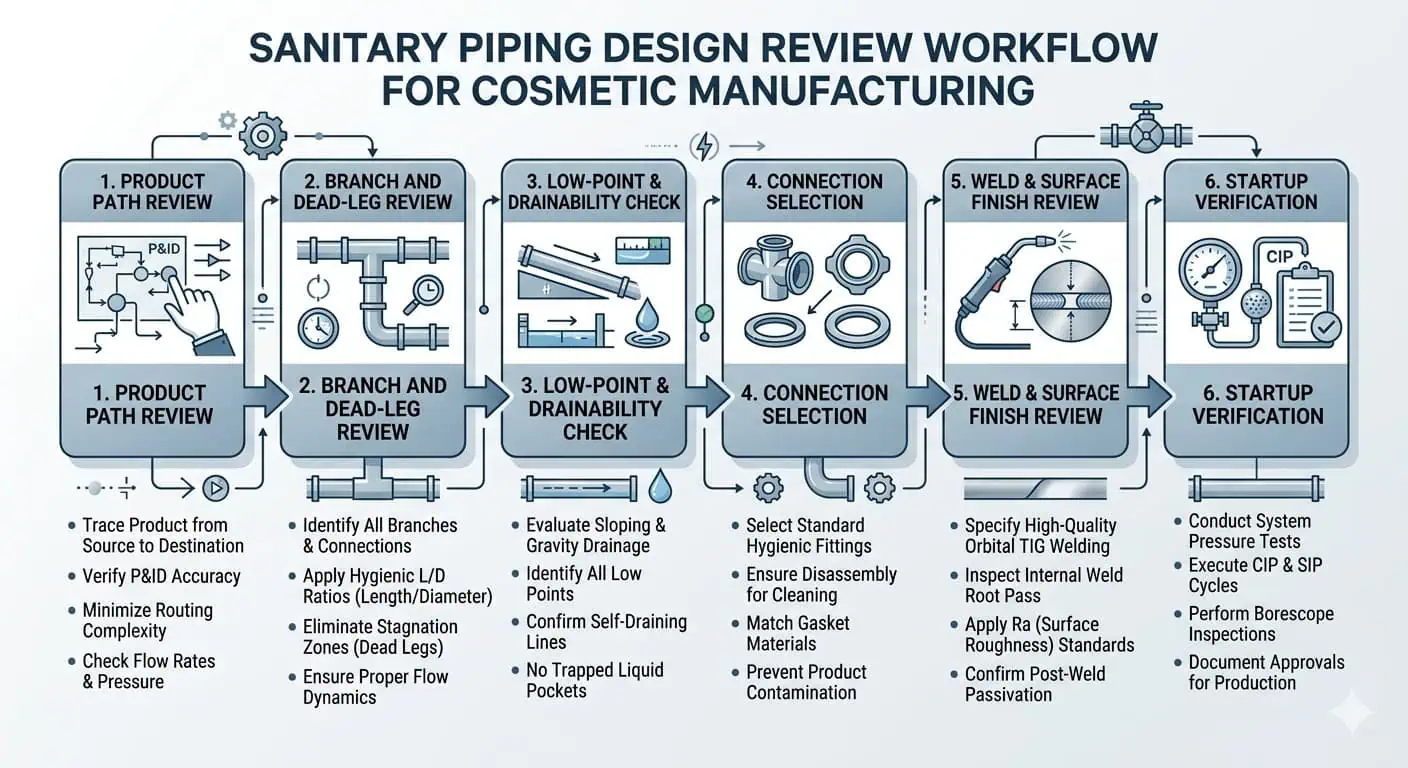
Domande da Porre Prima di Finalizzare il Layout
- Ogni ramo a contatto con il prodotto è davvero giustificato e drenabile igienicamente?
- Dove può rimanere residuo dopo lo spegnimento o il drenaggio parziale?
- Ci sono tratti morti, tasche cieche o falsi punti bassi evitabili?
- Il pendio previsto sarà ancora presente dopo l'installazione dei supporti e il montaggio in campo?
- I punti di campionamento e strumentazione corrispondono alla sensibilità igienica del prodotto effettivo?
Domande da Porre Prima di Rilasciare RFQ
| Argomento RFQ | Cosa Dovrebbe Essere Definito |
|---|---|
| Ambito dei materiali | Chiarire quali parti sono veri componenti a contatto con il prodotto |
| Finitura superficiale | Definire la finitura igienica richiesta per le aree bagnate |
| Qualità della saldatura | Specificare le aspettative per la continuità interna e la gestione delle riparazioni in campo |
| Trattamento post-fabbricazione | Specificare l'ambito di pulizia/passivazione dove richiesto |
| Standardizzazione delle connessioni | Ridurre i dettagli non standard che creano lacune nella revisione igienica |
| Revisione della drenabilità | Richiedere conferma che il percorso installato supporti il drenaggio igienico |
Domande da porre prima dell'avvio
La revisione di avvio dovrebbe confermare che la linea installata corrisponda ancora all'intento progettuale. Verificare la pendenza effettiva, i punti bassi reali, le modifiche in campo, le connessioni aggiunte in ritardo e se eventuali raccordi temporanei siano rimasti sul lato a contatto con il prodotto. Una linea che era igienicamente razionale durante la progettazione può comunque perdere questa qualità durante l'installazione se questi controlli vengono saltati.
Suggerimento:
Trattare le modifiche tardive del progetto vicino a ramificazioni a contatto con il prodotto, collettori e punti di campionamento come modifiche di progettazione igienica, non come semplici comodità di fabbricazione. Queste sono le posizioni più probabili per creare problemi di sanificazione ripetuti in seguito.
Errori di progettazione comuni nelle tubazioni alimentari cosmetiche
Trattare il grado dell'acciaio inossidabile come sostituto di una buona progettazione
Aggiornare il materiale non corregge una geometria scadente. Se la linea trattiene liquidi, intrappola prodotto o crea transizioni difficili da pulire, una lega superiore da sola non renderà il sistema alimentare.
Aggiunta di diramazioni in ritardo senza revisione della pulibilità
Punti di campionamento tardivi, raccordi a T per strumenti e modifiche temporanee sono fonti comuni di problemi ripetuti. Una diramazione aggiunta per comodità spesso aggira la revisione alimentare originale.
Utilizzo di connessioni convenienti in posizioni di contatto con il prodotto errate
La facilità di montaggio non deve prevalere sulla pulibilità. Particolari filettati a contatto con il prodotto e transizioni locali scelte in modo inappropriato possono creare più problemi a lungo termine di quanto risparmino durante la fabbricazione.
Ignorare il drenaggio dopo supporti e installazione
La linea installata, non il modello, determina le prestazioni igieniche. Abbassamento dei supporti, falsi punti bassi e modifiche di adattamento in campo possono rimuovere silenziosamente la drenabilità prevista dal progetto.
Consentire alle riparazioni in campo di bypassare gli standard di finitura igienica
Le risaldate in campo e le riparazioni locali devono essere controllate come lavori a contatto con il prodotto. Un sistema igienico può essere indebolito rapidamente se la qualità della riparazione viene trattata come una preoccupazione secondaria.
Domande frequenti sulla progettazione di tubazioni alimentari per cosmetici
Qual è la regola più importante nella progettazione di tubazioni alimentari per la produzione cosmetica?
La regola più importante è progettare fin dall'inizio per la pulibilità e lo scaricabilità. Una linea di processo per cosmetici non dovrebbe solo trasportare il prodotto; dovrebbe anche evitare intrappolamenti, supportare una pulizia prevedibile e minimizzare il numero di dettagli locali che possono trattenere prodotto o soluzione di pulizia.
Perché i tratti morti sono importanti nelle linee per lozioni, creme e shampoo?
Le gambe morte sono importanti perché i prodotti viscosi e soggetti a residui rendono più visibili le tasche a risciacquo lento. Queste aree possono ritardare il recupero della pulizia, trattenere il prodotto e creare punti di attenzione ripetuti per la sanificazione durante i cambi di produzione.
L'utilizzo dell'acciaio 316L rende automaticamente un sistema di tubazioni alimentare?
No. L'acciaio 316L può migliorare il margine di corrosione e supportare il servizio igienico, ma non può correggere da solo un percorso scadente, una geometria di diramazione errata, un controllo di saldatura debole o una scaricabilità inadeguata.
Come devono essere progettati i punti di campionamento nelle tubazioni alimentari per cosmetici?
I punti di campionamento devono essere mantenuti giustificati igienicamente, scaricabili, facili da pulire e coerenti con la progettazione a contatto con il prodotto circostante. Le connessioni di campionamento aggiunte in ritardo o per comodità spesso creano rischi igienici locali se non vengono esaminate correttamente.
Perché una linea alimentare può ancora fallire la pulizia anche quando la disposizione sembra corretta sulla carta?
Perché il sistema installato potrebbe comportarsi diversamente dal modello. L'abbassamento dei supporti, i falsi punti bassi, le risaldature in campo, le transizioni locali e i dettagli dei rami possono tutti modificare il modo in cui la linea si scarica e risponde durante la pulizia.
Le connessioni a morsetto sono sempre l'opzione migliore per servizi cosmetici igienici?
No. Le connessioni a morsetto sono utili in molti sistemi alimentari, ma la connessione giusta dipende ancora dalla pulibilità, dalle esigenze di manutenzione, dalla geometria locale e dal fatto che la posizione sia un vero punto di rischio di contatto con il prodotto.
Considerazione Finale di Ingegneria
La progettazione di tubazioni alimentari per la produzione cosmetica è una disciplina di pulibilità e scaricabilità prima di diventare una questione di materiale o approvvigionamento. Una linea che funziona bene di solito deriva da percorsi del prodotto più brevi e semplici, geometria dei rami controllata, scaricamento affidabile, connessioni giustificate igienicamente, saldatura disciplinata e una revisione realistica di come il sistema installato verrà pulito e riavviato. Quando queste scelte progettuali vengono gestite in anticipo, è più probabile che la linea funzioni in modo coerente durante la produzione, la pulizia e il cambio prodotto—non solo durante l'avvio iniziale, ma per tutta la sua vita operativa.
Expert Insight:
Gli ingegneri spesso chiedono quale materiale, raccordo o finitura renderà igienica una linea cosmetica. La domanda più pratica è se la linea è stata progettata in modo che il prodotto e la soluzione di pulizia possano lasciare il sistema in modo altrettanto prevedibile di come vi sono entrati.
Se stai esaminando una linea per lozioni, shampoo, creme, gel o prodotti per la cura personale multi-prodotto, inizia mappando il percorso reale del prodotto, quindi identifica ogni ramo, punto basso, connessione di campionamento e modifica in campo che potrebbe influenzare la scaricabilità o la coerenza della pulizia.
Letture Correlate
- Acciaio Inossidabile 316L per Linee di Produzione di Cura Personale
- Raccordi Tubo Alimentari vs Industriali
- Considerazioni sulla Pulizia nei Sistemi di Processo Igienici
- Soluzioni per l'Industria dei Cosmetici e della Cura Personale
- Linee Guida FDA GMP per Cosmetici / Lista di Controllo Ispezione
- Linee Guida ISO 22716 GMP per Cosmetici
- Studio ASME per Gambo di Diramazione per Attrezzature di Bioprocessamento
- ASTM A380 Linee Guida per Pulizia, Decapaggio, Sgrassaggio e Passivazione
- ASTM A967 Trattamenti Chimici di Passivazione per Acciaio Inossidabile




